C7.3 Preparation of salts
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What is a salt?
Salts are compounds formed when a metal or ammonium ion (NH₄⁺) replaces the H+ ions in an acid
What is the composition of a salt?
Have cation and anion:
Cation comes from the metal ions from either the metal (hydroxide, oxide, carbonate) or ammonium reactant
Anion comes from the acid used
What is a hydrated substance?
A substance that is chemically combined with water
What is an anhydrous substance?
A substance containing no water
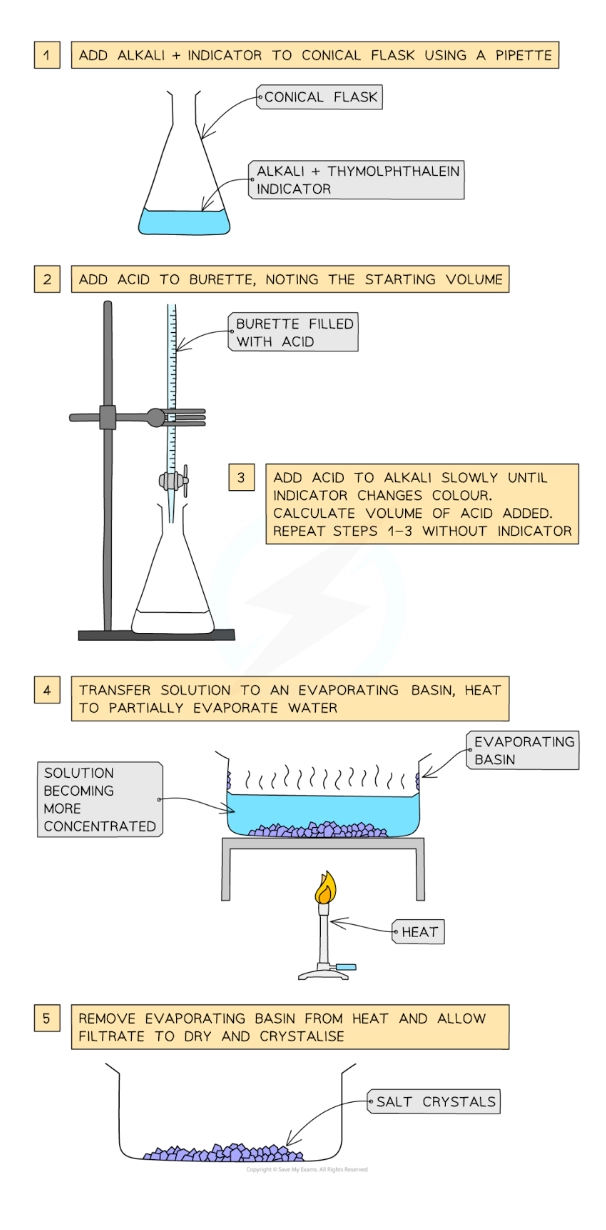
Describe the preparation, separation and purification of soluble salts by the reaction of an acid with an alkali (soluble base) by titration
Use a pipette to measure the alkali into a conical flask and add a few drops of indicator (phenolphthalein or methyl orange)
Add the acid into the burette and note the starting volume
Add the acid very slowly from the burette to the conical flask until the indicator changes to the appropriate colour
Note and record the final volume of acid in the burette and calculate the volume of acid added (starting volume of acid - final volume of acid)
Add this same volume of acid into the same volume of alkali without the indicator
Heat the resulting solution in an evaporating basin to partially evaporate, leaving a saturated solution (crystals just forming on the sides of the basin or on a glass rod dipped in and then removed)
Leave to crystallise, decant excess solution and allow crystals to dry
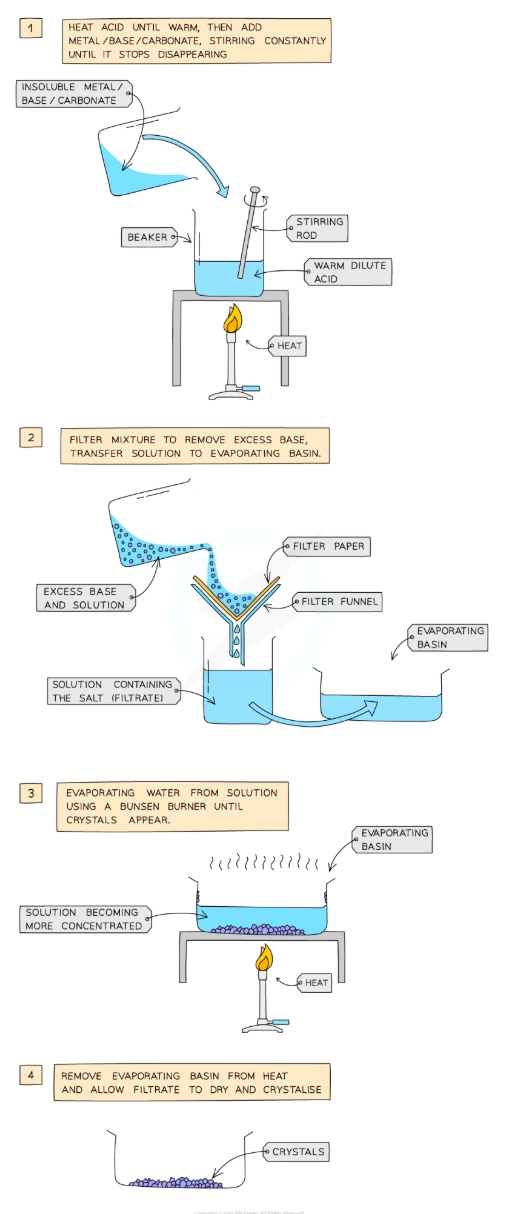
Describe the preparation, separation and purification of soluble salts by the reaction of an acid with excess metal/excess insoluble base/excess insoluble carbonate
Add dilute acid into a beaker and heat using a bunsen burner flame
Add the insoluble metal, base or carbonate, a little at a time, to the warm dilute acid and stir until the base is in excess (i.e. until the base stops disappearing and a suspension of the base forms in the acid)
Filter the mixture into an evaporating basin to remove the excess base
Heat the solution to evaporate water and to make the solution saturated. Check the solution is saturated by dipping a cold, glass rod into the solution and seeing if crystals form on the end
Leave the filtrate in a warm place to dry and crystallize
Decant excess solution and allow crystals to dry or blot to dry with filter paper
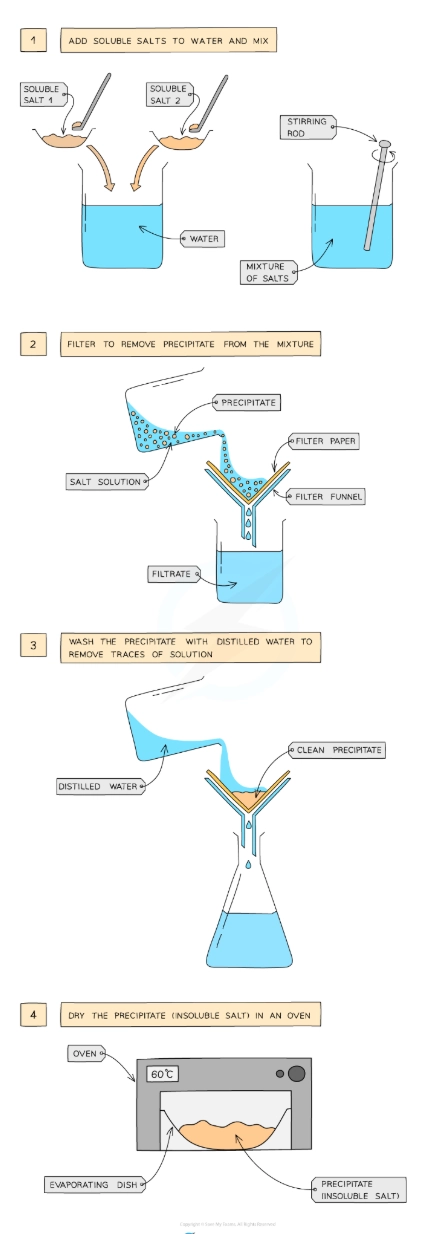
Describe the preparation of insoluble salts by precipitation
To prepare an insoluble salt, a precipitation reaction is used since the salt is formed in the form of a precipitate
Dissolve two soluble salts in water and mix together using a stirring rod in a beaker
Filter to remove precipitate from mixture
Wash the precipitate with distilled water to remove traces of other solutions
Leave in an oven to dry