Kinetics
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
State why the curve starts at the origin
EQ
No molecules with no energy
Explain the effect that lowering the temperature would have on the rate of reaction
EQ
Faster rate of reaction
Explain the effect that this change has on the rate of reaction
EQ
Amount of gas prevent reduced
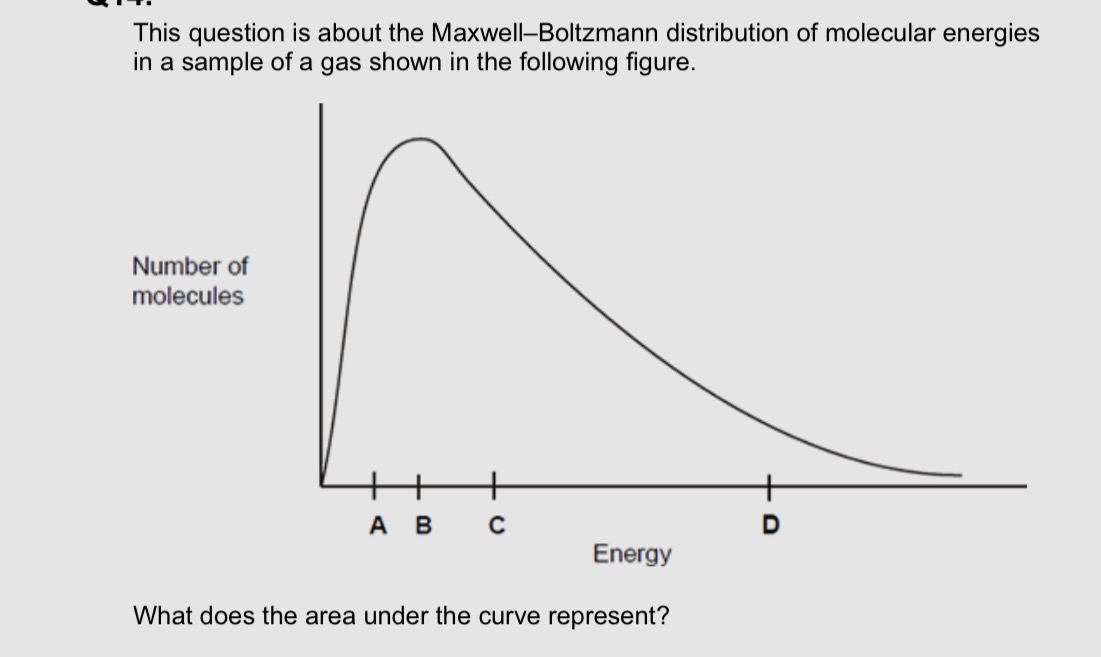
What does the area under the curve represent?
The total number of practicals
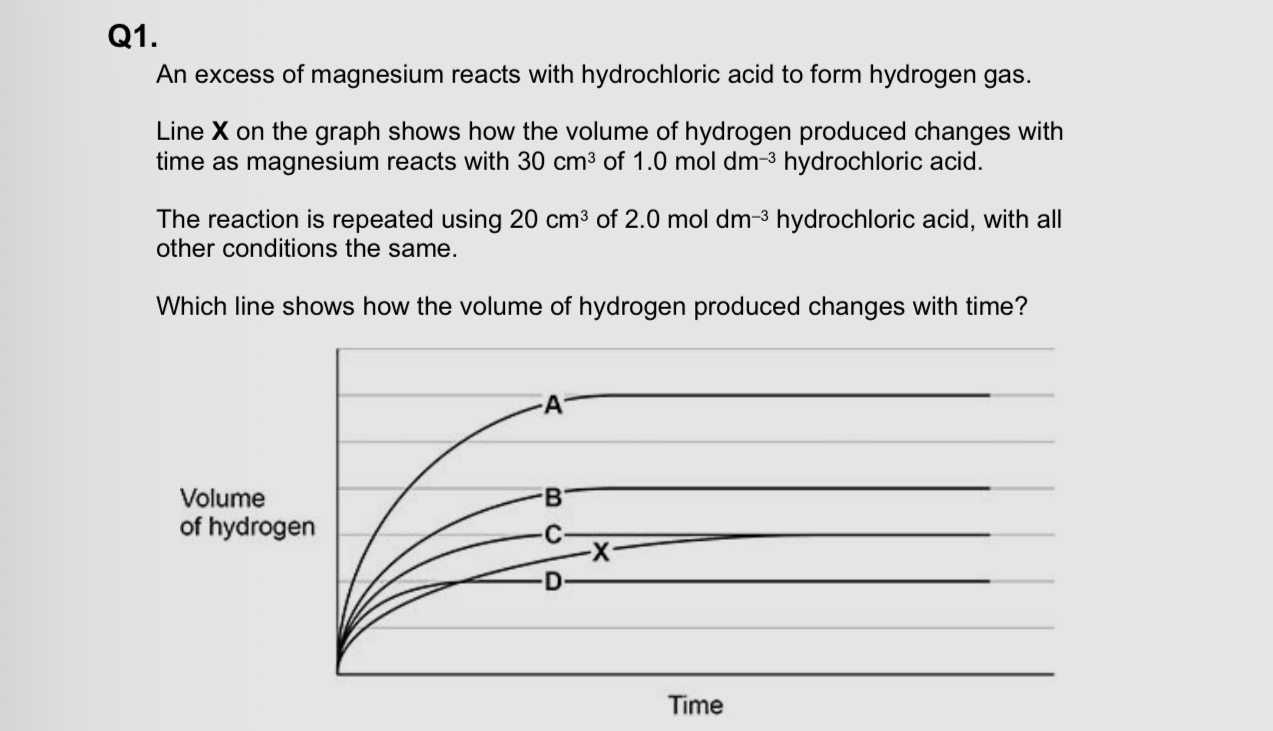
B

A
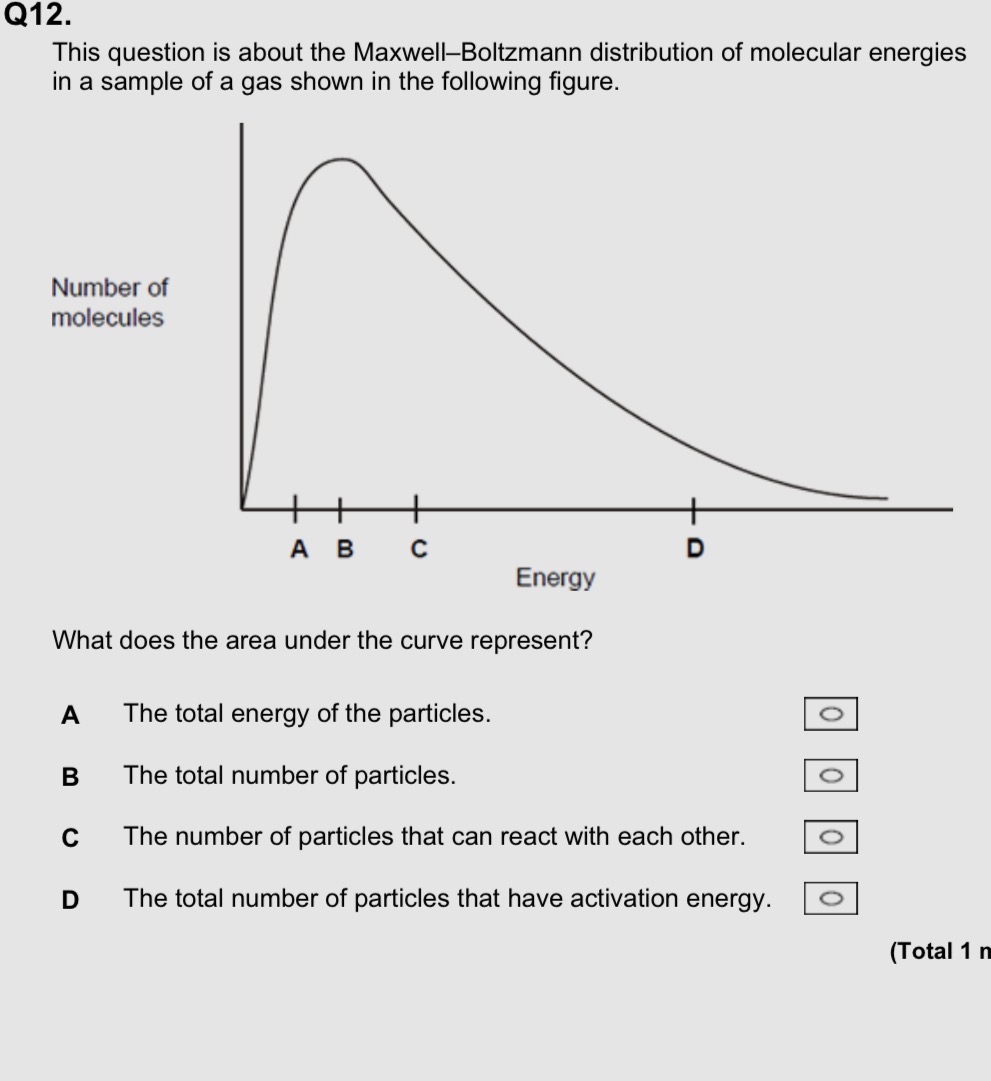
B
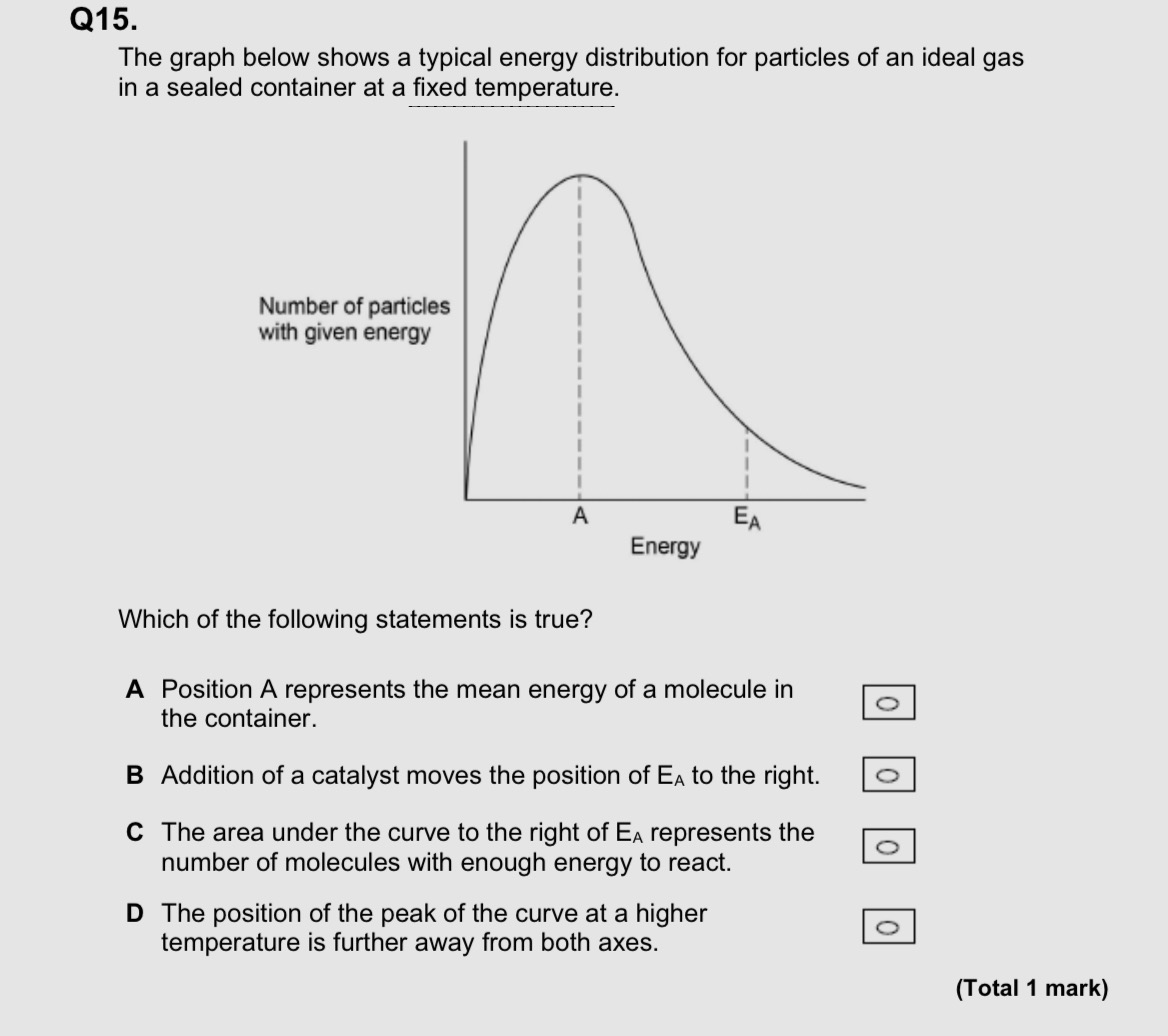
C
Which
Which statement is correct for the distribution curve of molecular energies in a gas?
True or false
Curve is symmetrical about the maximum
False
Is this likely to decrease the accuracy of the experiment?
Rinsing the flask with acid before each new experiement
EQ
Yes
Which statement is correct about the time taken for the cross to disappear when using the larger conical flask?
EQ
The time taken will be increased by using the larger conical flask
Suggest why the student record the times to the nearest second and not the nearest 0.1 s
EQ
Hard to judge to the nearest 0.1 second
Suggest why the students chose not to carry out experiments at temperatures in the range 1-10C
EQ
Reaction would take too long
Do particles in a sample have the same kinetic energy at the same temperature
EQ
Yes
How does Maxwell Boltzmann distribution curve change at a higher temp
Lower and right
How does the maxwell botlzmann curve change at lower conc
Lower graph shape by same factor lower
Explain why a small increase in temperature causes a large increase in the rate of reaction
EQ
More particles have the activation energy
More frequent and successful collision per unit volume
Which statement is correct for the distribution curve of molecular energies in a gas?
True or false
There are always some molecules with zero energy
EQ
False
Which statement is correct for the distribution curve of molecular energies in a gas
True or false
The position of the maximum of the curve is not dependant of the temperature
False
Which statement is correct for the distribution curve of molecular energies in a gas
True or false
The position of the maximum of the curve is not dependant on the temperature
False
Which statement is correct for the distribution curve of molecular energies in a gas
True or false
The mean energy of the molecules is greater than the most probable energy of the molecules
True