Distinguishing Tests
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
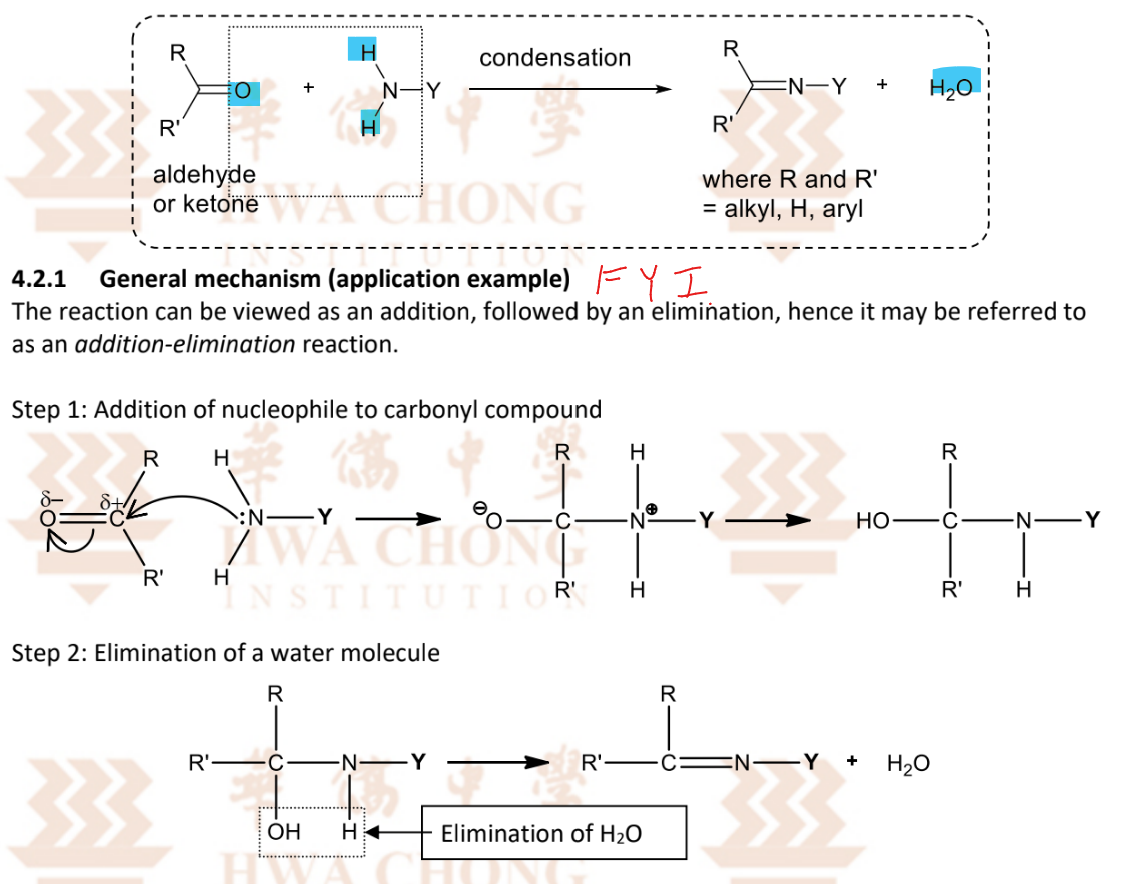
Overall condensation reaction
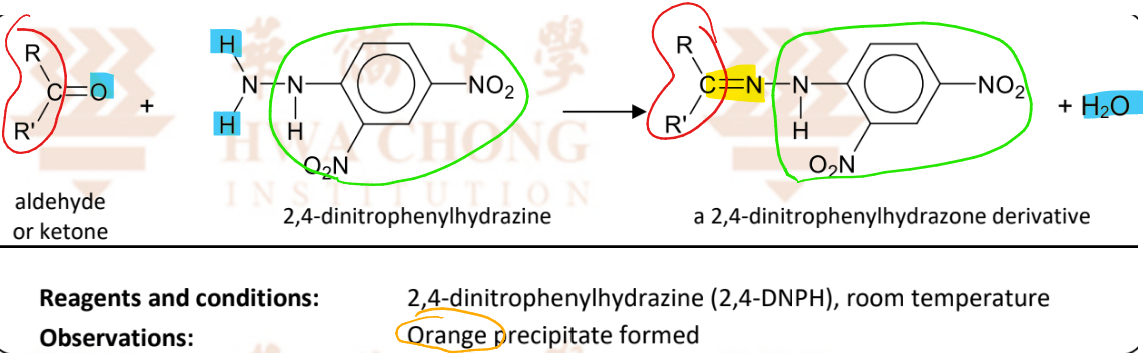
Condensation reaction with 2,4-dinitrophenylhydrazine (2,4-DNPH)
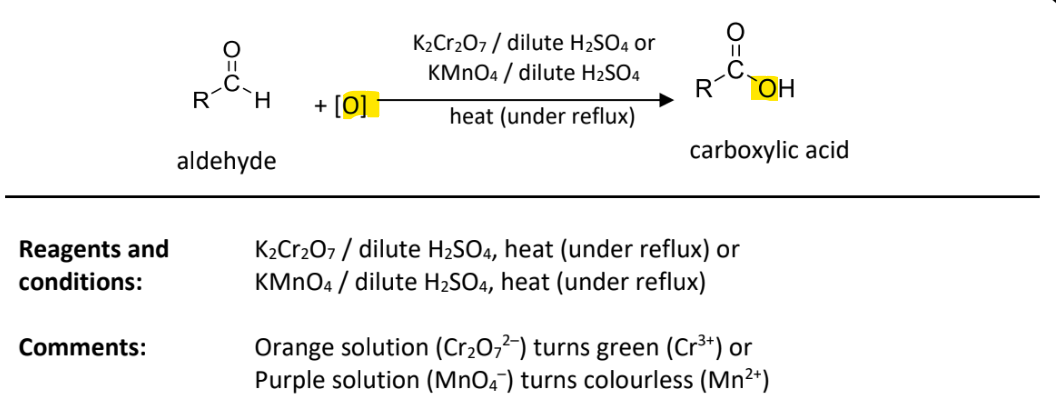
Oxidation with acidified potassium dichromate(V) or acidified potassium manganate(VII)
RnCs: K2Cr2O7 in the presence of dilute H2SO4, heat under reflux
OR KMnO4 in the presence of dilute H2SO4, heat under reflux
Observation: Orange(Cr2O72-) solution turns green(Cr3+)
Purple(MnO4-) solution turns colourless(Mn2+)
Why can’t ketones by oxidised with the same reagents?
no hydrogen attached directly to the carbonyl carbon (FYI: hydrogen needed because C-H bond is weak enough to be broken so electrons can be left behind for formation of new C-O bond)
Oxidation with Tollens’ reagent (silver mirror test)
RnCs: Tollens’ reagent( contains [Ag(NH3)2]+ ), warm
Observation: silver mirror formed
Note: [Ag(NH3)2]+ is unstable and needs to be freshly prepared
one drop of NaOH(aq) added to 3cm3 of AgNO3(aq) to produce Ag2O(dark brown ppt)
dilue NH3(aq) added dropwise until ppt dissolves
![<p>RnCs: Tollens’ reagent( contains [Ag(NH<sub>3</sub>)<sub>2</sub>]<sup>+</sup> ), warm</p><p>Observation: silver mirror formed</p><p></p><p>Note: [Ag(NH<sub>3</sub>)<sub>2</sub>]<sup>+</sup> is unstable and needs to be freshly prepared</p><ul><li><p>one drop of NaOH(aq) added to 3cm<sup>3</sup> of AgNO<sub>3</sub>(aq) to produce Ag<sub>2</sub>O(dark brown ppt)</p></li><li><p>dilue NH<sub>3</sub>(aq) added dropwise until ppt dissolves</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/8b6cfe15-51b8-405a-84c0-00160a734768.png)
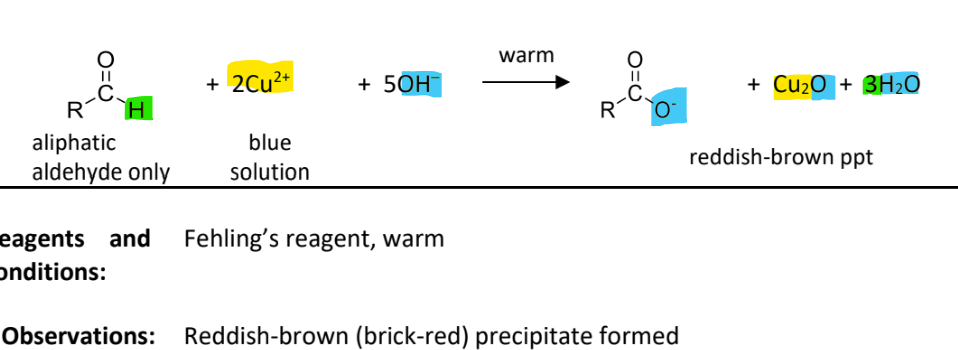
Oxidation with Feling’s reagent
RnCs: Fehling’s reagent(contains copper(II) tartrate), warm
Observation: reddish-brown(brick red) ppt formed
Note:
ONLY aliphatic adlehydes can react
Fehling’s reagent deteriorates on keeping and need to be freshly prepared:
mix Fehling’s solution A (CuSO4 solution) and Fehling’s solution B (sodium potassium tartrate + excess NaOH)
obtain a deep blue solution owing to the formation of copper(II) tartrate, the tartrate ions present prevent the precipitation of Cu(OH)2 by complexing with the copper(II) ions.
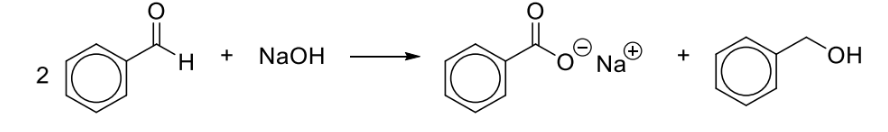
What happens when benzaldehyde reacts with Fehling’s reagent due to excess NaOH?
benzaldehyde undergoes dispropotion instead of oxidation
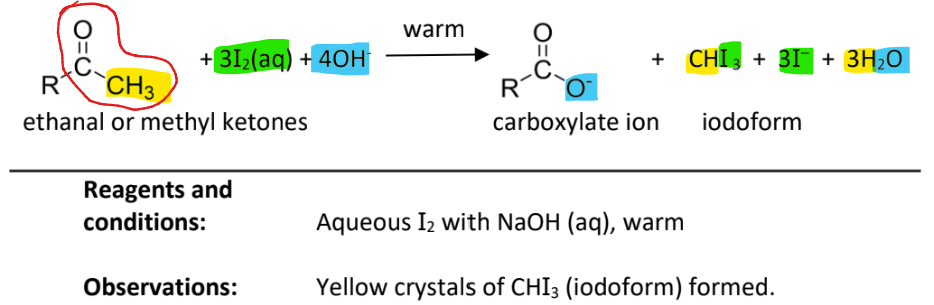
Tri-iodomethane(iodoform) Test
RnC: I2 (aq) with NaOH(aq), warm
Observation: Yellow ppt of yellow crystals of CHI3 formed
note:
carbonyl compound must be ethanal or methyl ketones (the group circled in red must be present in the compound)
Results of distinguishing tests when testing aldehyde
2,4-DNPH: ✅ (orange ppt)
K2Cr2O7 or KMnO4: ✅ (orange turn green/ purple turn colourless)
Tollen’s reagent: ✅ (silver mirror)
Fehling’s reagent: ✅ (reddish-brown ppt)
tri-iodomethan test: ONLY IF ETHANAL ✅ (yellow ppt)
Results of distinguishing tests when testing benzaldehyde
2,4-DNPH: ✅ (orange ppt)
K2Cr2O7 or KMnO4: ✅ (orange turn green/ purple turn colourless)
Tollen’s reagent: ✅ (silver mirror)
Fehling’s reagent: ❌ (benzaldehyde undergoes disproportion, no reddish-brown ppt)
tri-iodomethan test: ❌ (don’t have the CO(CH3) )
Results of distinguishing tests when testing ketone
2,4-DNPH: ✅ (orange ppt)
K2Cr2O7 or KMnO4: ❌ (no H attached to carbonyl C, remains orange/ purple)
Tollen’s reagent: ❌ (no H attached to carbonyl C, no silver mirror)
Fehling’s reagent: ❌ (no H attached to carbonyl C, no reddish-brown ppt)
tri-iodomethan test: ONLY if methyl ketone ✅ (have the CO(CH3) )