Chem- chap 8 & 9
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Bond (intermolecular)
Holds atoms together
Ex: Ionic, polar covalent, nonpolar covalent
Force (intermolecular)
Attraction between molecules
Ex: dipole-dipole, hydrogen bonding, dispersion
Nonpolar covalent
Electrons are shared equally
Example: O2, CH4
Rule: 0 - 0.4
Polar covalent
Electrons are shared unequally
Ex: H2O, HCL
Rule: 0.5 - 1.7
Ionic
Higher than 1.7
If the shape is symmetrical?
Cancels out and it’s nonpolar
If the shape is assymetrical?
Polar
London forces dispersion
Present in all molecules
Only force in nonpolar molecules
Ex: CH4
If nonpolar- dispersion only
Dipole- dipole
Between polar molecules
Ex: HCl
Rule: polar molecule - has dipole-dipole
Hydrogen bonding
H is bonded to N, O or F
Ionic interactions
Between ions (full charges)
Strongest attraction
Ex: na+ and cl-
Solids
Shape
Volume
Boyle’s Law
Pi Vi = Pf Vf
Charles Law

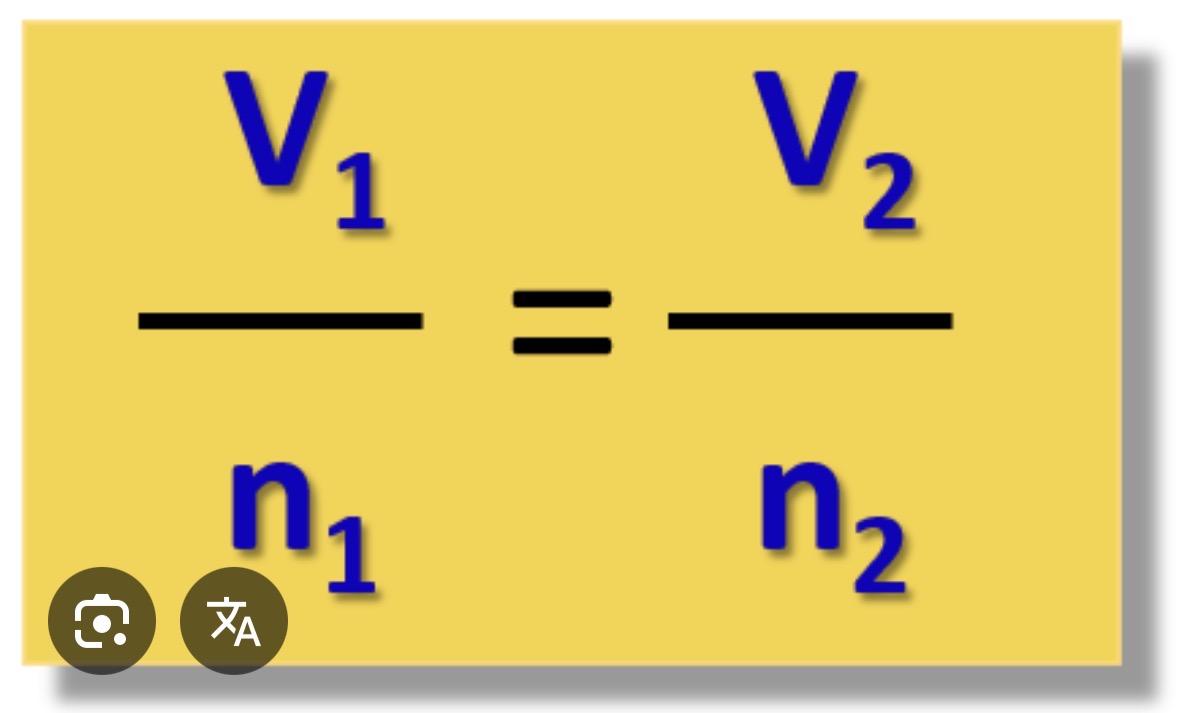
Avogrado’s law
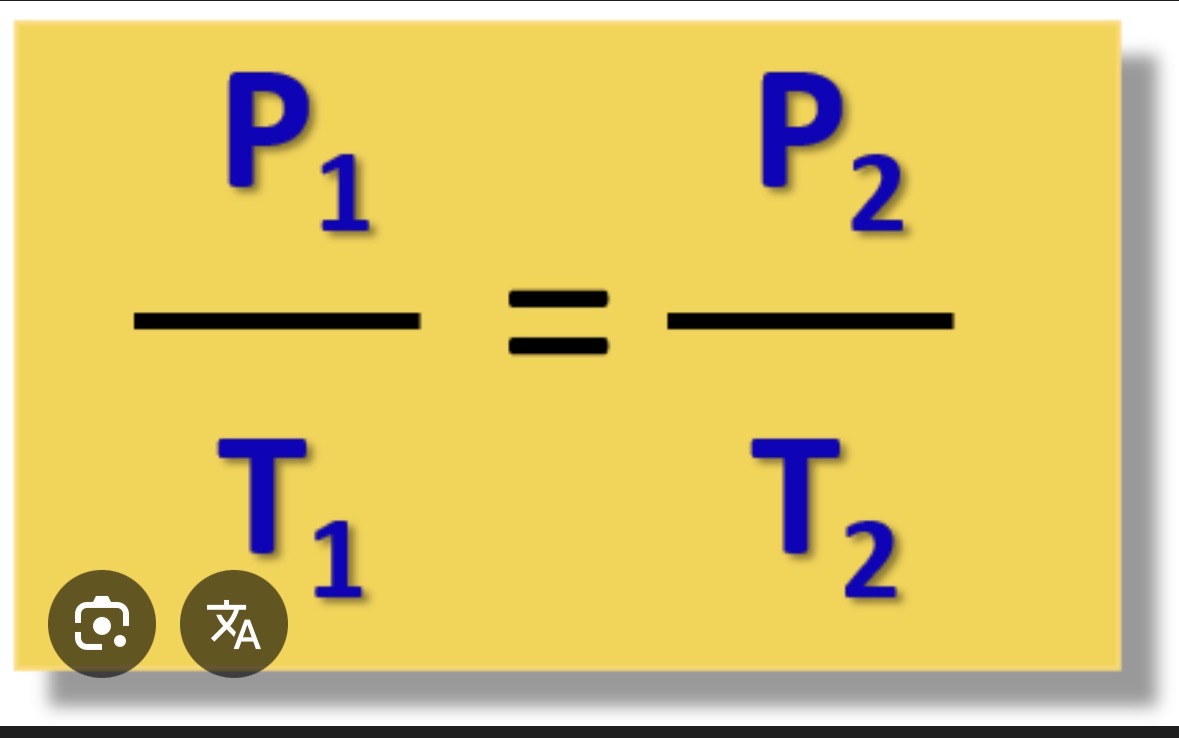
Gay lussac’s
Ideal gas law
