BIOL2021 - Chapter 17 - Cell cycle
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
What is the correct order of mitosis?
Prophase
Prometaphase
Metaphase
Anaphase
Telophase
What happens in prophase of the cell cycle?
Replicated chromosomes condense into sister chromatids
Mitotic spindles assemble between centrosomes
What happens in prometaphase of the cell cycle?
Nuclear envelope breaks down
Chromosomes attach to spindles at their kinetochores
What happens in metaphase of the cell cycle?
Chromosomes align at the equator
Kinetochores attach chromatids to opposite poles of the spindle
What happens in anaphase of the cell cycle?
Sister chromatids separate towards poles and form daughter chromosomes
Kinetochore microtubules get shorter
What happens in telophase of the cell cycle?
Daughter chromosomes arrive at poles and decondense
Nuclear envelope reassembles
contraction ring forms
What happens during cytokinesis in the cell cycle?
Cytoplasm is divided into two by the contractile ring
Cell divides into 2 daughter cells
Which does a cell spend more time doing; growing and increasing protein mass and organelles, or undergoing cell division?
Growing and increasing protein mass and organelles
When does cell growth occur?
During interphase
What happens during S phase?
Chromosome (DNA) replication
Replication DNA held together as sister chromatids
Centrosome replication in animal cells
What happens during M phase (M for mitosis)?
Mitosis
Cytokinesis
What are the 4 phases of the eukaryotic cell cycle?
G1 (gap 1) phase
S phase
G2 (gap 2) phase
M phase
What happens during gap phases (G1 and G2)?
Growth
creating proteins and organelles required for next stage
Monitoring readiness
Which phases are included in Interphase?
G1
S
G2
What phase does a cell enter of conditions/signals are not suitable for cell division?
G0 phase/quiescence (not dividing or preparing to divide)
What is a post differentiated state?
A state a cell can be in
a G0 phase where the cell cannot re-enter the cell cycle
What are senescence cells?
Type of G0 cell
Will not re-enter the cell cycle
Caused by stress
What system triggers the major events of the cell cycle?
The cell-cycle control system
When and what are the main checkpoints in the cell cycle control system
G2/M transition; all DNA is replicated and favorable environment
Begin Anaphase and cytokinesis; Are chromosomes attached to spindle
Start cell cycle/S phase; Favorable environment
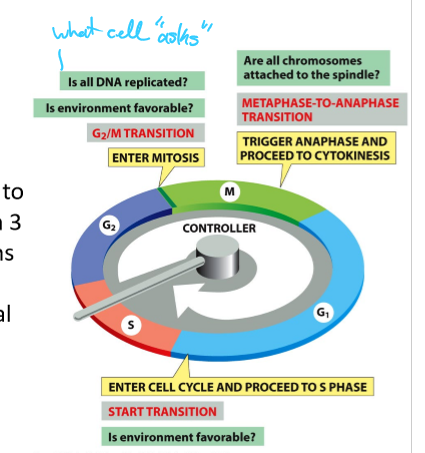
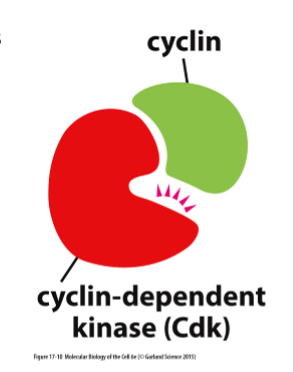
What are the “master regulators” of the cell-cycle control system?
Cyclin-dependent kinases (Cdks)
What are Cdks regulated by?
Cyclins (major regulators)
Phosphorylation (G2/M transitions)
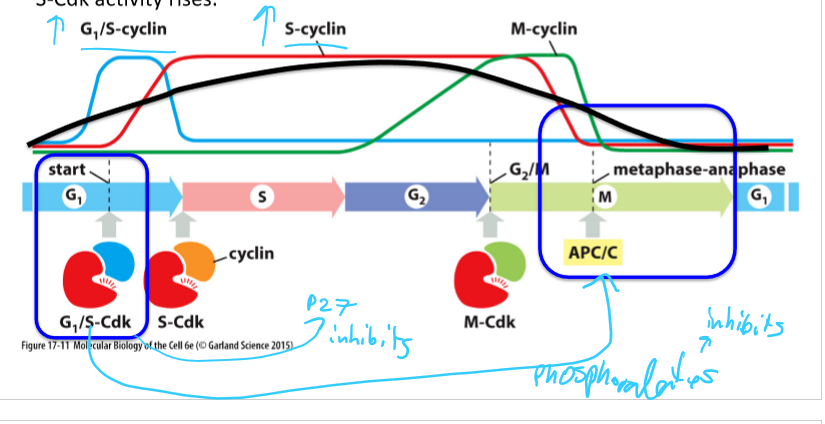
How do Cdk protein levels vary through the cell cycle?
They stay constant
How do cyclin protein levels vary during the cell cycle (in general)?
Rise and fall by synthesis and degration
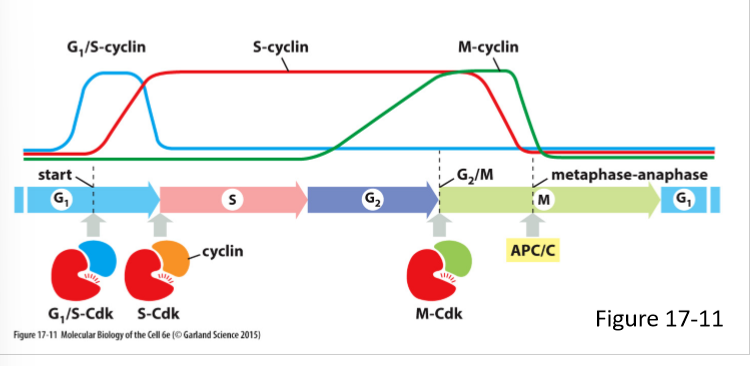
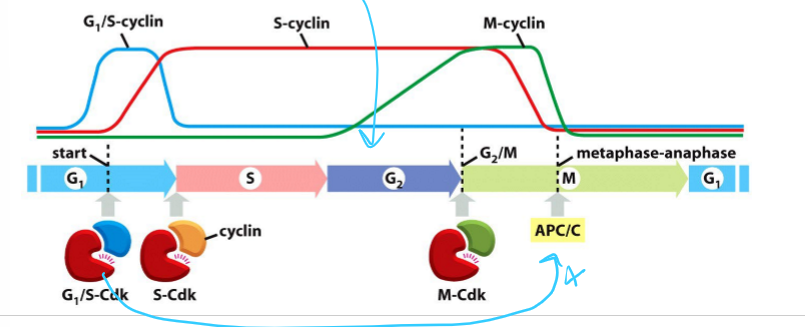
What are the 3 main classes of cyclins? What do they form?
G1/S-cyclins; G1/S-Cdk complex
S-cyclin; S-Cdk complex
M-Cdk; M-Cdk complex
G1-cyclin;
How do G1-cyclin levels vary in the cell cycle? What do they control?
Rise gradually during G1 phase and fall during M phase
Regulate G1/S-Cdk activity
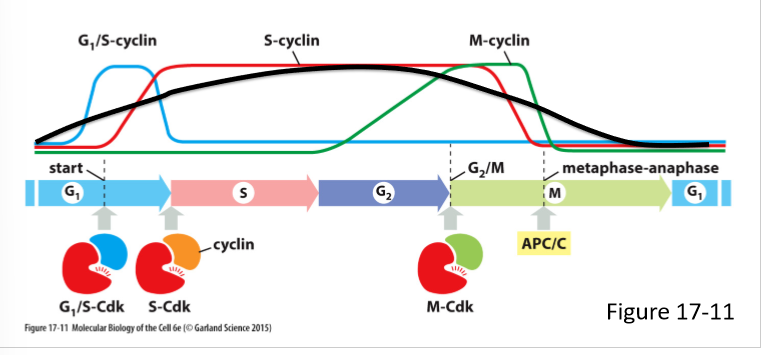
True/False; All eukaryoticncell have G1/S cyclin, S-cyclin, M-cyclin, and G1-cyclin
False; only most have G1-cyclin
How do cyclins work?
Bind and activate kinases that phosphorylate proteins related to cell cycle progress
What do G1/S-cyclin do? How does its concentration vary in the cell cycle?
Forms G1/S-Cdk complex
Activates Cdk in late G1 and helps trigger progression through the start
Help commit to cell-cycle entry
Levels rise before the start in G1 phase, and fall in S phase
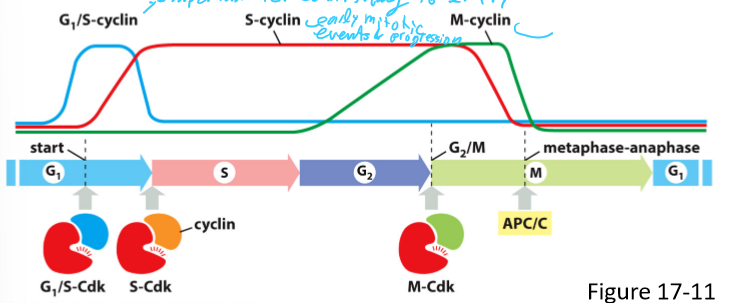
What do S-cyclin do? How does its concentration vary in the cell cycle?
Forms S-Cdk complex
Binds Cdk soon after progression through the start of the cell cycle
Helps stimulate chromosome duplication (S phase) and some early mitosis activities
Levels rise after the start in G1 phase and remain elevated until mitosis
Contributes to the control of some early mitotic events
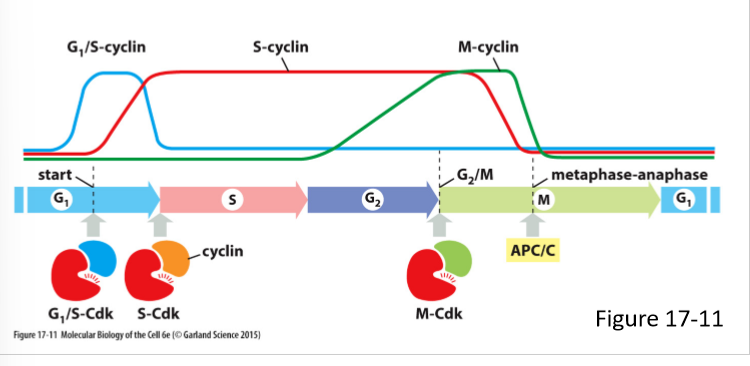
What do M-cyclins do? How does its concentration vary in the cell cycle?
Forms M-Cdk complex
Activates Cdk that stimulate entry into mitosis at G/M transition
Levels rise in G2 phase and fall mid mitosis (just before metaphase-anaphase transition)
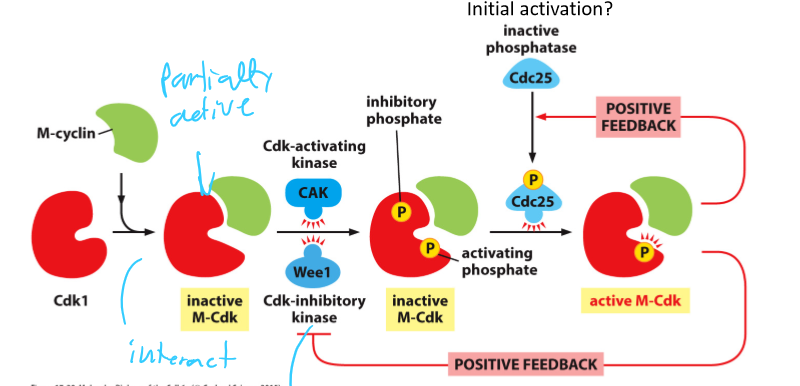
What is CAK and what does it do?
Cdk-activating kinase (CAK)
Phosphorylates Cdk active site
Induces full activity
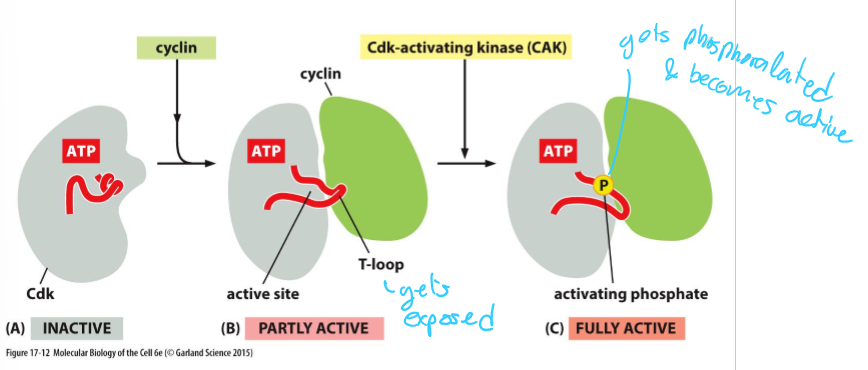
What part of a Cdk does cyclin bind/attach to?
The T-loop
What is required for Cdk to be fully active?
Cyclin binding
Phosphorylation by CAK
What does Wee1 do to a Cdk complex?
Adds 2 inhibitory phosphatases to the roof of its active site
Inactivates it
What phosphatase removed inhibitory phosphates from a Cdk complex?
Cdc25 phosphatase
What are CKIs and what do they do?
Cdk inhibitor proteins
suppressed Cdk activity
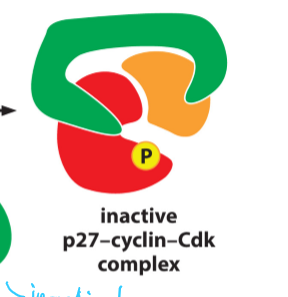
What is p27 and what does it do?
a CKI
Binds to both Cdk and cyclin
Changes conformation of its active site
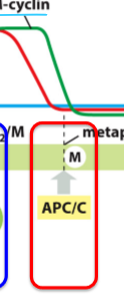
What is the Met-Ana transition triggered by?
Proteolysis (protein degradation) by Anaphase-promoting complex or cyclosome (APC/C)
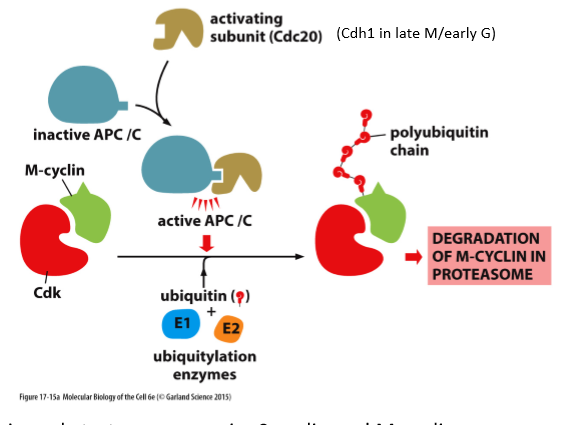
What are Anaphase-promoting complex and Cyclosome (APC/C) and what do they do?
Ubiquitin ligase
Initiates anaphase and destruction of M and S cyclins
Remains active into G1
What controls levels of cyclin?
Transcriptional regulators
What Cdk activity happens during the start of the cell cycle in G1?
G1-Cdk stimulates transcription of G1/S and S-cyclin genes
G1/S-Cdk activity inactivates APC/C (phosphorylation) and CKIs that inhibit S-Cdk, initiates centriole duplication in animal cells at the start of S phase
S-Cdk activity rises
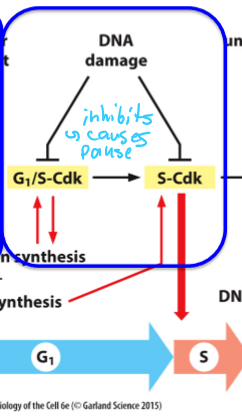
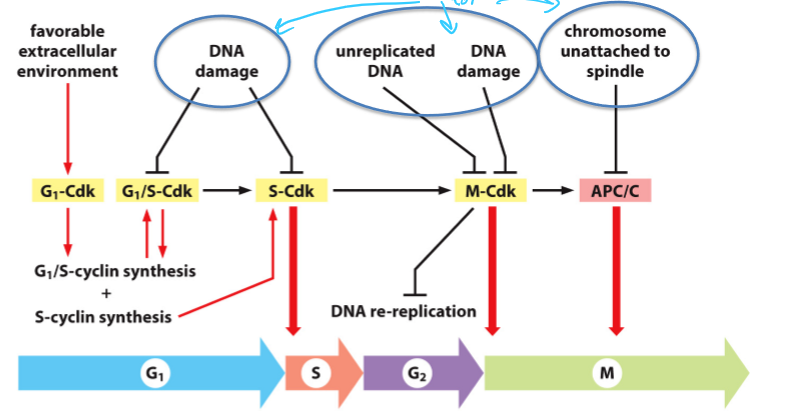
What Cdks and stage of the cell cycle is blocked by DNA damage?
G1/S-Cdk and S-Cdk are inhibited, which blocks the start (in G1)
M-Cdk is inhibted,which blocks the transition into M
What is DNA replication and Histone synthesis initiated by and how?
S-Cdk; phosphorylates proteins that activate helicase and recruit DNA synthesis machinery, which increases histone synthesis
How many times does S-Cdk initiate DNA replication in the cell cycle?
1
What does DNA replication start with?
DNA helicase; Unwinds double-stranded DNA
How is it ensured that chromosome duplication only occurs once per cycle?
Initiation phase of DNA replication is divided into 2 distinct steps
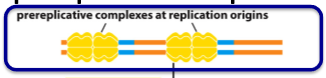
What are prereplicative complexes?
A pair of inactive DNA helicases loaded on the replication origins from late mitosis to early G1
What happens when DNA helicases are activated by S-Cdk?
DNA winding
Initiation of DNA synthesis
What do DNA helicases move out of the replication origins with? How does the ensure replication only happens once?
They move with the replication forks, meaning the origins cannot be reused until new replicative complexes form at the end of mitosis
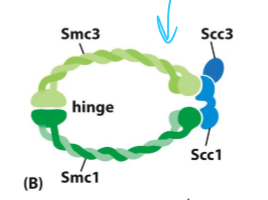
What are sister chromatids are held together by?
Cohesin
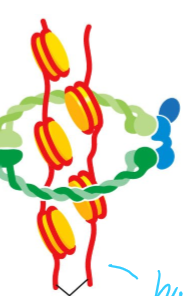
What is cohesin composed of?
two Smc molecules (Smc3 and Smc1) held together at their hinge
An Scc1 bound at the ATPase domain of the Smc, with an Scc3 bound to it
When does M-Cdk become active?
At the G2/M transition
What fully activates M-Cdk at the onset of mitosis?
Dephosphorylation of the inhibitory phosphate by Cdc25
What Cdk and stage does unreplicated DNA inhibit?
Inhibited M-Cdk, which blocks the transition into M
What early mitotic events does M-Cdk activate?
Chromosome condensation
Nuclear envelope breakdown
Separation of centrosomes and setting up mitotic spindle
How does M-Cdk help chromosome condensation and nuclear envelope breakdown?
Phosphorylates condensins
Phosphorylates nuclear pore complex (NPC) subunits, causing disassembly
Phosphorylates nuclear Lamins, causing disassembly
How does M-Cdk drive formation of the mitotic spindle?
By phosphorylating:
Microtubule associated proteins and other proteins to create shorter, more dynamic microtubules
Centrosome proteins; more gamma-tubulin complexes, more microtubule polymerization
Motor proteins; centrosomes move apart, spindle sets up
What is the bipolar spindle set up by?
Microtubules
Motor proteins
What happens during the formation of the mitotic spindle?
Kinetochore microtubules + ends attach to sister chromatids
Astral microtubules radiate out from spindle pole and contact the cell cortex (actin filaments under plasma membrane)
Interpolar microtubules interdigitate at the equator and cross link with other microtubules
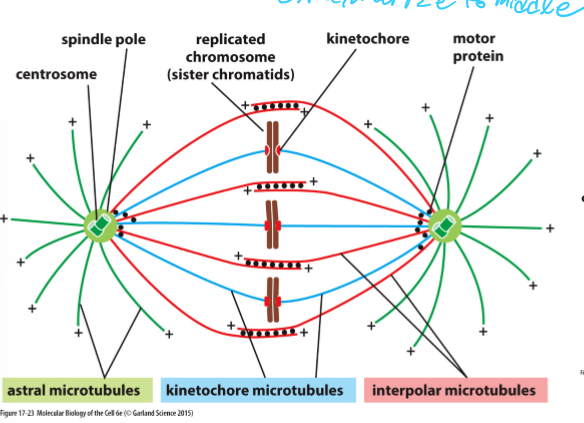
When does spindle assembly begin?
Early mitosis
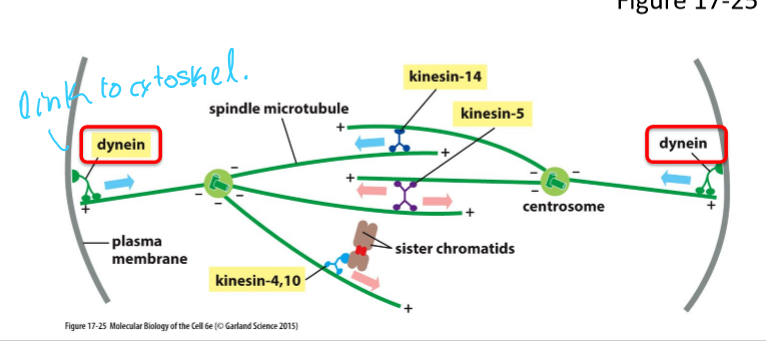
What do kinesin-5 motors do and what are they activated by?
Phosphorylated by M-Cdk and other protein kinases
Drive centrosome separation
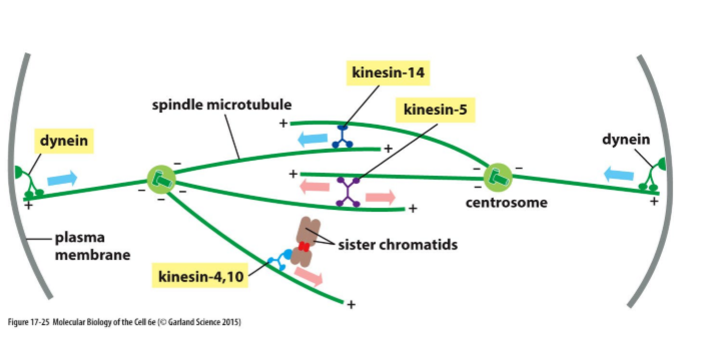
What do microtubule-dependent motor proteins govern?
Spindle assembly and function
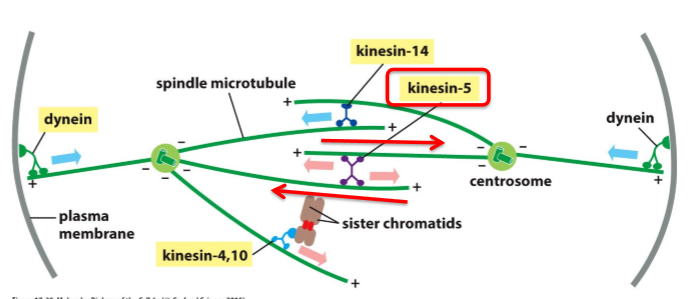
What is Kinesin-5 and what does it do?
Motor protein with 2 motor domains that move toward + end
Pushes poles of the cell apart
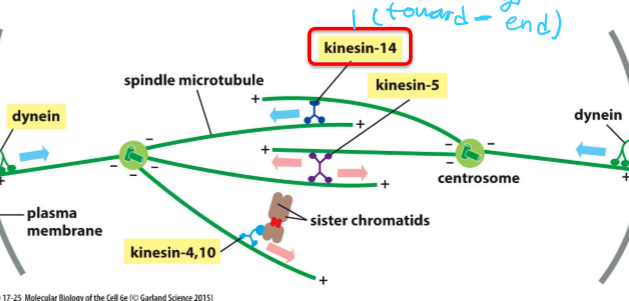
What is Kinesin-14 and what does it do?
Single domain motor protein
Interacts with antiparallel microtubules
Pulls poles together by moving towards - end
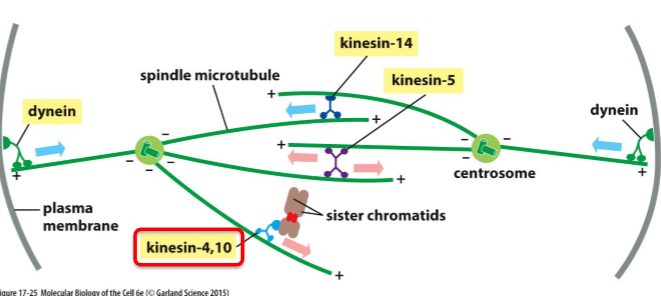
What are Kinesin-4 and Kinesin-10 and what do they do?
Type of chromokinesins
Move towards + end
Associated with chromosome
Push chromosome away from pole
What role does Dynein have in spindle assembly and function?
Link astral microtubules with actin cytoskeleton at the cell cortex
Pulls spindle poles towards cell cortex and away from each other, by moving towards - end
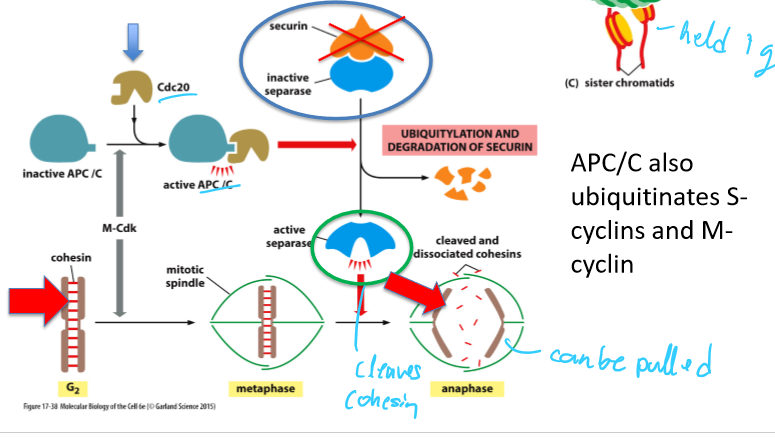
What does APC/C trigger?
Sister chromatid separation (anaphse)
Completion of mitosis
What is APC/C activated by?
M-Cdk or Cdc20
What does APC/C activation by Cdc20 trigger?
Destruction (ubiquitination) of securin (releases separase) and cyclins
Leads to sister chromatid separation and completion of mitosis
What molecules does APC/C ubiquitinate?
Securin
S-cyclins
M-cyclins
What is the key regulator of the M-A transition?
APC/C
Which of the following is most likely to be inhibited at the M-A checkpoint if chromosomes are improperly attached?
APC/C complex
What would be likely to happen during mitosis to a mammalian cell that would normally be dividing if you injected it with high levels of a mutant securin protein that could not be ubiquitlated, keeping levels of active securin high at all times?
The chromosomes would not be able to separate in anaphase
What does Cdk inactivation allow for, before the completion of mitosis?
Allows phosphatases to dephosphorylated
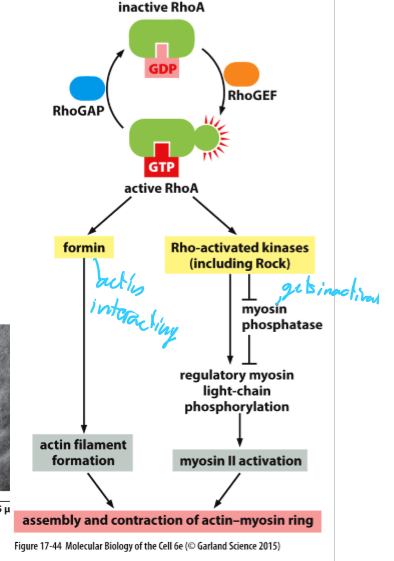
What happens during cytokinesis?
Rho-A GEF activates Rho-A at cell cortex
This activates
formin; stimulates actin filament polymerization
Rho-activated kinases, which activate myosin II
What allows for cells to escape from G1 or G0?
External signals
Unicellular organism; Nutrient
Multicellular organism; Mitogen
What is a mitogen?
An external signal that can allow cells to escape from G1 or G0, to stimulate cell division, in multicellular organism
What stimulates cells to grow?
growth factors
What signals cells to survive/suppress apoptosis?
Survival factors
What happens in the Mitogen pathway?
Mitogen interacts with a mitogen receptor
Mitogen receptor interacts with Ras
Ras interacts with MAP kinase
Map kinase causes activation of transcription regulatory protein
Causes gene expression
Transcription regulatory protein is synthesized
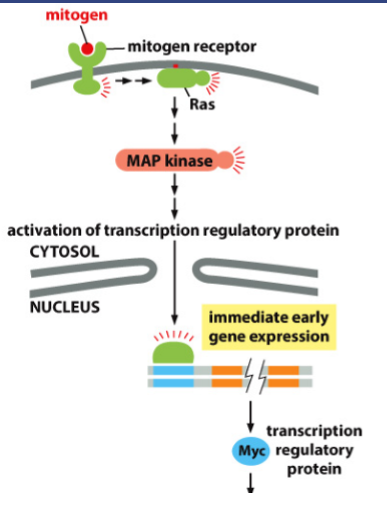
What does Myc activate?
Transcription of G1-cyclin, which activates G1-Cdk, leading to transcription of other genes involved in cell growth and cell division, including G1/S cyclin.
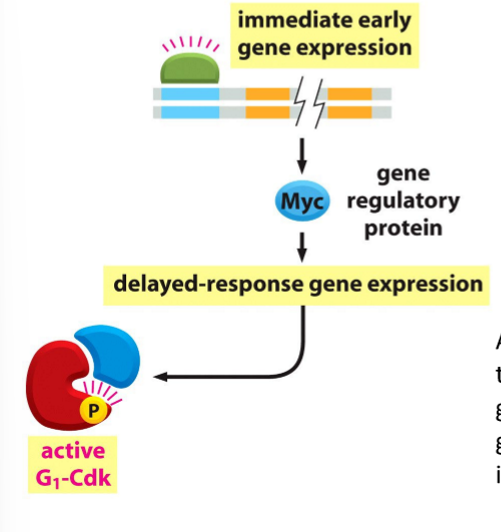
What are E2Fs?
Gene regulatory proteins
Bind promotors to stimulate transcription of G1/S cyclin, S-cyclin, DNA synthesis proteins, and other proteins required for S-phase
What are E2F proteins regulated by?
Rb proteins, which are inactivated by phosphorylation by G1/Cdk
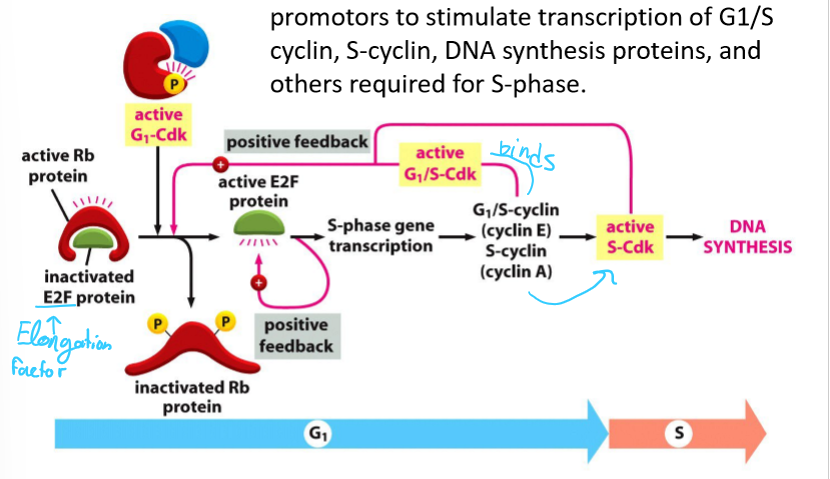
Why do activated APC/C and CKI levels need to be regulated? What are they regulated by?
APC/C will ubiquitylate S-cyclin
CKI will inhibit Cdks
Both are phosphorylated by G1/S Cdk
What happens if problems are detected at checkpoints in the cell cycle control system?
Transition is delayed
Inhibitory mechanisms act on cyclin complexes
What happens of DNA damage is detected by protein kinases?
p53 is phosphorylated, removing Mdm2
active p53 bids to regulatory region of p21 gene, transcribing it
CKI is produced
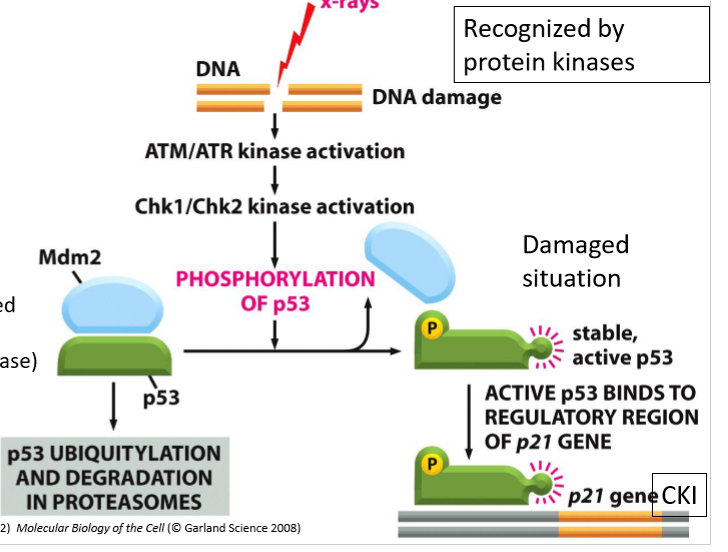
What happens to Mdm2 and p53 in cells with undamaged DNA?
Mdm2 tags p53
p53 is ubiquitinated and degraded by proteasomes
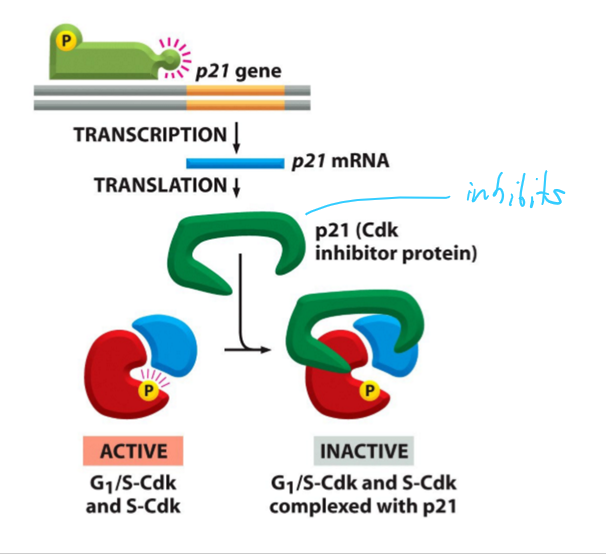
What is p21?
A CKI (Cyclin-dependent kinase inhibitor proteins) for G1/S-Cdk and SdK
Why are p53 mutations commonly found in cancers?
p53 stops the cell cycle until DNA damage is repaired
p53 failure means that damaged DNA will be replicated
extending damaged DNA into other cells increases the number of mutated cells and can lead to cancer
How many cancers is p53 mutations found in?
Half of all cancers
True/False; Many pathways stimulate cell division and cell growth
True