RAD 107 FINAL EXAM STUDY GUIDE
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
ALARA stands for…
As Low As Reasonably Achievable
Mass vs weight
mass - described in an object based on energy equivalence
weight - force exerted on an object by gravity
Energy equivalence equation
E=mc²
Conservation of Energy law
energy can neither be created nor destroyed
Conservation of matter law
matter can not be created nor destroyed
Alpha particle
Particle released during alpha emission, 2 protons 2 neutrons but 0 electrons
Beta particle
“electron” (except from different origin) that emerges from the nucleus during beta decay
Positron
electron with a positive charge
X-rays and gamma rays
electromagnetic waves usually emitted with particles, travels at the speed of light
radiation exposure measured in air
traditional: Roentgen
SI: Graya
Medical radiation exposure to a patient, measured in tissue
Traditional: RAD
SI: Grayt
Occupational exposure to radiation
Traditional: REM
SI: Sievert
Roentgen definition
Unit of radiation exposure in the air, measures using only x-rays and gamma rays
Atoms vs Molecules
atoms are individual units of matter
molecules are groups of 2+ atoms
Radiation definition
Energy emitted and transferred through space
Matter definition
anything that has mass and takes up space
Ionization definition
the removal of an electron from an atom
Energy definition
the ability to do work
Types of energy
potential, kinetic, chemical, electrical, thermal, nuclear, electromagnetic
Inertia definition
property of matter that resists change in motion or at rest
potential vs kinetic energy
potential energy is stored energy of a position or configuration
kinetic energy is the energy associated with the motion of an object
Formula for kinetic energy
KE = ½ mv²
m= mass
v²= velocity squared
final units: kg(m/s)²
Speed
rate at which objects cover distance (rate of change of position)
Acceleration
rate of change of velocity
Acceleration equation
a = (vt - vo)/ t
final units: m/s²
Fundamental units of measurement
MKS:
distance = meters
mass = kilograms
time = seconds
Derived units of measurements
made up of fundamental units
acceleration = m/s²
velocity = m/s
force = kg(m/s²)
Work = N (kg(m/s²)) * m = Joules (J)
Momentum
mass x velocity
greater velocity = greater momentum
final unit = kg(m/s²) Newton (N)
Properties of matter
has mass, takes up space
Periods (periodic table)
horizontal rows, determines the number of electron shells an element will have (1-7)
Groups (periodic table)
vertical columns, sorted according to number of electrons in the outer shell of an element (1-8)
Atomic Mass Number
number of nucleons (protons and neutrons) in an element
Atomic Mass Unit
unit of mass of an element based on the mass of the Carbon-12 atom
Atomic Mass
includes the number of protons, neutrons, and electrons
Atomic number
number of protons
Elemental mass
weighted average of all atomic isotopes of an element
Noble/Inert Gases
group 8, completely stable and does not react to other elements, full outer shell
Nucleons
protons and electrons
Binding energies
inner shell has the strongest binding energy
outer shell has the weakest binding energy
charges of subatomic particles
proton = +
neutron = O
electron = -
what determines the chemical properties, number of electrons, and number of protons in an atom?
the atomic number
Alpha particles vs helium atom
alpha particle has 2 protons and 2 neutrons but no electrons
Centripetal force
center seeking force that keeps electrons in orbit
Centrifugal force
outward seeking force, helps electrons maintain distance from nucleus
Formula for maximizing the number of electrons in any shell (excluding outer shell)
2n² (n=period/shell number)
Chemical notation
1 1 H (1 proton, 1 neutron, 1 electron)
199 F- (9 protons, 10 neutrons, 10 electrons) (negative charge = add an electron)
199 F+ (9 protons, 10 neutrons, 8 electrons) (positive charge = lose an electron)
neutrons = top number - bottom number
Radioactivity
emission of particles and energy from an atoms nucleus that is unstable (to try and achieve stability)
What type of radioactivity occurs most often
beta emission/decay
Half-life definition
the time it takes for a quantity of radioactive material to be reduced to ½ its original value
Photon vs particle
photons are forms of electromagnetic energy with no mass or charge
particles are the same but posses mass
How much bigger are protons and neutrons than electrons?
2000x
Unit of a quantity of radioactive material
Becquerel = 1 disintegration/second
Speed of light
3.0 × 108 m/s
What fields make up the electromagnetic wave?
electric and magnetic fields
Electromagnetic waves move…
at the speed of light (3.0 × 108 m/s)
Amplitude (waves)
½ the range from crest to valley (or from 0 to crest/valley)
Frequency (waves)
Number of cycles per second
1 Hz = 1 cycle/s
1 cycle = 1 wave
Wavelength (waves)
distance from crest to valley
Wavelength of red light
red = 700 nm
Wavelength of violet light
violet = 400 nm
Where is Planck’s constant used?
used in Planck’s quantum equation: E = hf
E = eV
h = Planck’s constant
f = photon frequency in Hz
What is the relationship of frequency and wavelength in light waves?
Frequency and wavelength are inversely related
The electromagnetic spectrum
(lowest frequency) Radiofrequency — microwaves — infrared — visible light — ultraviolet — x-rays and gamma rays (highest frequency)
Wave equation
Velocity = Wavelength x Frequency
c = λf
How is the wave equation used regarding electromagnetic waves?
velocity is constant and is equal to the speed of light
How is the wave equation used with sound waves?
the velocity varies depending on the density of the material it passes through
density decreases = speed increases
Resistor
inhibits electron flow

Battery
provides electrical potential

Capacitor (condenser)
momentarily stores electric charge

Ammeter
measures electric current

Voltmeter
measures electric potential

Switch
turns circuit on or off by providing infinite resistance

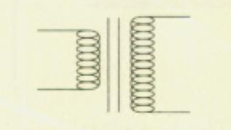
Transformer
increases or decreases voltage by fixed amount (AC only)
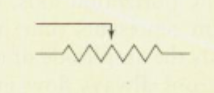
Rheostat
variable resistor
Diode
allows electrons to flow only in one direction

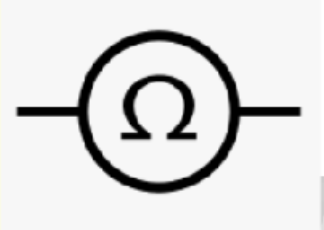
Ohm meter
measures resistance
Short circuit
an unintended pathway that allows the current to pass directly across the electric potential instead of through an element that uses the power
Open circuit
a break in a circuit that stops the flow of electrons
Conventional current
Positive to negative terminal, assumes positive electron charged flow
Electron flow
negative to positive terminal, assumes negative electron charged flow
Volts
joules/coulomb
3 ways for electrification
friction - rubbing electrons off one atom onto another
contact - touching an object so the charge transfers
induction - one charged object attracts molecules and cannot freely move
Electrostatics
study of stationary electric charges
Electrodynamics
study of electric charges in motion
Superconductors
materials with no resistance to conduction (niobium and titanium)
Conductors
Any substance where electrons move freely (copper and aluminum)
Semiconductors
materials that sometimes insulate and sometimes conduct (silicon and germanium)
Insulators
materials that do not allow any electron flow (rubber, wood, glass)
Power
electric rate of doing work (measured in Watts)
What it takes to have a current in a circuit
potential difference (voltage) and a closed conducted path
Coulomb’s Law
Electrostatic force increases directly as the product of the two charges increases. Force decreases in an inverse relation with the square of the distance between 2 objects (leads to inverse square law)
F = K (Q1Q2)/D2
How do electric fields radiate?
Points outward from a positive charge and in towards a negative charge
Ohm’s Law
Potential difference equals the current flow times resistance
V = IR
V= volts
I = electric current in amps
R = electric resistance in Ohms