chemistry unit two
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
alpha rays
nuclei (He+2) bend toward the negative plate because they have a positive charge.
the most common isotope of carbon
mass 12
atomic # 6
carbon 12
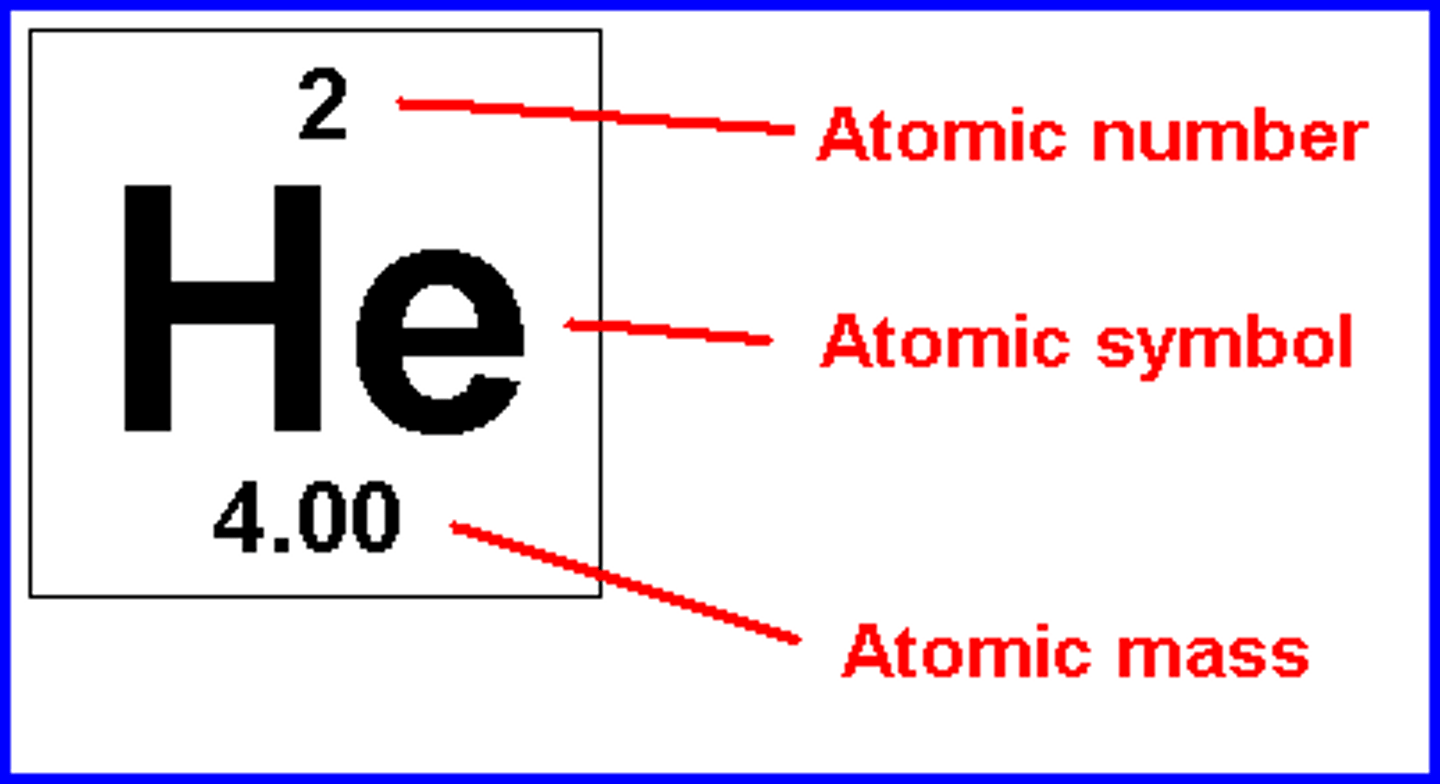
what are the five ways in which a nuclear reaction differs from a chemical reaction?
-nuclear reactions involve nuclei not molecules
-the rate of a nuclear reaction is not affected by change in temp, pressure or catalyst
-nuclear reactions of an atom are the same weather it is in a chemical compound or elemental form.
-different isotopes of an element have essentially the same behavior in chemical reactions but often different behaviors in nuclear reactions
Beta rays
electrons(e-) bend toward the positive plate because they have a negative charge.
gamma rays
photons(y) that do not bend toward either plate because they have no charge.
how can a nucleus emit an electron during beta decay when there are no electrons present in the nucleus to begin with?
during beta decay a weak nuclear force causes a neutron to transform into a proton and in the process emits an electron
what is the difference between an alpha particle and a helium atom?
An alpha particle is the positively charged nucleus of a helium atom, lacking electrons, while a helium atom is electrically neutral with two orbiting electrons.
alpha particle
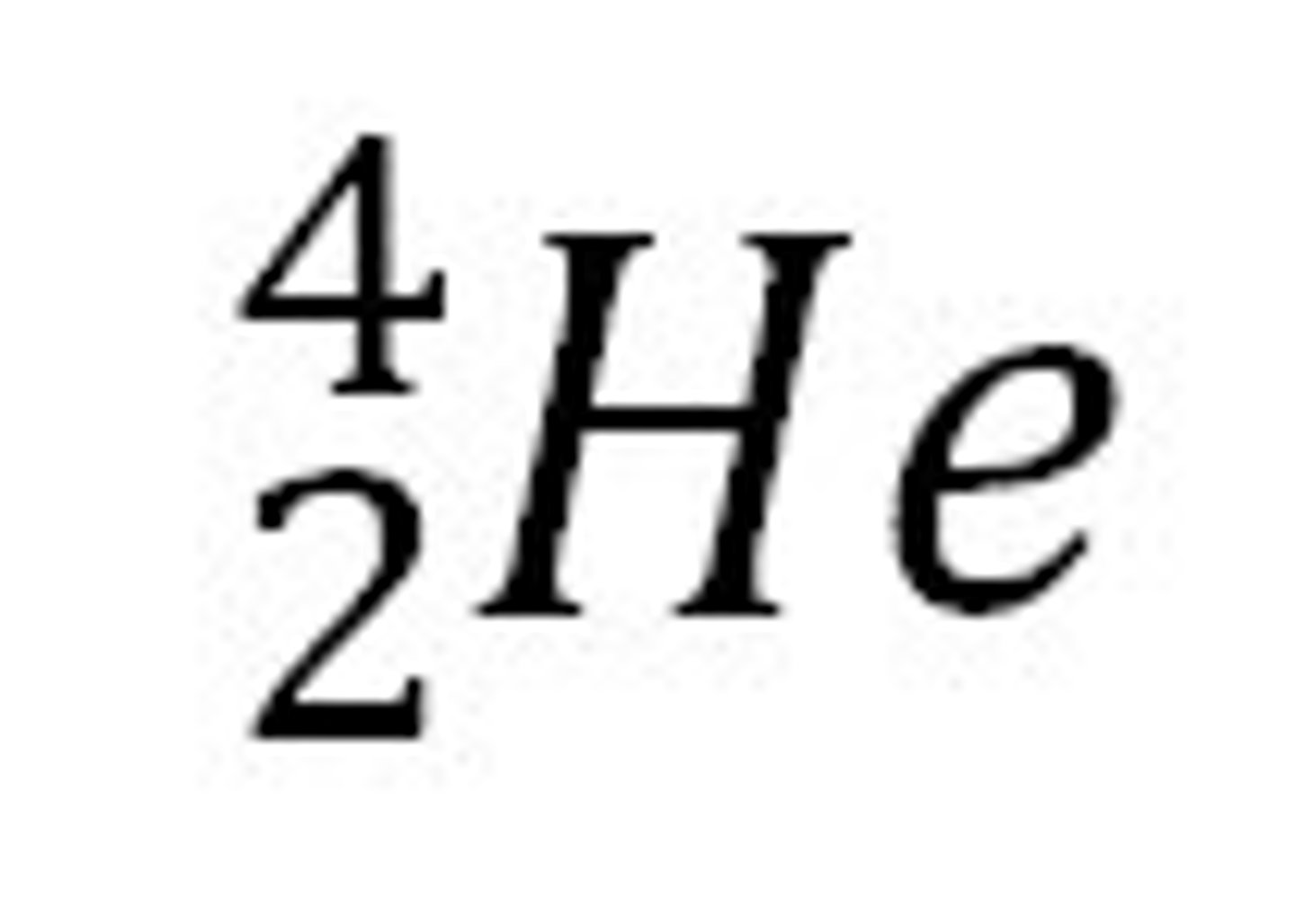
beta particle
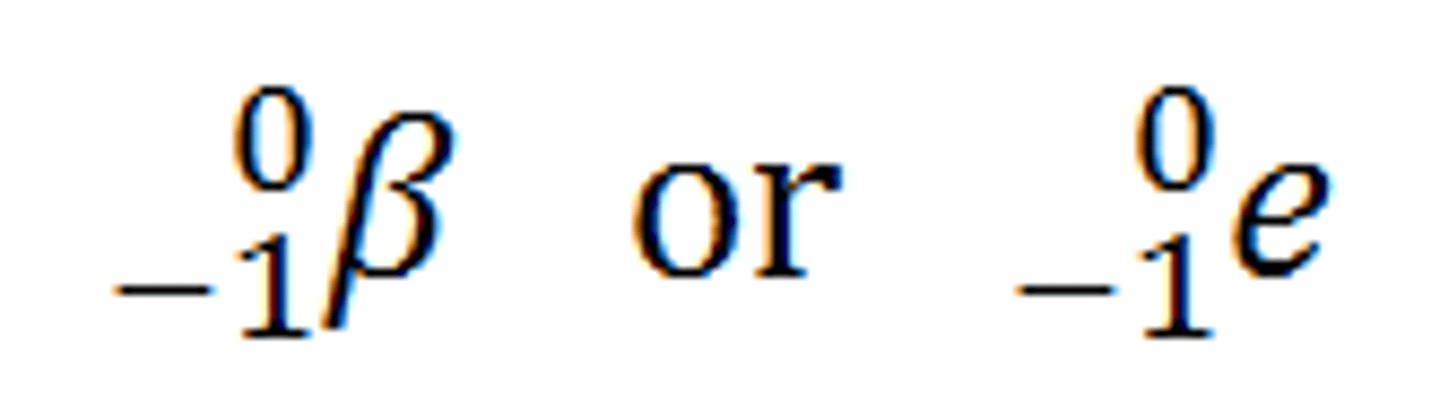
alpha emission equation for uranium-238
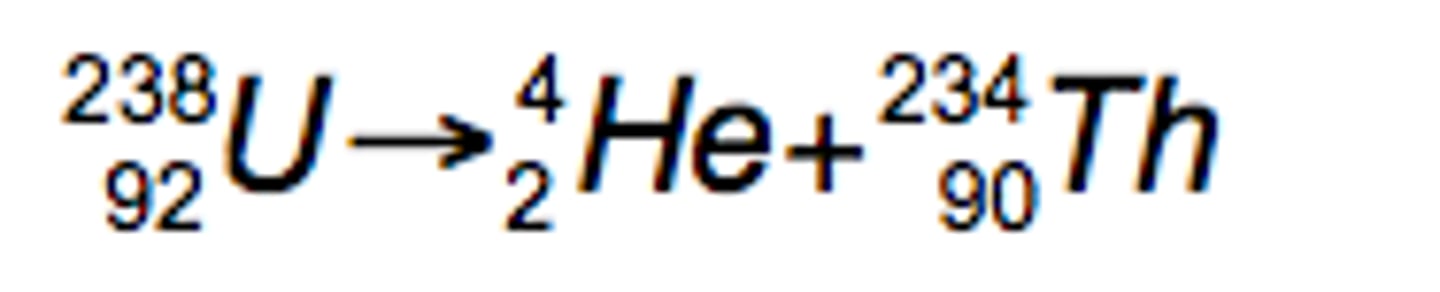
beta emission equation for iodine-131
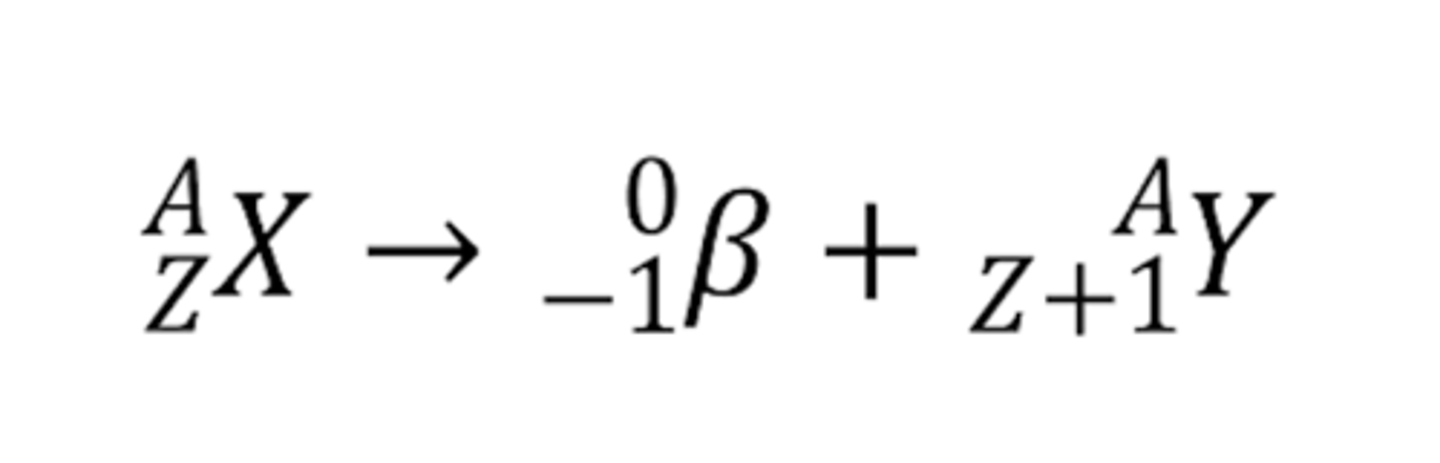
positron emission for potassium-40
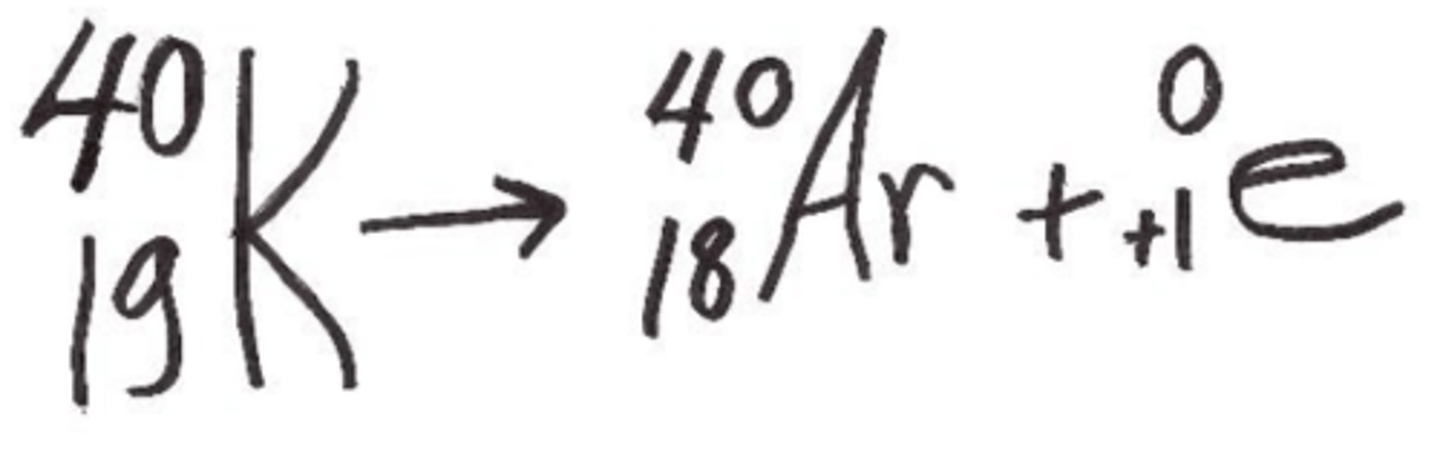
electron capture
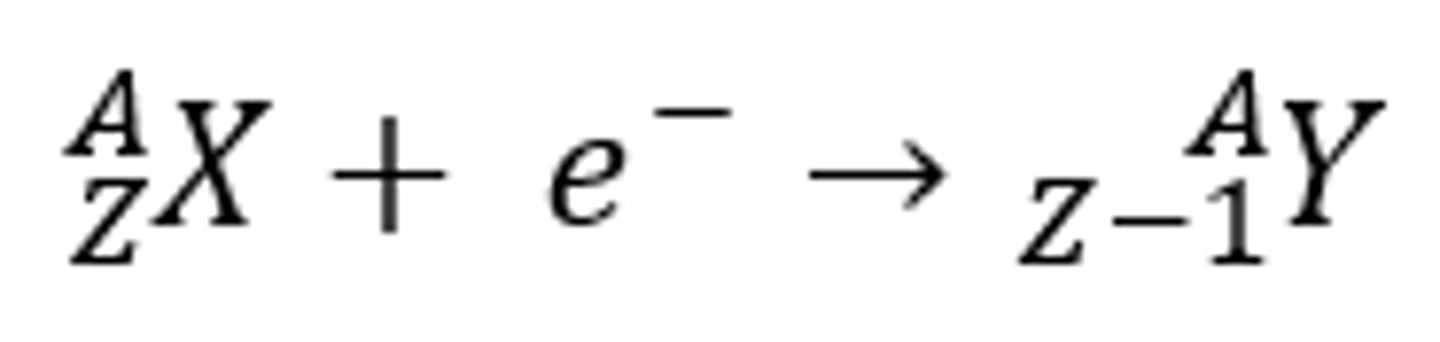
delta g determines
if the reaction is spontaneous
what indicates a spontaneous reaction
ΔG < 0
percent yield

a substance that is oxidized causes
reduction
a substance that gives up an electron is
a reducing agent
a substance that is reduced is
oxidizing agent
delta g =
delta H - T delta S or G products - G reactants
if bonds broken is greater than bonds formed then energy in - energy out =
+ delta H endothermic, heat absorbed, unfavorable
delta H =
bonds broken - bonds formed
energy in - energy out
if bonds broken is less than bonds formed than energy in - energy out =
- delta H, exothermic, releases heat, favorable
delta S =
entropy of products (SP)-entropy of reactants (SR)
delta H
change in heat (enthalpy)
delta S
change in disorder (entropy)
entropy of products greater than entropy of reactants, than
delta S is favorable
entropy of products is less than entropy of reactants, than
delta S in unfavorable
Oil Rig
oxidation is gaining electrons
Reduction is gaining electrons
Avogadro's law
V1/n1=V2/n2
the volume of gas is directly proportional to its molar amount at a constant pressure and temperature
STP
=0c=273.15k and 1atm=760mmHg
standard molar volume
22.4 L/mol
r=
0.0821 L atm/mol K
always soluble
NH4+, Li+, Na+, K+, NO3-, ClO4-, CH3CO2-
usually soluble
Cl-, Br-, I-, SO42-
insoluble everything else
e.g. CaCO3