phase transformations exam 3
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Spinodal decomposition occurs when a homogeneous phase becomes thermodynamically unstable (composition fluctuations cause a ______ in free energy), but nucleation and growth occur when a homogeneous phase becomes metastable and a secondary phase would be more stable (fluctuations cause a local _____ in free energy)
maximum, minimum
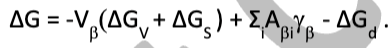
In what type of interface do you consider Gs contribution to change in free energy during a phase transformation?
coherent
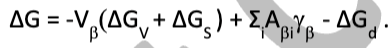
Define the Gd contribution
elimination of defect involved with heterogeneous nucleation
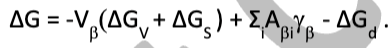
What factors hinder the secondary phase precipitation?
interfacial energy
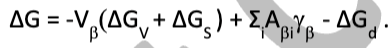
What factors drive the secondary phase precipitation?
Gv, Gs, and Gd
Vacancies, Dislocations, Stacking Faults, Grain Boundary Corners, Grain Boundary Faces, External Surfaces, and Internal Porosity
list the highest to lowest potency of the sites above for heterogeneous nucleation
external surfaces, internal porosity, GB corners, GB faces, stacking faults, dislocations, vacancies
To determine the critical value at which heterogeneous nucleation dominates precipitation rate, you take the ratio of what two values?
Nhet/Nhom (known boundary thickess over grain size, exp term must be larger than that ratio)
Assuming that changes in the molar volumes of primary and secondary phases can be neglected, the solute diffusion-controlled growth rate of incoherent flat interface is _____
parabolic
At low driving force for nucleation, grain ____ nucleation dominates. As driving force increases, grain edges and then ____ dominate as nucleation sites.
corner, boundaries
Partly coherent interfaces migrate by ______ ______, but incoherent interfaces are highly mobile.
ledge mechanism
When do you typically see planar incoherent interfaces?
after grain boundary nucleation
What is it called when the diffusion fields of separate particles begin to overlap and growth decelerates rapidly until it stops.
soft impingement
Given a needle-like precipitate with a curved end, will the concentration of solute at the interface be more or less than the equilibrium composition?
more
For a given plate precipitate thickness, what is the relationship between lengthening rate and time?
linear (or constant)
Why is thickening of precipitates by ledge migration so difficult?
nucleation mechanisms cannot supply ledges with constant spacing
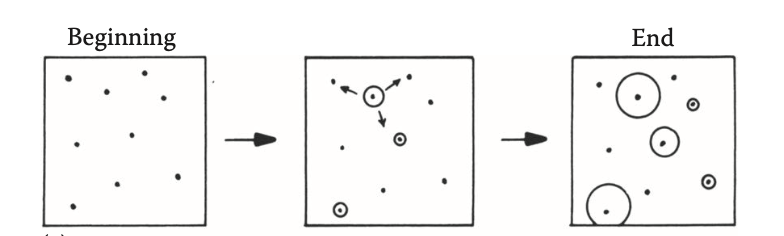
describe the nucleation rate
constant
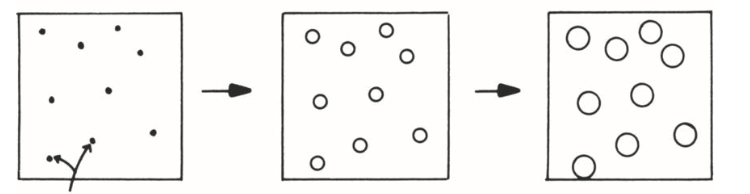
describe the nucleation rate, what is this phenomenon called?
nucleation only occurs at beginning of transformation, site saturation
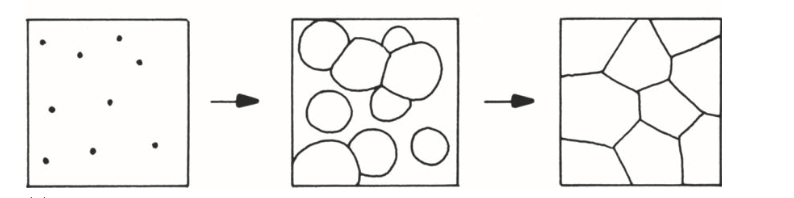
what type of transformation is this?
cellular, due to hard impingement
GP zones are ____ with the matrix and thus have a very low interfacial energy. What type of nucleation do they seem to undergo?
coherent, homogeneous
If an alloy is heated to a temperature above the GP zone solvus line then the zones will dissolve, what is this process called?
reversion
Close to a grain boundary the vacancy concentration will be the equilibrium value for the aging temperature, but away from the boundary it will be that for the ____ ______ temperature. Thus the precipitate free zone (PFZ) forms on the ______.
solution treatment, boundary
The width of the PFZ is narrower at ___ temperatures and ___ quench rates. They can also form on ____.
low, high, defects
Spinodal decomposition occurs when the alloy is unstable because small fluctuations in _____ that produce A-rich and B-rich regions will cause an overall decrease in free energy.
composition
Spinodal decomposition occurs for any alloy composition where the free energy has a _____ curvature.
negative
Spinodal decomposition occurs by “____-hill” diffusion, nucleation and growth occurs by “__-hill” diffusion
up, down
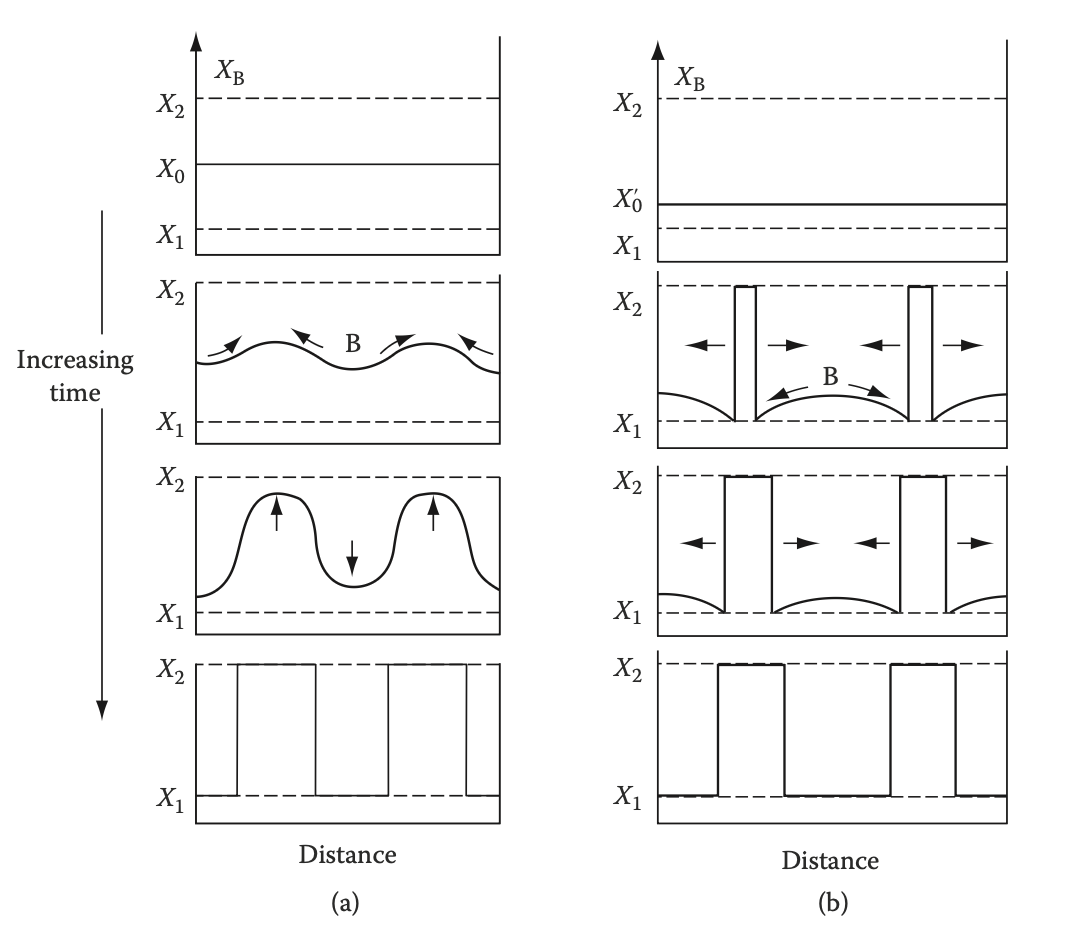
which one is spinodal decomposition and nucleation and growth?
a=sd, b=ng
During spinodal composition, fluctuations in composition ____ exponentially with time and the interdiffusion coefficient is ____ than zero.
increase, less
The miscibility gap on a phase diagram corresponds to the equilibrium compositions of _____ phases. The one defining _____ phases is the result phases from spinodal decompositions.
incoherent, coherent
A high density of small precipitates will tend to coarsen into a lower density of larger particles with a ____ total interfacial area. What is this phenomenon called?
smaller, ostwald ripening
Growth behind a planar incoherent interface can be simplified using a _____ approximation
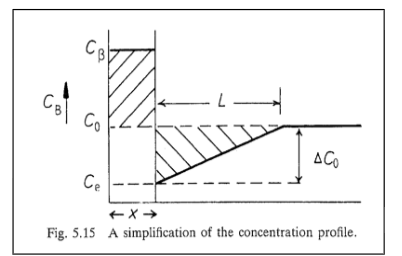
Zener
For a C-curve shaped TTT diagram transformed fraction f is defined at the volume of ____ at time t divided by the final volume of ___(same as before)
beta
For a S-shaped f(t,T) vs log(t) diagram the transformed fraction f is defined by the ____ equation, useful if there are multiple ____ mechanisms
avrami, growth
There is a Tmax corresponding to a maximum growth rate vmax, when T>Tmax then the growth rate is _____ limited, but when T<Tmax then the growth rate is _____ limited.
thermodynamically, kinetically
Nucleation rate depends on the _____ ______ for creation of a critical volume of nucleus and the activation energy for _____ _____
activation barrier, atom migration
Rate of isothermal transformation is determined by nucleation and growth, but overall fraction of material volume transformed is dominated by ______.
growth
There is a maximum growth rate between T=0 and T=___. There is a maximum nucleation rate between T=0 and T=___.
Te, Tm
When considering the nucleation rate, when T is close to Tm, there is a large activation barrier G* and ___ thermodynamic driving force. When T « Tm, there is ___ mobility of atoms and also a ___ rate of nucleation.
small, low, small
____ impingement refers to when the growth of a new phase is restricted by the presence of existing phases that act as obstacles to the growth process.
hard
When aging occurs at a temperature above the GPZ solvus line, there is a ____ peak hardness during aging; a _____ dispersion of particles and _____ number fraction of precipitates.
lower, coarser, smaller
What type of precipitate can form by spinodal decomposition, i.e. there is no nucleation barrier?
GPZ
The Gibbs–Thomson effect causes the solute concentration in the matrix adjacent to a particle to increase as the radius of curvature ______.
decreases
The microstructure that forms due to spinodal decomposition is uniform and unaffected by grain boundaries, thus there is no formation of _____
PFZ
In age hardening of Al-Cu alloys, peak age hardening is achieved as
(i) a single phase with coherent interface
(ii) a single phase with semi-coherent interfaces
(iii) a mixture of two phases, one with coherent and one with semi-coherent interfaces
iii
In age hardening, overaging is associated with the formation of ____ precipitates, what else results in the loss of hardness?
incoherent, greater precipitate spacing
For constant molar volumes, it can be assumed that Xb=Cb(__) and that (Cb-Ce)=(Cb-___)
Vm, Co
The concentration gradient and thus supersaturation _____ with time because solute is drained from the supersaturated matrix, parent phase.
decreases
For needle (or plate) shaped precipitates the lengthening and thickening rates show different growth rate characteristics. The interface sections associated with the head/cap of the needle shape precipitate are typically ______ and have curvature with radius of curvature r, where r is of magnitude approximately equal to ____ of the width of thickness of the needle (or plate).
incoherent, half
Lengthening involves _____-controlled migration of a curved incoherent interface section. Here the interfacial curvature related to the Gibbs Thomson effect or capillary force (surface tension) ___ the chemical potential derived thermodynamic driving force for migration, which is proportional to the supersaturation ∆Xo.
diffusion, opposes