APSC 278 - Ceramics
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
what are applications for ceramics?
glasses (glasses, glass ceramics)
clay products (structural clay products, whitewares)
refractories (clay, nonclay)
abrasives
cements
ceramic biomaterials
carbons (diamond, graphite, fibers)
what is a ceramic?
“burnt material”
the desirable properties of ceramics are typically developed through a high-temperature heat treatment process known as firing
what is the composition of ceramics?
metallic + nonmetallic, two nonmetallic elements, larger multi-ion atoms
what are the bonds in ceramics?
range from purely ionic to purely covalent
could have a combination of both bonding types, with the degree of ionic character depending on the difference in electronegativity between the atoms
what is the AX type of ionic ceramic crystal structure?
metallic ions are cations (A) and nonmetallic ions are anions (X)
equal number of cations and anions
can form several crystal structures, each named after a material that exhibits that structure
eg.: NaCl, CsCl, and ZnS
what are the AmXp and AmBnXp types of ionic ceramic crystal structure?
if the charges on the cations and anions are not the same, a compound can exist with
AmXp formula, e.g.: AX2: CaF2, ZrO2, and ThO2
AmBnXp formula (A and B are cations) eg.: BaTiO3
what are silicates?
covalent ceramics composed primarily of silicon and oxygen
eg.: soils, rocks, clays, and sand
basic building block: (SiO4)4 tetrahedron
one Si atom with an O atom bonded to each orbital
each O- ion is left with a single negative charge
equal separation and distribution of the 4 O atoms in space (tetrahedron shape)
different silicate structures exist that are the basis of commercial glasses
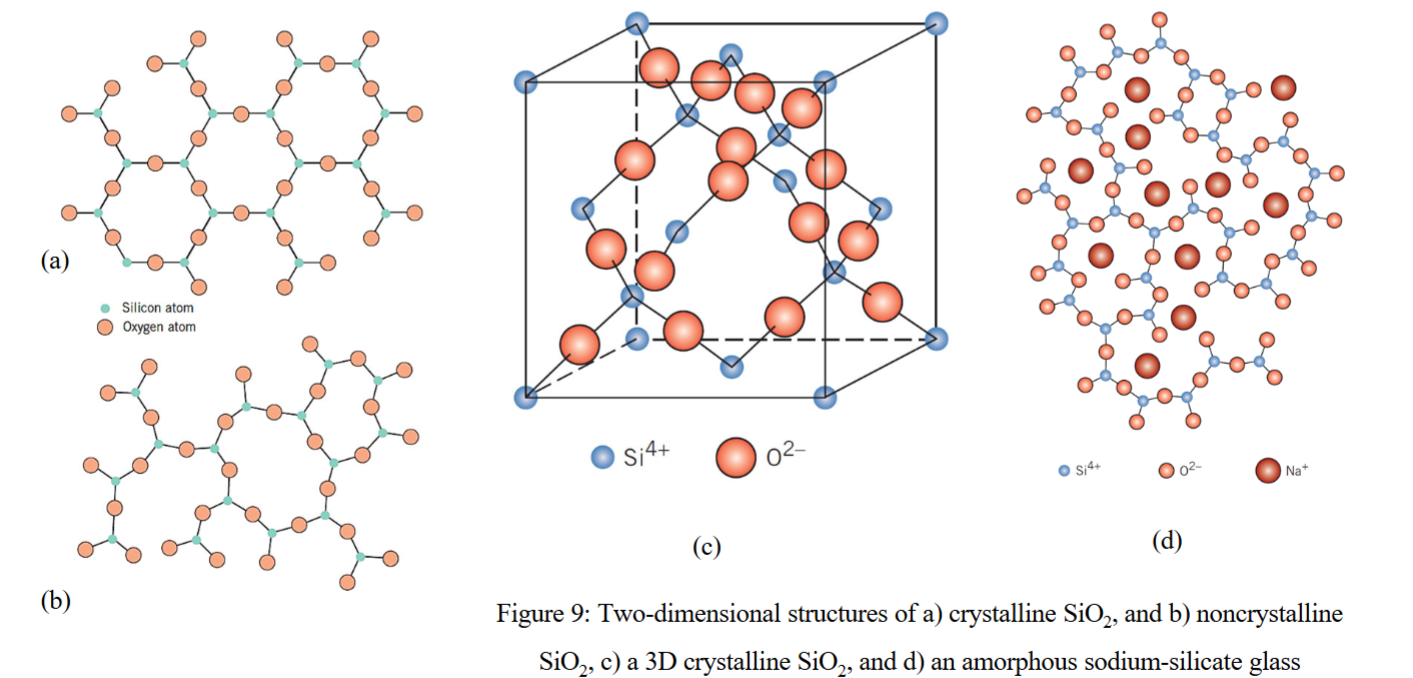
what is the simplest silicate?
silicone dioxide (silica / SiO2)
3D network, where the corner O atoms in each tetrahedron are shaped by adjacent tetrahedra
if tetrahedra are arrayed in an ordered manner, a crystalline structure is formed
can also be a non crystalline or amorphous solid (glass) with atomic randomness
potential silica crystalline structures
what are network modifiers?
oxides that have been added to modify the SiO44- network to form inorganic glasses
what is Na2O (sodium oxide, “soda”)?
a network modifier that reduces the glass viscosity and melting point, allowing the glass to be formed at lower temperatures
what is CaO (calcium oxide, “lime”)?
a network modifier that strengthens the glass structure and improves durability
what is Al2O3 (aluminum oxide)?
a network modifier that increases hardness and scratch resistance
what is K2O (potassium oxide)
a network modifier that creates compressive stresses
what network modifier makes up 90% of all commercial glasses?
soda-lime (sodium oxide - calcium oxide)
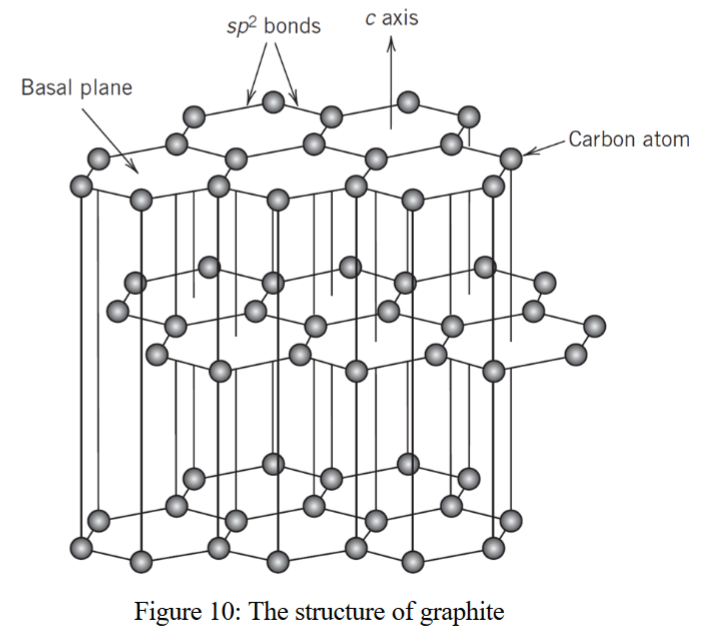
what is carbon as a covalent ceramic structure?
carbon exists in two allotropic forms: diamond and graphite
in graphite:
C atoms are located at corners of interlocking regular hexagons that lie in parallel (basal) planes
strong, covalent bonds between carbon atoms on the base planes
weaker van der waal bonds between layers
what are example applications of carbon as a ceramic structure?
lithium-ion battery anodes
solid lubricant
carbon fiber composites in aircraft structures
smartphones and laptops
what are advantages of properties of ceramics?
high melting point (excellent thermal insulator)
high creep resistance
high elastic modulus
high hardness and wear resistance
excellent compressive strength
chemically inactive (resistant to most acids, alkalis, and organic solvents)
high electrical resistivity
what are disadvantages of properties of ceramics?
low ductility (very brittle)
low fracture toughness (~0.5 MPa m0.5)
potentially difficult to manufacture
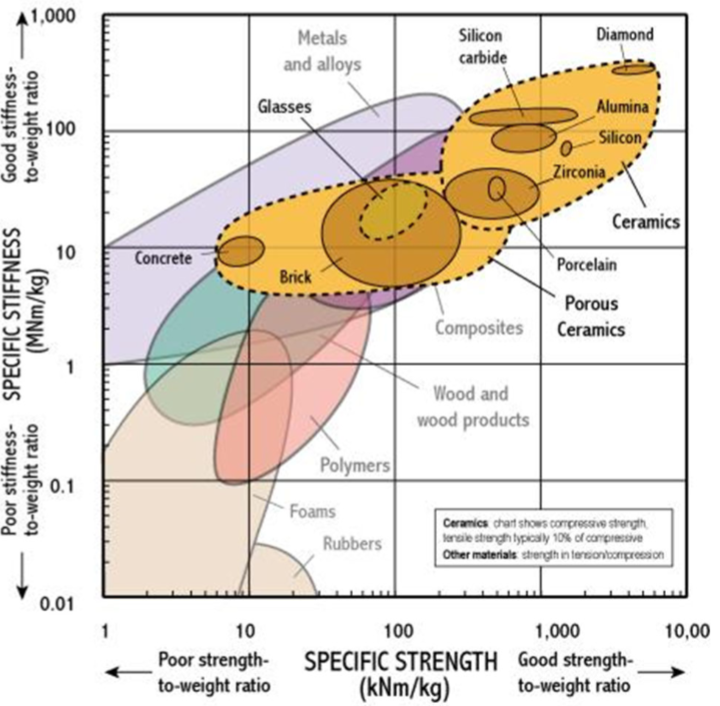
specific strength vs specific stiffness plot for ceramics
classification of ceramic fabrication techniques
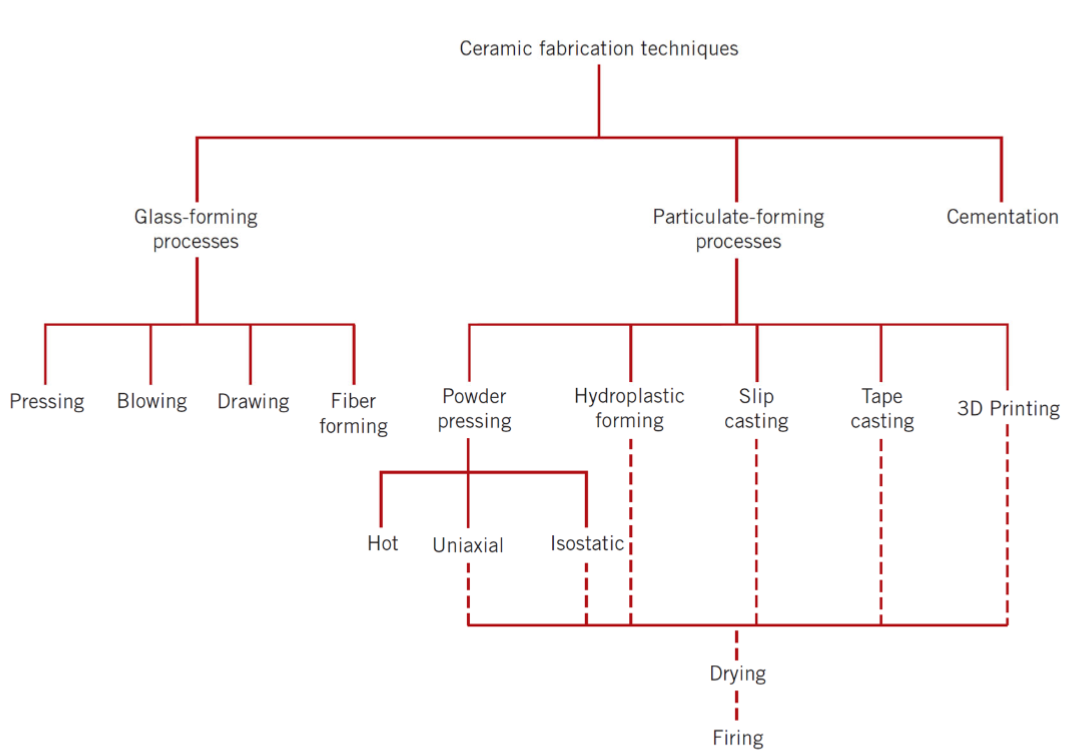
what are the steps in the particulate forming process?
powder
forming
drying
firing
what is the first step of the particulate forming process?
powder
mill (grind) and screen constituents to obtain the desired particle size
minerals and materials like clay, silica, ect. extracted from the earth and ground into powder
what is the second step of the particulate forming process?
forming
mix the powder with water or keep dry
press or cast the powder into the desired shape
resulting part is still soft and pliable
what is the third step of the particulate forming process?
drying
remove the residual water before firing
what is the fourth step of the particulate forming process?
firing
fire at high temperatures for vitrification or sintering of powder
what are the three main ways to shape ceramic powder into the desired form in the forming stage?
powder pressing
hydroplastic forming
slip casting
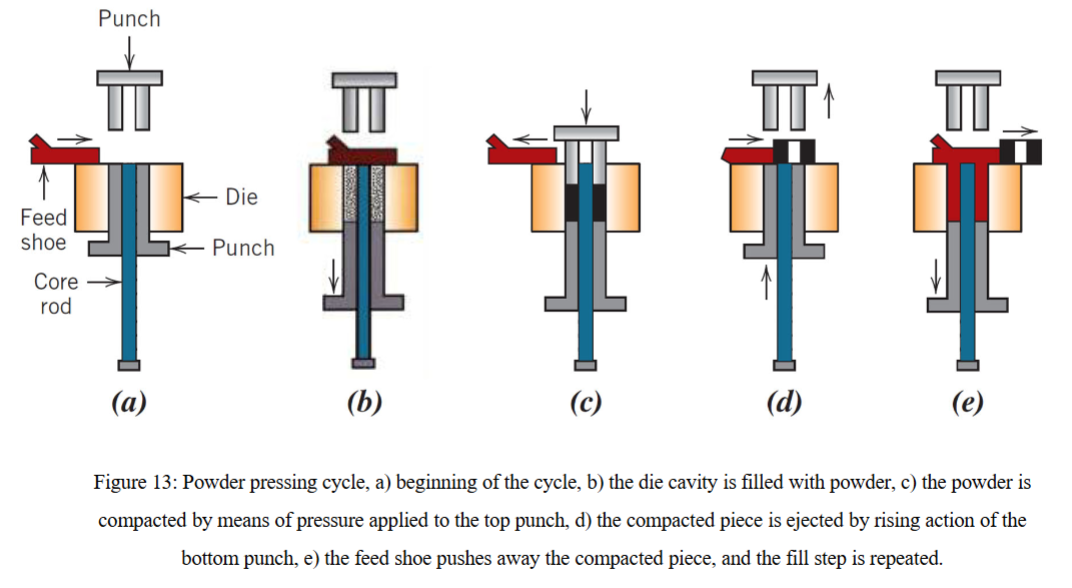
what is powder pressing?
can be used for both clay and non-clay compositions (such as metals)
dry process, no water added
what is hydroplastic forming?
when mixed with water, clay is highly plastic and may be molded without cracking
yield strength is low but sufficient enough to permit a form ware to maintain its shape during handling and drying
extrusion is the most common technique
a stiff plastic ceramic mass is forced through a die orifice having the desired cross-sectional geometry
eg.: brick, pipe, ceramic blocks, tiles
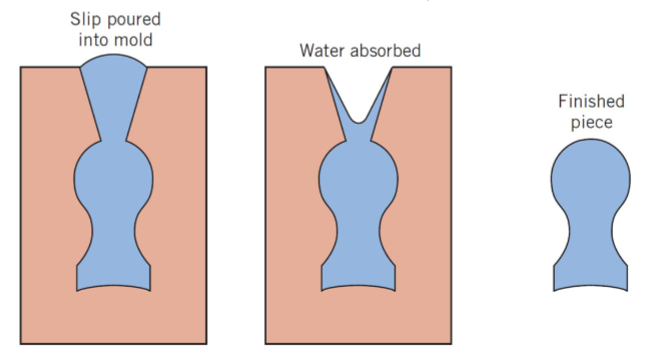
what is slip casting?
for clay based compositions
slip poured into a porous mold, water is absorbed from the slip into the mold, and leaves behind a solid layer on the mold wall, and the thickness depends on time
what is solid casting?
repeated slip casting process until the entire mold cavity becomes solid
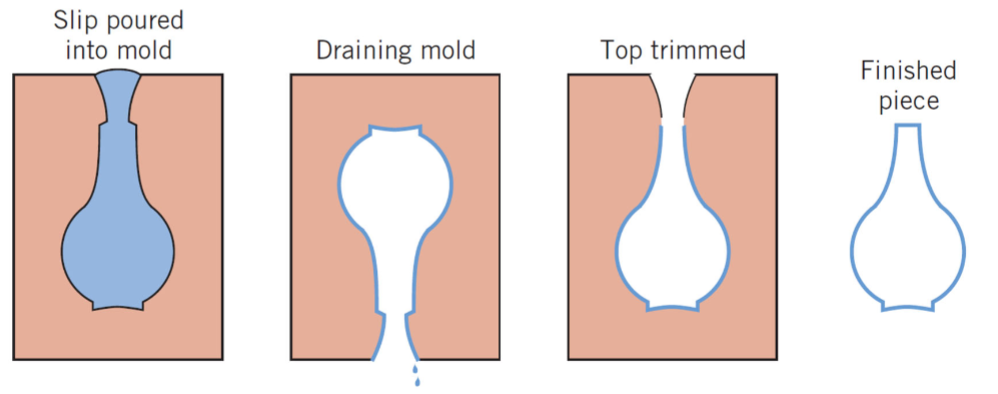
what is drain casting?
terminating the slip casing process when the sold shell wall reaches the desired thickness by inverting the mold and pouring out the excess slip
as the cast piece dries and shrinks, it pulls away from the mold wall and the mold may be disassembled and the cast piece removed
what happens during the drying and firing process?
as water is removed and drying progresses, the interparticle separation decreases and shrinkage occurs
if drying is occurs too quickly, the sample may warp or crack due to non-uniform shrinkage
what are outcomes from heat treatment between 900 - 1700°C?
sintering
vitrification
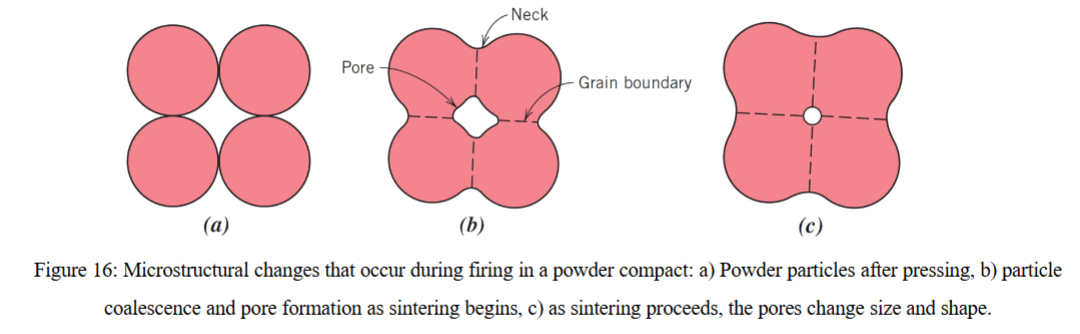
what is sintering?
occurs when a firing piece below the melting temperature (no liquid is formed) and where the piece has been powder pressed (no added water)
the particles coalesce due to an overall reduction in surface area due to surface energy
gaps between particles are also reduced
microstructural changes that occur during firing in a powder compact
SEM micrograph of after sintering a powder compact

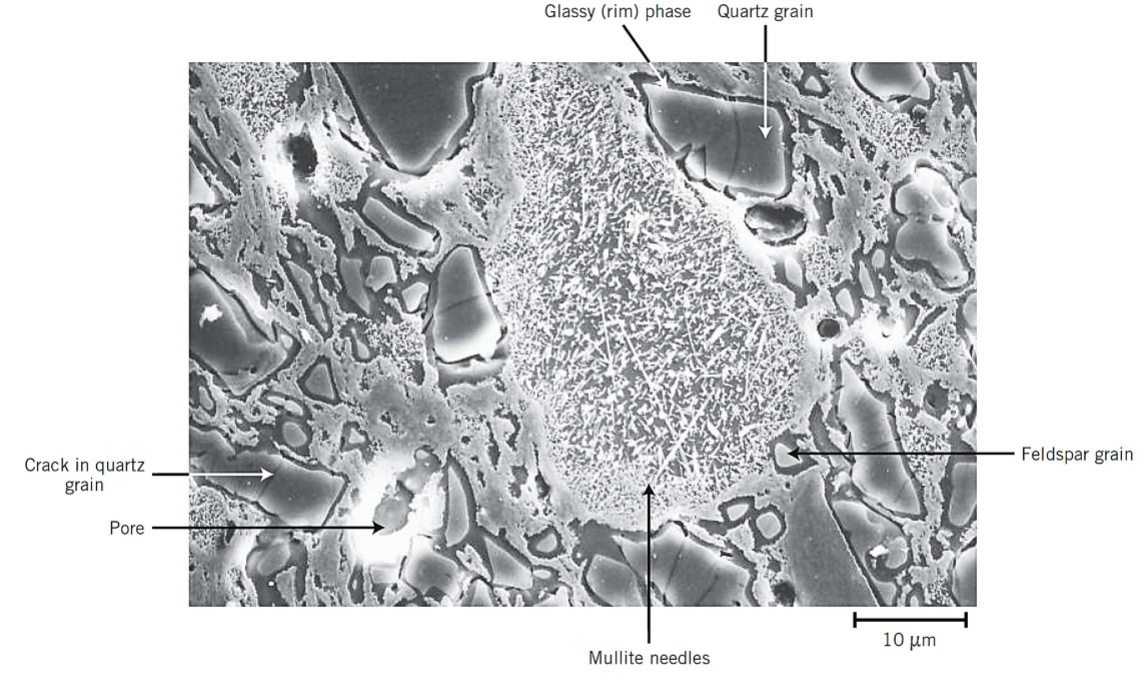
what is vitrification?
the gradual formation of a liquid glass that flows into and fills some of the pore volume
temperature at which the liquid phase forms is lowered by the addition of fluxing agents
fused phase flows around the remaining unmolten particles and fills in the pores through capillary action
shrinkage also occurs during this poress
upon cooling, this fused phase forms a glassy matrix that results in a dense, strong body
SEM of fired porcelain
how do glassy materials solidify compared to crystalline materials?
glass becomes more viscous as temperature decreases, and there is no specific temperature at which liquid transforms to solid, as with crystalline materials
what is needed to form distinct crystallographic structures involving SiO44- tetrahedra
extremely slow cooling - time for the tetrahedra to organize into the complex crystal structures
nucleating agents are added - assists in forming the initial crystallization sites (phosphorus and boron can do this for glass)
silica can exist in a supercooled liquid below melting temperature
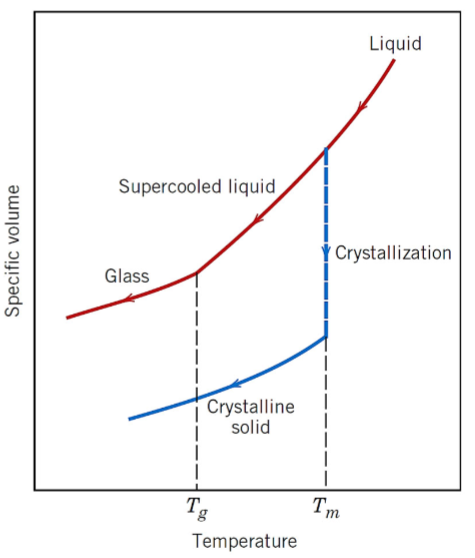
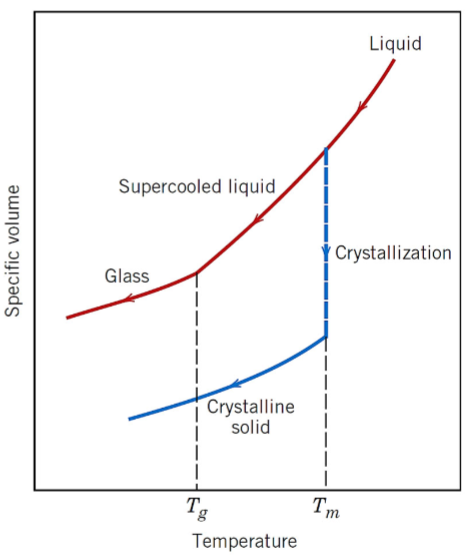
explain the specific volume vs temperature behaviour of crystalline and non crystalline materials using the graph (Tg = glass transition temperature, Tm = melting temperature)
crystalline materials solidify at the melting temperature
for cooling of a supercooled liquid, a change in slope occurs at Tg (glass-transition temeprature)
below Tg, the material is considered a glass
above Tg, it is considered a supercooled liquid (cooled below its conventional Tm)
during heating, if the material is in crystalline solid form, it melts when it reaches the Tm
what is the effect of temperature on viscosity?
viscosity increases with decreasing temperature
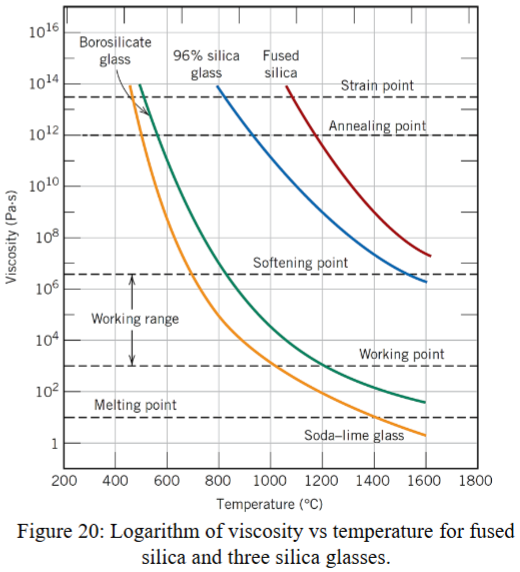
what is the strain point?
the temperature at which the viscosity is v = 3×1013Paꞏs
the glass will fracture without plastic deformation under this temperature
what is the annealing point?
v = 1012 Paꞏs
sufficient atomic diffusion to remove residual stress
what is the softening point?
v = 4×106 Paꞏs
maximum temperature at which glass can be handled without significantly deforming it
what is the working point?
v = 4×106 Paꞏs
the glass is easily deformed
what is the melting point?
10 Paꞏs
the point where glass is considered liquid
what are thermal stresses?
internal stresses developed during cooling from high temperatures
arise from different cooling rates and thermal contraction between the surface and interior'
ceramics are cooled slowly to avoid thermal stresses
what is thermal shock?
when the material is weakened or fractured due to high thermal stresses
how can thermal stresses be reduced if they are already present?
heat the glass to the annealing temperature
cool slowly to room temperature
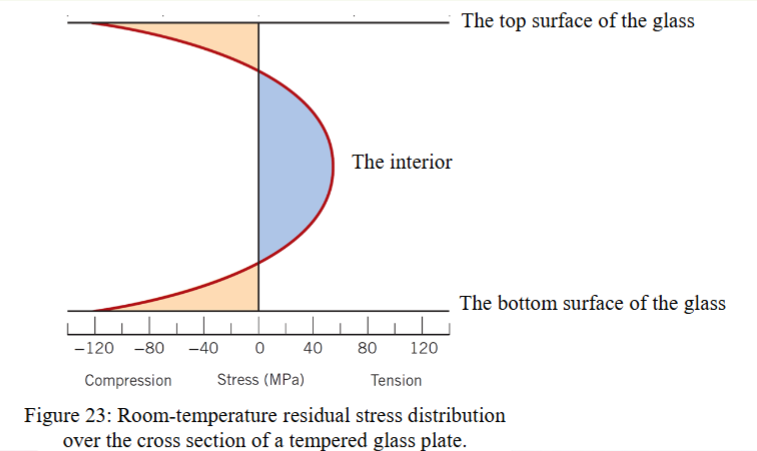
what is thermal tempering?
heat treatment that induced compressive residual stresses at the surface to increase the strength of glass
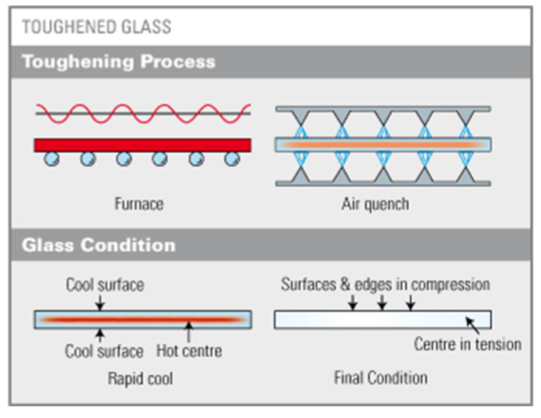
how does thermal tempering work?
the glass is heated above the glass transition temperature but below the softening point
glass is rapidly cooled using air jets or sometimes an oil bath
the surface cools faster and becomes rigid first
the interior cools more slowly and remains plastic for a longer time
as the interior cools and contracts, it pulls against the rigid surface
results in compressive stresses at the surface and tensile stresses in the interior
room temperature residual stress distribution over the cross section of a tempered glass plate