Lab 9: Amide Synthesis
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
what is an amide?
think of a carboxylic acid but replace the OH with NH2, NHR, or NR2
how do amides typically form?
from the nucleophilic substitution of a carboxylic acid derivative with a primary or secondary amine
what is the point of having aluminum oxide
it provides a common basic polar surface or will also be an acid absorbent to neutralize any acetic acid by product
techniques
solid-liquid extraction, gravity filtration, IR, HNMR, and melting point
table of reagents
acetic anhydride, aluminum oxide, and ethyl acetate
is this reaction exothermic or endothermic?
exothermic
broadly explain the procedure
add aluminum oxide to your unknown amine
add acetic anhydride dropwise and let reaction occur for the next 10 minutes
once the reaction is done, add ethyl acetate to the reaction mixture and extract (aka your product should be in the organic layer)
filter your product with gravity filtration to get the rest out and wash it again with ethyl acetate to ensure all of the product is out of the aluminum oxide
evaporate all the ethyl acetate (product should crystallize)
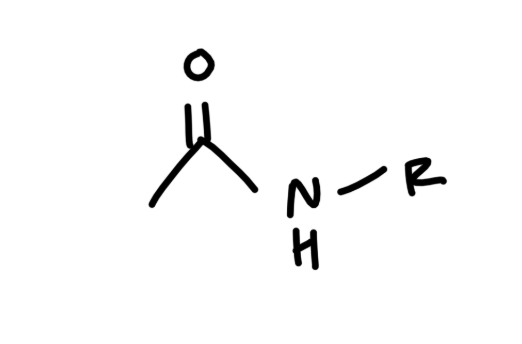
draw a generalized structure of an amide
think carboxylic acid but OH group replaced by NHR
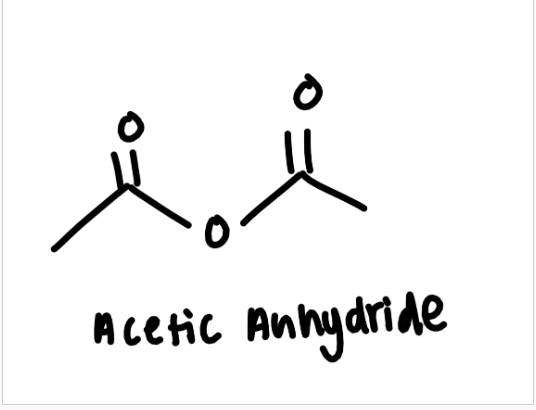
draw acetic anhydride
what classification of starting material will act as the nucleophile
primary amine
what is the chemical formula of the aluminum oxide catalyst
Al2O3
what is the solvent used to extract the amide?
ethyl acetate
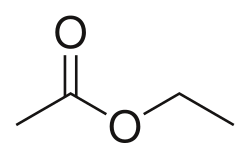
what is the balanced equation
everything is a 1:1 ratio
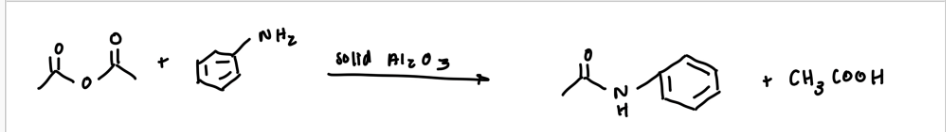
reaction mechanism
nucleophilic attack of amine and the electrons from an alkene goes to the oxygen (will be negative), hydrogen on N will get deprotonated, then the electrons from the alkoxide will come down to form carbonyl, and then the other ester gorup will leave
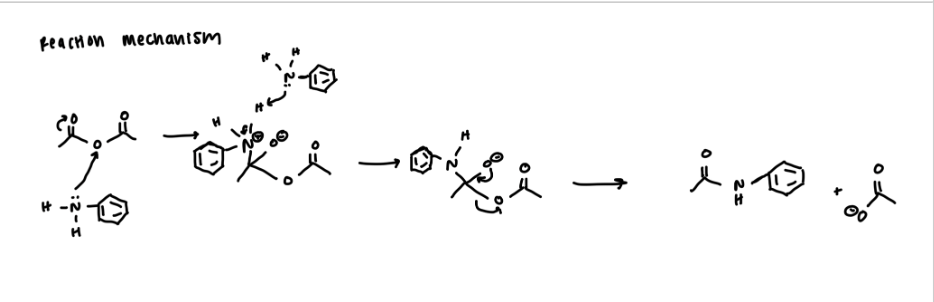
limiting reagent
unknown amine
did your IR change to see if you had an amide form?
yes, first i looked for a strong narrow peak to see the adjacent carbonyl group. the second is if there was a broad peak around 3100-3500 (but not as wide as an OH signal). this represents the amide but see how many defined peaks there are to see if it is a primary amide (two H’s) or secondary amide (one H)
Why is it essential that no water or alcohols are present during the amide synthesis reaction you performed in lab this week?
because they will be competing nucleophiles and will want to react with acetic anhydride to form a carboxylic acid or ester instead of an amide (see balanced equation)

Provide the appropriate starting materials and reagents needed to form the following compounds using this week’s reaction parameters:
N-(3-ethylpentyl)-2-methylpentanamide:
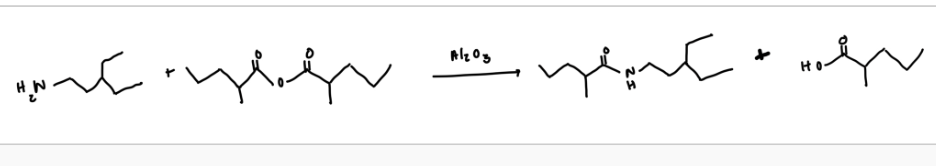
Provide the appropriate starting materials and reagents needed to form the following compounds using this week’s reaction parameters:
2-methyl-N-(3-methylcyclohexyl)butanamide:
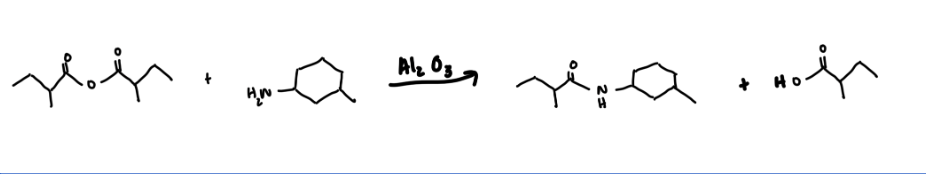
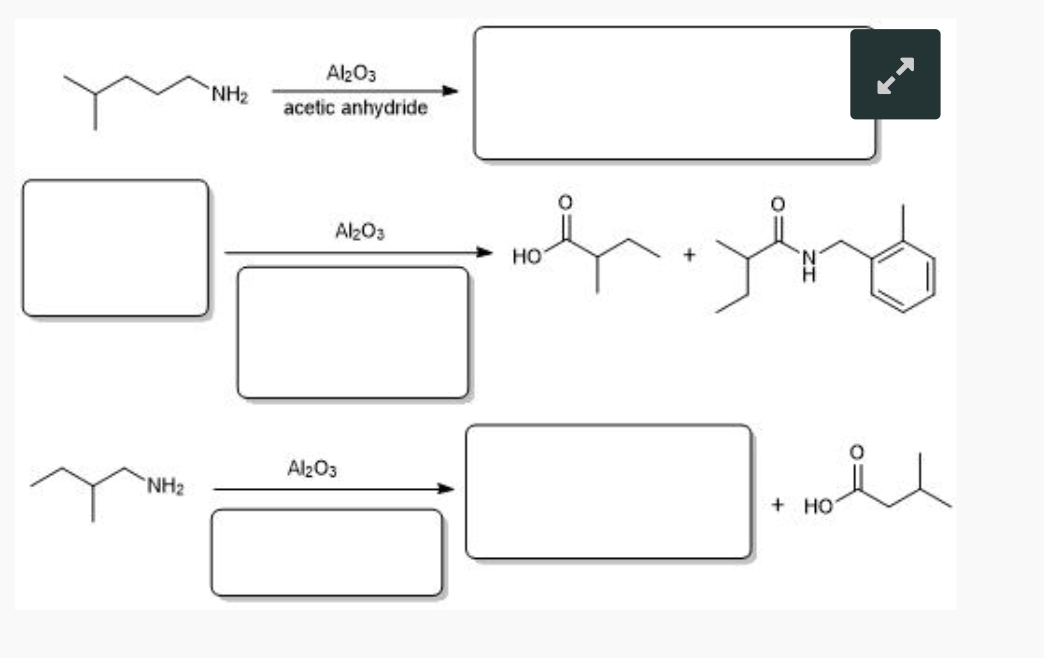
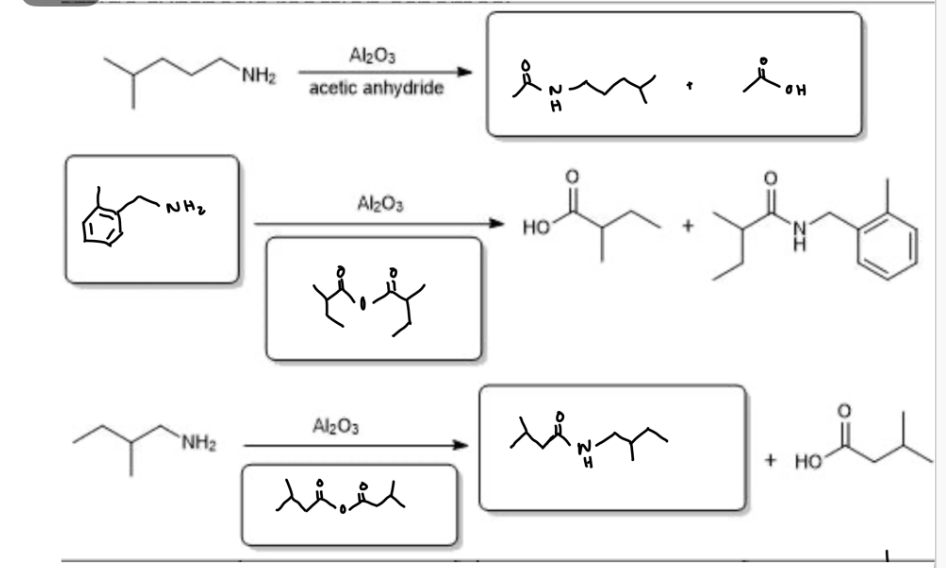
What are the appropriate starting materials and/or products needed to complete this synthesis