alcohold & ethers
0.0(0)
Studied by 0 peopleCard Sorting
1/5
Earn XP
Description and Tags
some stuff idk whats going on and if it even pertains to alcohol & ethers
Last updated 10:45 PM on 4/28/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
1
New cards
prepping alcs
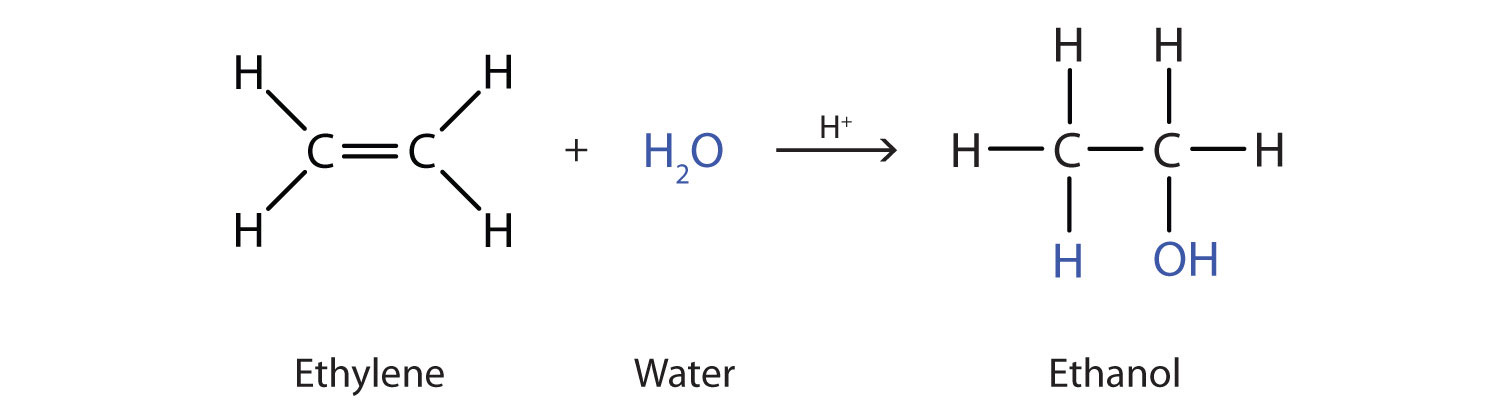
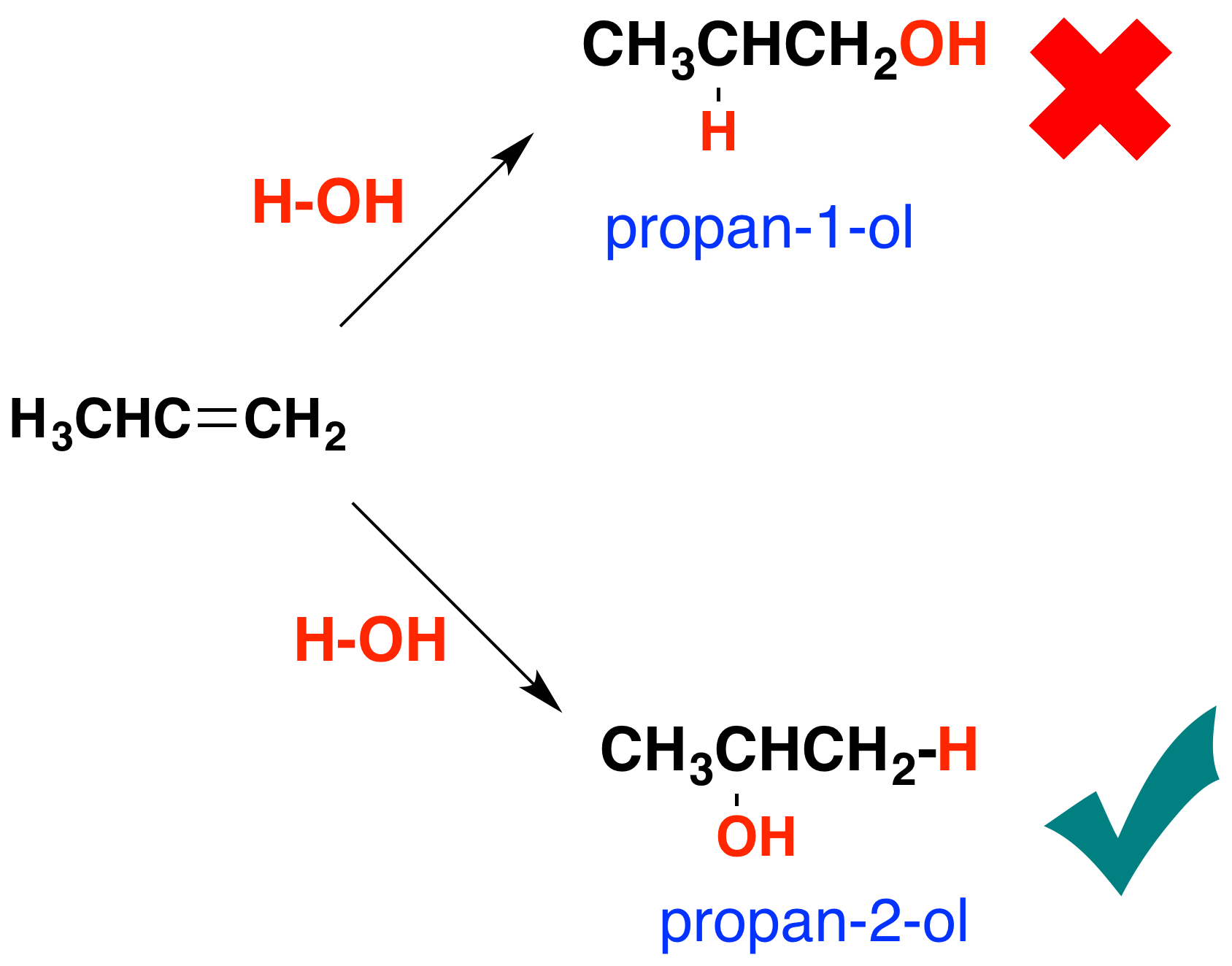
addition reaction of OH to carbon with last number of hydrogen
2
New cards
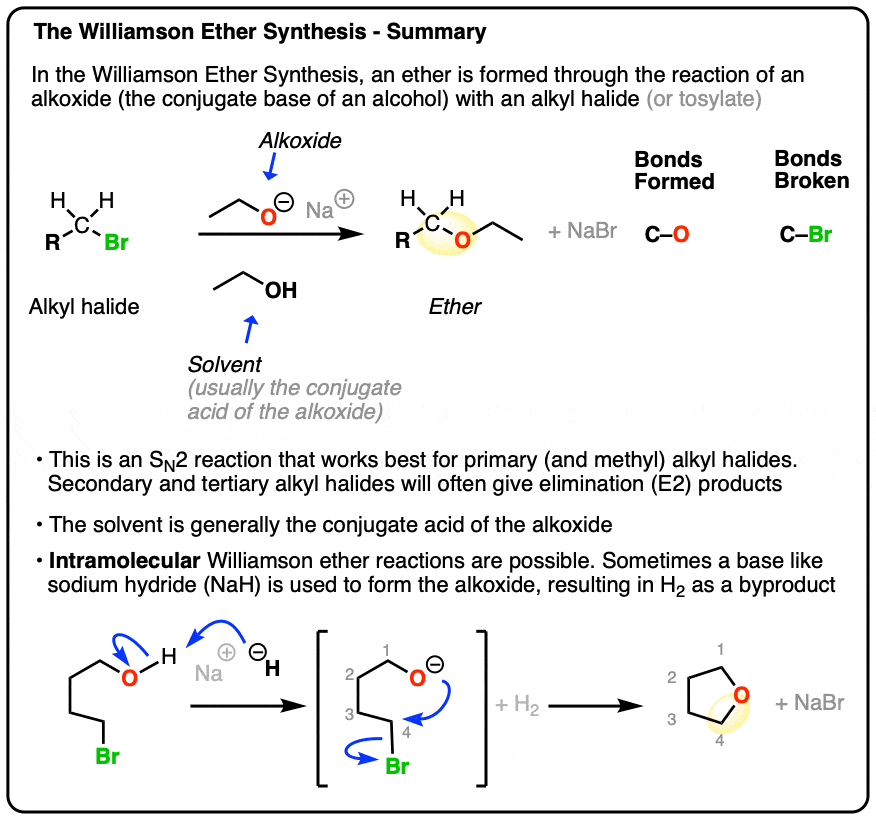
ethers ( functional group )
cannot form H-bonds but introduce polarity
2 alcs react in the presence of H2SO4. Elimination, water removed (condensation)
-oxy added to prefix of smaller HC group and joining it to alkane name of larger HC group
3
New cards
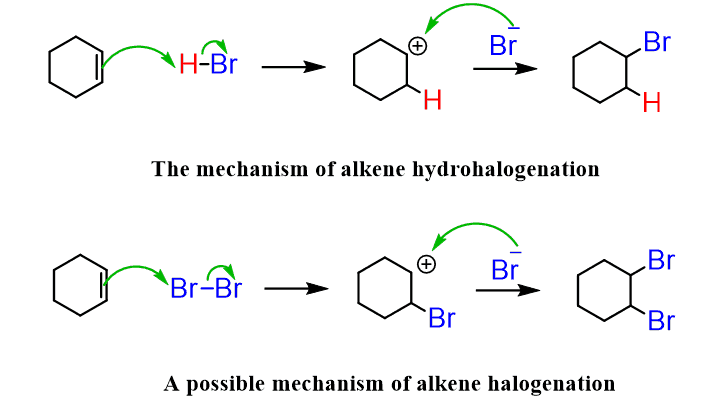
halogenation
halogen + alkene/alkyne ; addition
4
New cards
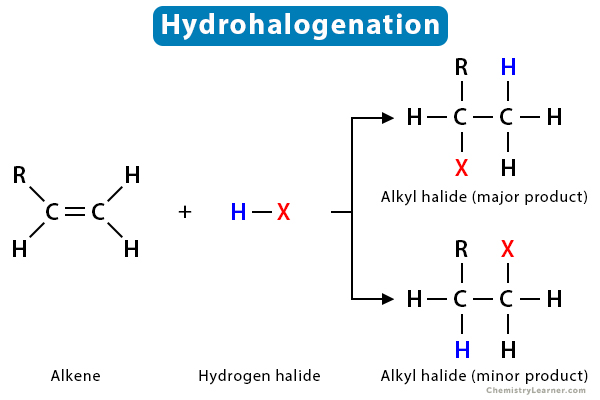
hydrohalogenation
H bonded to halogen (eg. HF, HCL)
5
New cards
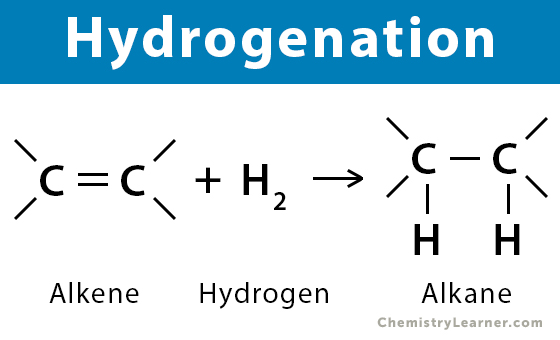
hydrogenation
H bonded to organic molecule
6
New cards
hydration
adding H2O to organic molecule