Basic concepts organic chemistry
1/11
Earn XP
Description and Tags
Chapter 11
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Application of IUPAC rules of nomenclature
Find the longest carbon chain → parent name.
Number the chain for the lowest locants.
Highest priority functional group = suffix (
-ol,-oic acid, etc.).Side groups/substituents = prefixes (
methyl-,chloro-, etc.).Write substituents alphabetically with their positions.
Use commas between numbers and hyphens between numbers/words.
Combine into the full IUPAC name.
General, structural and displayed formula
General: the simplest algebraic formula of a member of a homologous series (e.g. alkane - CnH2n+2)
Structural: the minimal detail that shows the arrangement of atoms in a molecule (e.g. butane - CH3CH2CH2CH3 or CH3(CH2)2CH3)
Displayed formula: the relative positioning of atoms and the bonds between them (e.g. alkene functional group C=C)
Skeletal formula
Simplified organic formula shown by removing the hydrogen atoms from alkyl chains, leaving carbon skeleton and functional groups
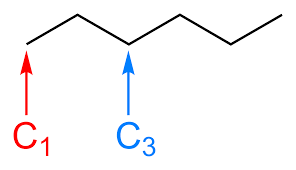
Homologous series
A series of organic compounds having the same functional group but with each successive member differing by CH2
R may be used to represent alkyl groups, but also other fragments of organic compounds not involved in reactions.
Functional group
A group of atoms responsible for the characteristic reactions of a compound
Saturated compound contains just C-C bonds and unsaturated contains C=C, C triple bond C and aromatic rings
Degree of saturation
The degree of saturation describes how many rings or multiple bonds are present in a compound.
More multiple bonds/rings → higher degree of saturation
Includes:
double bonds (C=C)
triple bonds (C≡C)
cyclic/ring structures
Alkyl group
Alkyls are the main hydrocarbon chain or molecule (-1 hydrogen) as a substituent with the formula of CnH2n+1
For example, methyl (CH3) is alkyl of methane (CH4)
Aliphatic, alicyclic and aromatic
Aliphatic (a compound containing carbon and hydrogen joined together in straight chains, branched chains or non-aromatic rings)
Alicyclic (an aliphatic compound arranged in non-aromatic rings with or without side chains)
Aromatic (a compound containing a benzene ring)
Structural isomer
Compounds with the same molecular formula but different structural formulae
Chain isomers → different carbon chain arrangement
Position isomers → same groups, different positions
Functional group isomers → different functional groups
Covalent bond fission
Homolytic fission (in terms of each bonding atom receiving one electron from the bonded pair, forming two radicals)
Heterolytic fission (in terms of one bonding atom receiving both electrons from the bonded pair)
Radical
A species with an unpaired electron, highly reactive, represented with a dot (Chlorine radical - Cl•)
Curly arrows
Curly arrows show the movement of an electron pair, which is used in reaction mechanisms and heterolytic fission
Half curly allows are used in homolytic fission to show the movement of one electron (forming radicals)