BDS 2 MCQ bioscience
1/227
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
228 Terms
what does HIV cause?
opportunistic infections
secondary to severe impairment of cell-mediated immunity
how is HIV transmitted?
blood- parenteral/ intravenous drug user
bodily fluids- sexual transmission; baby delivery/ breastfeeding
how to treat HIV if suspected exposure?
PEP- 4-72 hours after
How does HIV bind to host cells?
CD4+ Th receptors
replication of HIV
bind to CD4+ Th receptors on host cells
reverse transcriptase converts viral RNA —> DNA
integration to host DNA
genome packaged in protein coat by proteases
release of mature virus—> infects other cells
universal precaution of HIV
everyone is presumed to have HIV until proven otherwise
opportunistic infections caused by defective cell-mediated immunity caused by HIV
intracellular pathogens
myobacter
—> TB
opportunistic infections caused by defective cell-mediated immunity caused by HIV
viruses
cytomegalovirus
varicella zoster—> chicken pox—> shingles
opportunistic infections caused by defective cell-mediated immunity caused by HIV
fungal
meningitis
oral candidiasis
opportunistic infections caused by defective cell-mediated immunity caused by HIV
protazoal
cerebral toxoplasmosis
opportunistic infections caused by defective cell-mediated immunity caused by HIV
cancers
kaposi’s sarcoma- gumline/ roof of mouth
HPV
classification of HIV
<200 CD4+ Th
dental symptoms of HIV
angular chelitis- fungal
glossitis
candida
oral hairy leukoplakia
gingivitis
parotitis
kaposi’s sarcoma on gumline/ roof of mouth
oral ulcers
roughly how long does HIV stay latent?
10 yrs
what are long-term non-progressors?
stay in latent phase of HIV
when are you cured of HIV?
HIV RNA undetectable
how does BIKTARVY treat HIV?
targets HIV replication cycle
increases immune system
side effect of BIKTARVY to treat HIV
induce/ inhibit Cytochrome P450
= subtherapeutic/ toxic levels of oral sedatives e.g. medazolam
test for HIV
antibody HIV + P24 antigen
current infection
what is window period?
takes up to 3 months to make HIV
what is anatomical dead space?
respiratory system volume excluding the alveoli
function of anatomical dead space
transports air to alveoli to stop big changes in CO2
ventilation inefficiency
25% of air stays in airways
not all air inspired used for gas exchange
tidal volume
500mls
what is alveolar dead space?
lung disease
alveolar not acting as respiratory membrane
what is physiological dead space?
anatomical + alveolar dead space
areas not involved in gas exchange
circulatory inefficiency
difference in alveolar and circulation pO2 due to venous shunting
what is alveolar pO2?
104mmHg
what is circulatory pO2?
95mmHg
where is venous shunting present?
bronchial circulation: deoxygenated blood from airways to left atrium
pulmonary circulation: deoxygenated blood from thebesian veins of heart to left ventricle
what is required for efficient gas exchange?
good match between alveolar ventilation (Va) and blood flow (Q)
what happens to Va/ Q when
embolus/ vasoconstriction?
decreased blood flow (Q)
increased alveolar dead space
what happens to Va/ Q when
mucus plug/ pneumothorax/ broncoconstriction?
decreased alveolar ventilation (Va)
increased alveolar dead space
consequences of hypoxia
close O2 sensitive K+ channels
induced vasoconstriction of pulmonary arteries
increased pulmonary pressure in pulmonary arteries
cor pulmonale
what happens to Va/ Q when
underventilated
low alveolar ventilation (Va)
lower pO2 no CO2 change
what happens to Va/ Q when
overventilated
high alveolar ventilation (Va)
insufficient blood flow (Q)
consequences of high CO2
lowers pH of CSF and blood
detected by carotid/ aortic bodies
autonomic control centre in pons/ medulla
resp. motor neurones—> muscles
= increase rate and depth of breathing= remove more CO2
consequences of O2 treatment being too high
removes hypoxic drive
type 1 respiratory failure
low PaO2 AND low PaCO2
examples of type 1 respiratory failure
acute asthma
lung fibrosis
pulmonary embolism
type 2 respiratory failure
low PaO2 and HIGH paCO2
examples of type 2 respiratory failure
decreased ventilatory drive= sedative overdrive
decreased neuromuscular= myopathy
failure to reset chemoreceptors= COPD
what is asthma?
REVERSIBLE
chronic
bronchial hyperesponsiveness
= Bronchoconstriction
atopic triggers for asthma
Allergen- IgE
non-atopic triggers for asthma
exercise
chemicals
smoke
pathophysiological signs of asthma
smooth muscle cell spasm
bronchial inflammation
goblet cell hyperplasia
immune reaction to triggers in asthma
mast cells secrete histamine/ leukotrienes
infiltration of Th2 —> nitric oxides
chemokines —> damage and irritation to epithelium
diagnosis of asthma
spirometry
Fev1/ FEC <0.7
asthma treatment
relievers
short-acting beta-2-agonist: stimulate beta receptors to increase cAMP= bronchodilators e.g. Salbutamol
antimuscarinics: block ACh binding to muscarinic receptors
xanthine= relax smooth muscle/ bronchodilator
asthma treatment
preventors
leukotriene receptor agonists= block leukotrienes causing bronchospasm
glucoticosteroid- synthesised from cholesterol to reduce pro-inflammatory mediators
monoclonal antibodies
long-acting Beta-2-adrenoreceptors- stimulate beta receptors to increase cAMP= bronchodilators e.g. Salmeterol
side effects of Beta-2-adrenoreceptors
muscle tremores
headaches
increased heart rate
hypokalaemia
side effects of glucoticosteroids
oral candidiasis- less T-cells
decreased bone density
adrenal supression
oral side effects of asthma/ COPD treatments
inhaler: dry mouth—> caries
bronchodilator: GERD
corticosteroids:oral candidiasis (reduced T-Cells)
what to do in dental setting if patient has Asthma
avoid triggers e.g. stress/ NO2
asthma pump and salbutamol available
avoid NSAIDS- inhibit COX1/2= less prostoglandins; more leukotrines= bronchospasm
pulse ox. <95% = low flow 24-28% O2 (avoid removing hypoxic drive)
what can cause normal bronchocontriction of the lungs
parasympathetic innervation
ACh
Histamine
what causes natural bronchodilation of the lungs?
Sympathetic innervation
beta-2 receptor activation
whar is COPD?
chronic obstructive pulmonary disease
IRREVERSIBLE
chronic
restricted air flow affecting gas exchange
chronic bronchitis
emphysema
what is chronic bronchitis?
chronic airway inflammation
= affects expiration and inspiration
pathophysiological signs of chronic bronchitis
increased mucus
narrowed airway
fewer cilia
signs and symptoms of chronic bronchitis
productive cough for 3 months within 2 years
elevated haemoglobin
oedema
overweight
cyanotic
what is emphysema?
enalrged alveoli
= affects expiration
pathophysiological signs of emphysema
damaged epithelium= increased cytokines
loss of elastase in alveolar connective tissue
large air spaces
signs and symptoms of emphysema
dyspnoea- increased CO2= barrel chested
overinflated lungs= smaller heart, flattened diaphragm
RHS heart failure
thin
treatments for COPD
anticholinergics- block ACh at receptors= relax smooth muscle and decrease Mucus
Beta-2 adregenic Agonists- stimulate beta receptors to increase cAMP= bronchodilators
corticosteroids- inhibit immune response
cardiac cycle definition
series of electrical and mechanical events dictating blood flow through the heart to the circulation with each beat
role of pacemaker cells
set intrinsic heart rate
location of pacemaker cells
SA node in RA
AV node
purkinje fibres
how do pacemaker cells work?
unstable resting potential in heart
cells spontaneously depolarise
effect of parasmpathetic autonomic innervation to heart rate
open more K+ channels
longer to reach resting potential
= slow HR
effect of sympathetic autonomic control on heart rate
open more Na+ channels
shorter time to reach resting potential
= increases Heart rate
effect of adrenaline hormone on heart rate
increases heart rate
normal Heart rate
50-90 BPM
why do the pacemaker cells in SA node usually set the heart rate?
fastest depolarisation
cardiac cycle electrical pathway
SA node
internodal pathway
AV node
AV bundle
Bundle of His
Purkynje fibres
why is electrical conduction across AV node slower than across atria?
allows Atrial systole
how is ventricular myocardium specialised for rapid cell-cell conduction?
intercalated disks
gap junctions
how does ECG measure cardiac cycle
amalgamation of ion movements/ all action potentials in heart
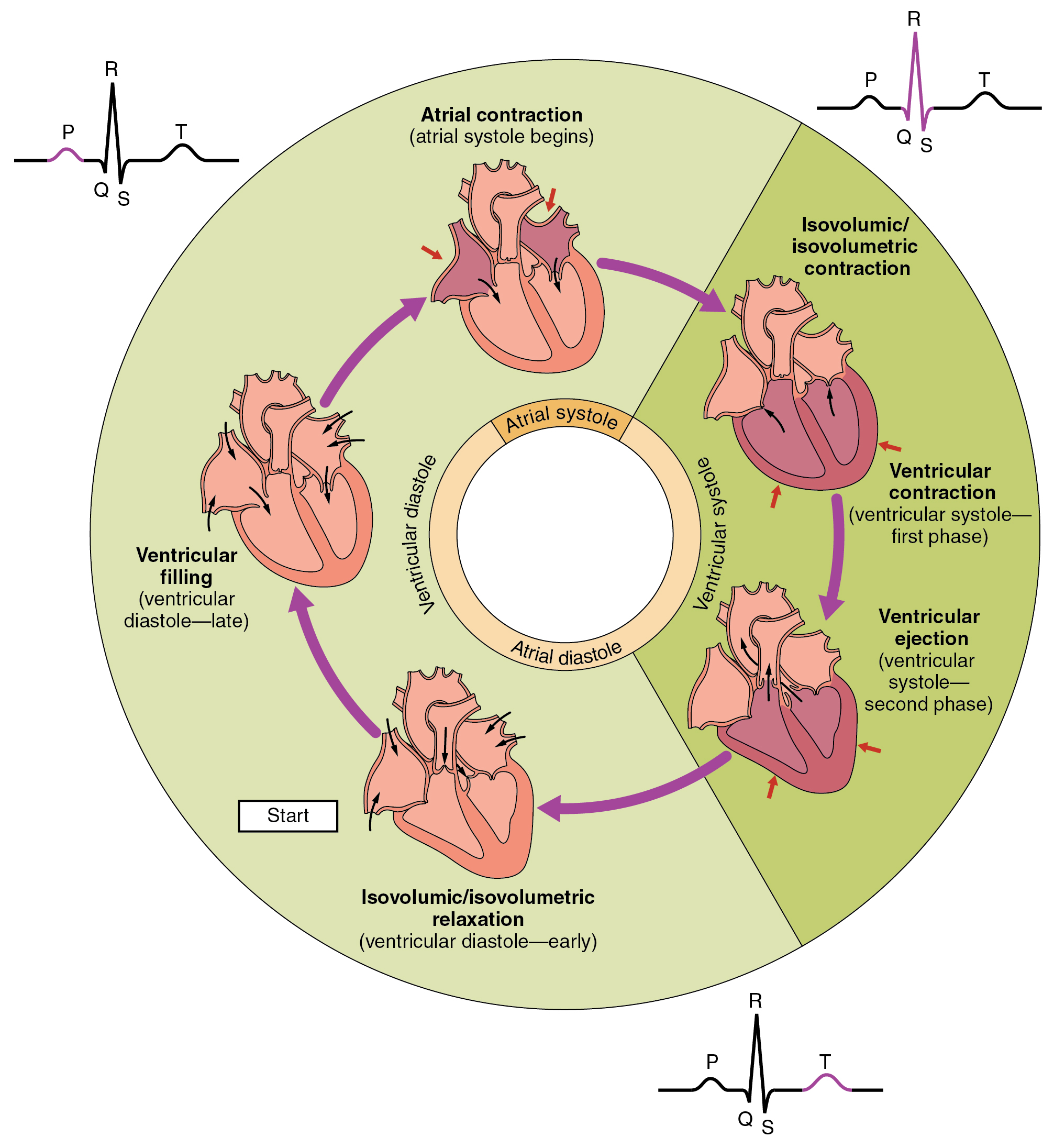
what does P-wave on ECG show?
impulse travels from SA to AV node
atrial depolarisation
= ATRIAL SYSTOLE
what does QRS complex on ECG show?
impulse travels from AV node —> bundle of His —> Purkinje fibres
ventricular depolarisation
= VENTRICULAR SYSTOLE
(+ atrial repolarisation)
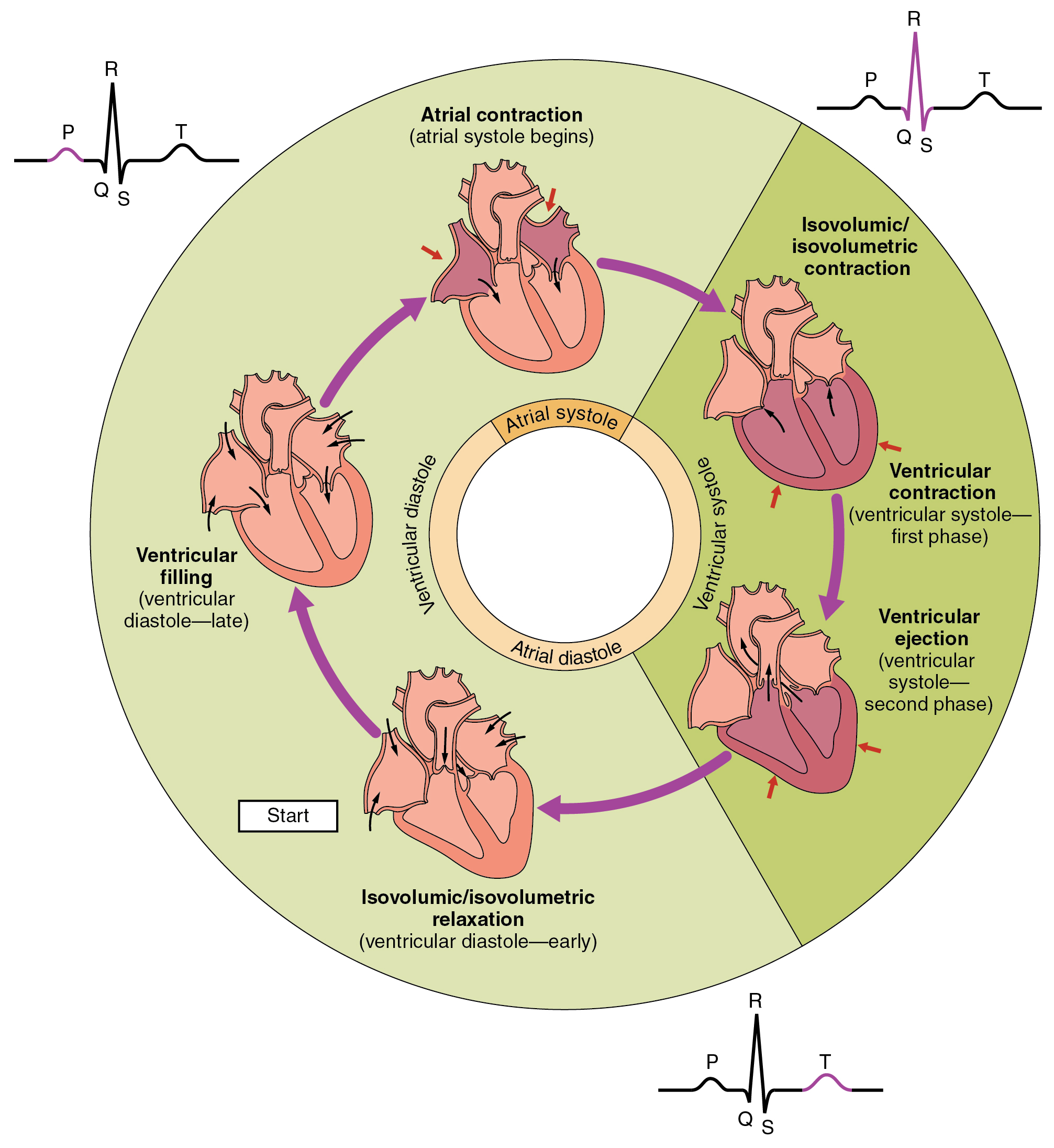
when does atrial repolarisation begin on ECG?
QRS complex
but masked because more ventricular cells
what occurs during T-wave on ECG?
ventricular repolarisation
= ventricular and atrial DIASTOLE
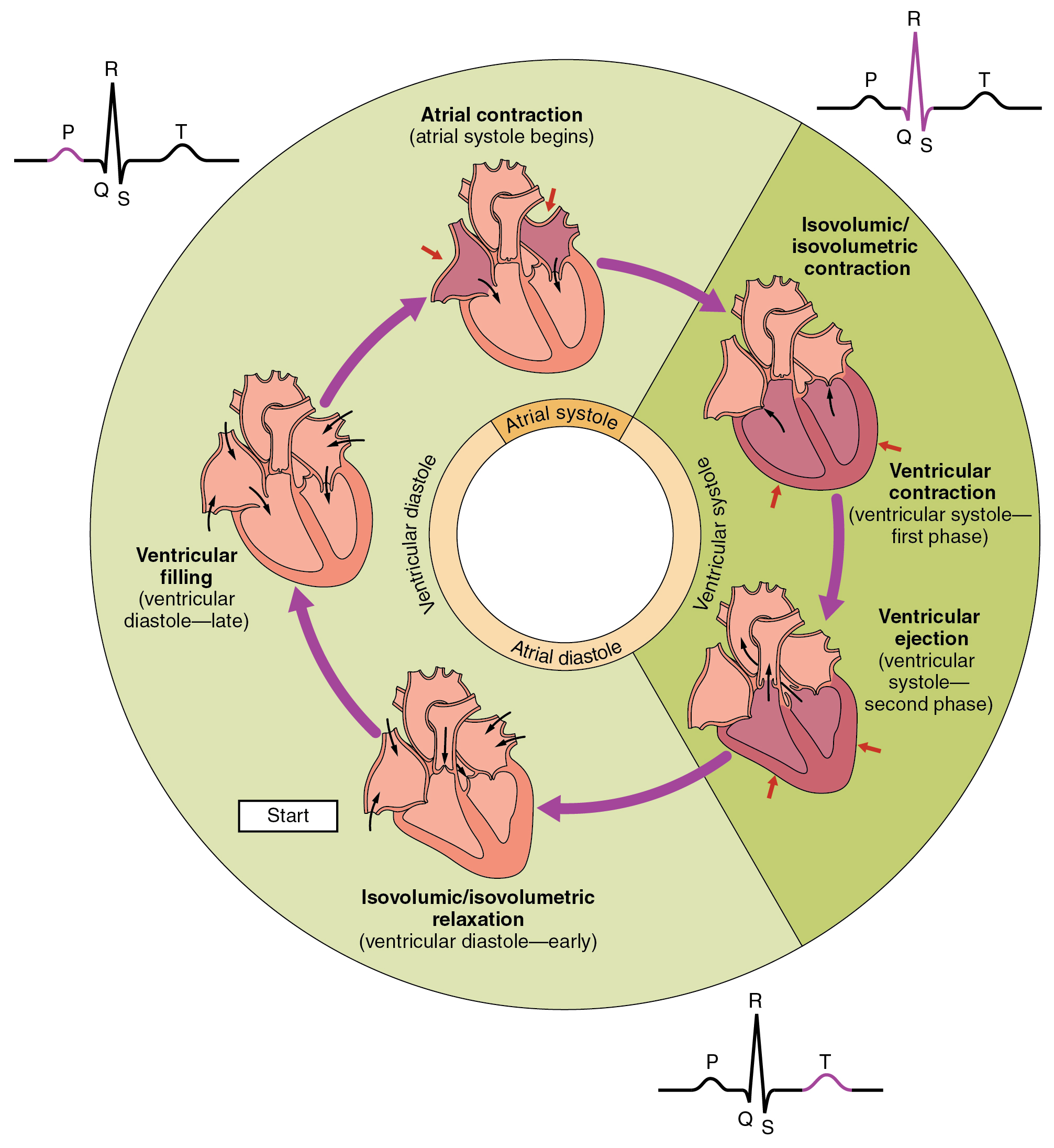
how does blood move throughout the heart?
triggered by electrical events
pressure changes
what can be used to visualise the mechanical events of the cardiac cycle?
Wigger’s diagram
what valves are open in P-wave?
mitral valve open
pressure in atria > ventricle
what valves are closed in P-wave?
Aortic valve
pressure in ventricle < Aorta
what valves are open in QRS complex?
aortic valve open
pressure in ventricle > aorta
what valves are closed in QRS complex?
mitral valve closed= LUB
pressure in ventricle > atrium
what valves are open in T-wave?
mitral valve open
pressure in atrium > ventricle
what valves are closed in T-wave?
aortic valve closed= DUB
pressure in aorta > ventricle
what do the heart sounds represent?
LUB= mitral valve closing in ventricular systole
DUB= aortic valve closing in diastole
what causes systolic murmur?
aortic valve not open
mitral valve not closed
= extra sounds when LV contracting
what causes diastolic murmurs?
mitral valve not closed
aortic valve not open
= extra sounds when LV relaxed
stenosis vs regurgitation
narrowing= valve not opening
Leaking= valve not closed
autoimmune disease definition
specific immune responses to self-antigens resulting in pathology
what can non-organ specific autoimmune diseases affect?
DNA
Histones
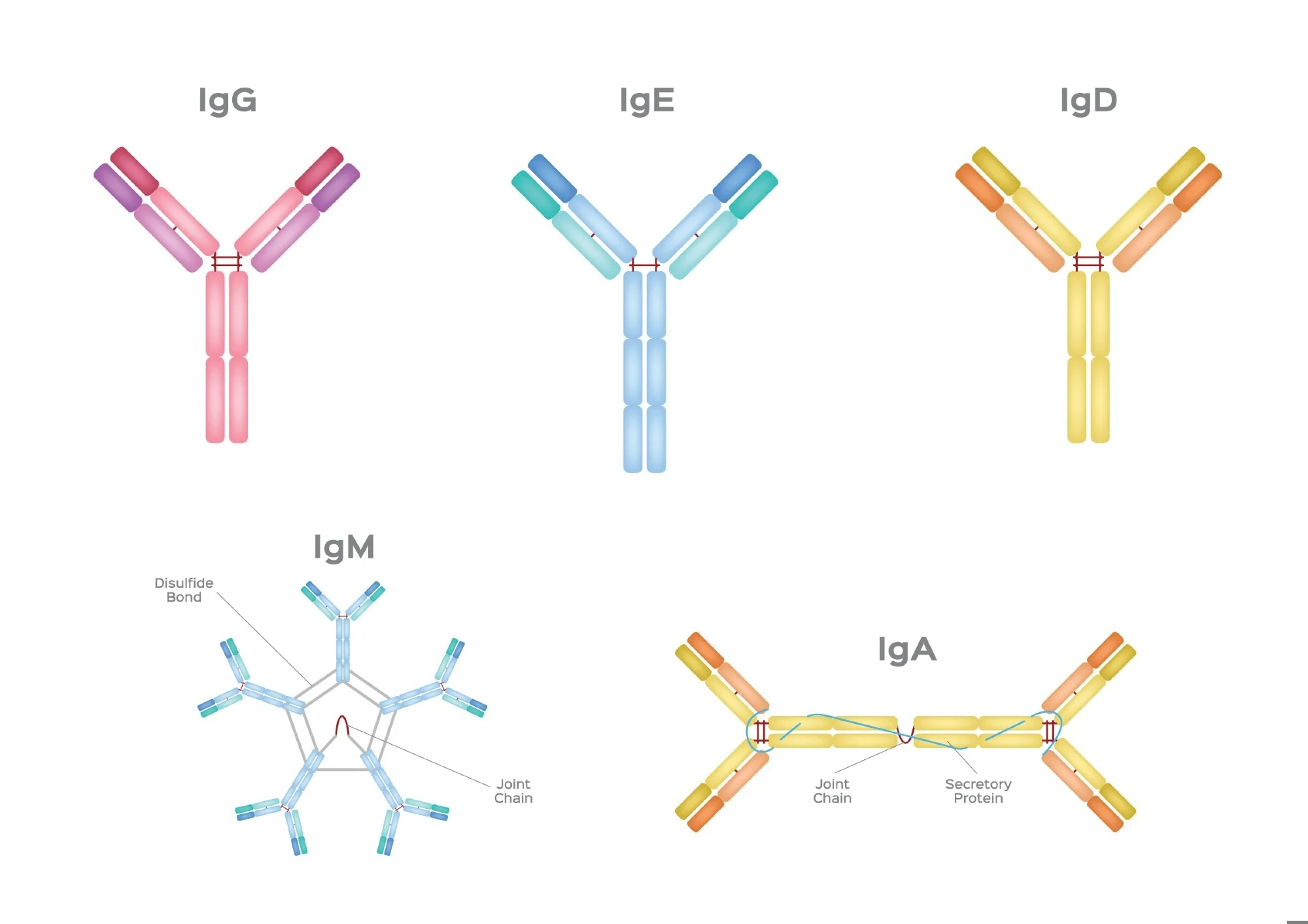
IgG
what is involved in innate immunity
macrophages; neutrophils- pattern recognition
protective barriers
what is involved in Adaptive immunity?
b-cells- recognise soluble antigens
T-cells- recognise foreign molecules expressed by self-cells
what are 3 key characteristics of adaptive immunity?
memory- cleared faster on reinfection
specificity to single antigen
discriminate between self (host) and non-self (foreign) cells
role of B-cells
bind to complementary antigens
clonal expansion into daughter cells that differentiate
memory cells
plasma cells- secrete antibodies
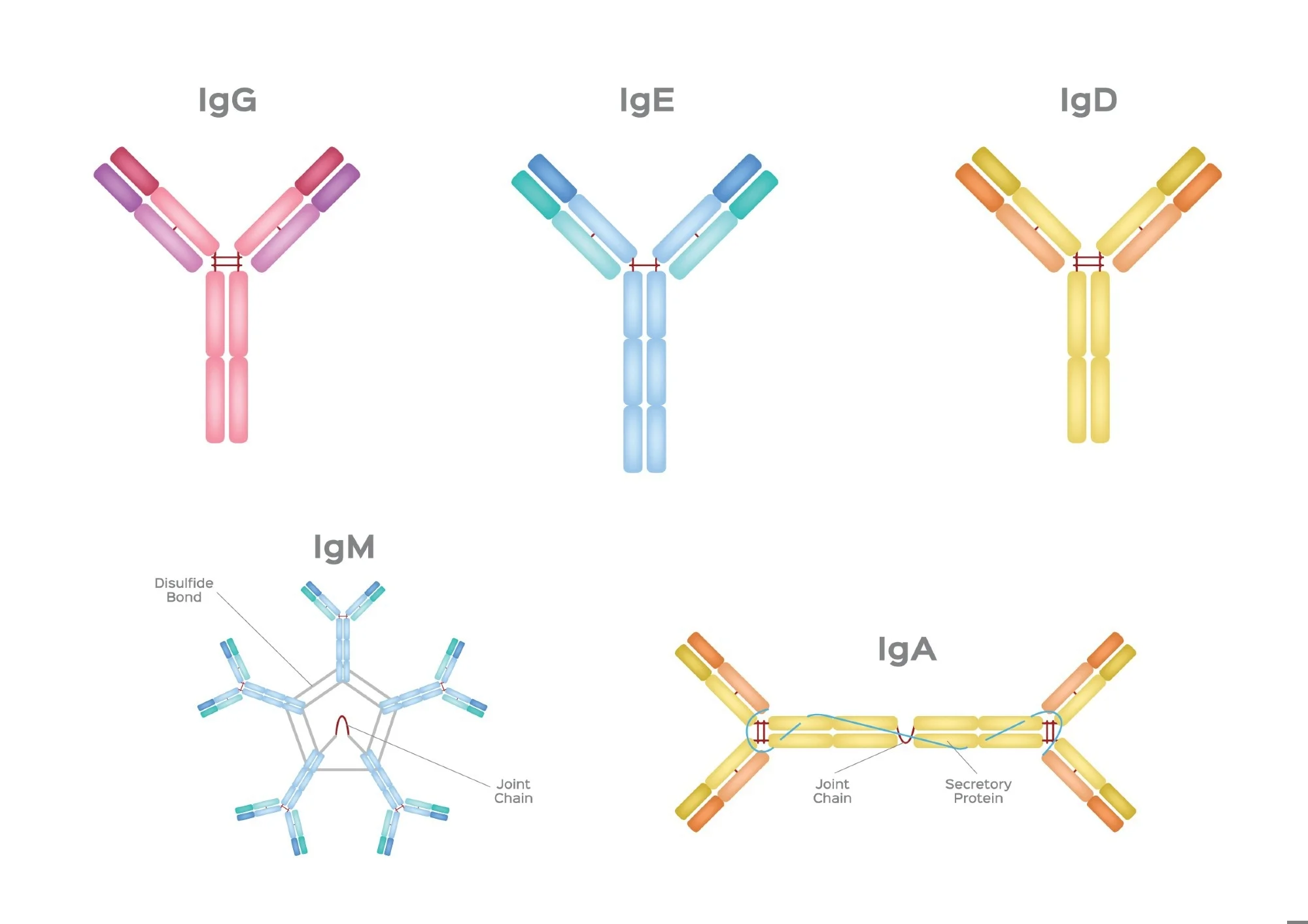
IgG
blood, tissue, placenta
fix complement
bind phagocytes
neutralise toxins