Environment - PATH2221
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
Name 6 environmental causes of disease
Air pollutants
UV radiation
Heavy metals and toxins
Smoking and alcohol
Therapeutic drugs
Nutrition
What is carbon monoxide?
Odorless, non-irritating gas produced by incomplete oxidation of carbon material
How does carbon monoxide cause disease?
It binds to haemoglobin with 200-fold stronger affinity (carboxyhaemoglobin) which prevent delivery of oxygen to tissue
What does carbon dioxide bind to other than haemoglobin?
Myoglobin: induces myocardial depression, hypotension and arrhythmias. Ferrous heme of cytochrome c oxidase: inhibits mitochondrial respiration
What brain injury occurs following carbon monoxide poisoning?
Bilateral necrosis (movement impairment); acute hypoxic-ischemic injury (neuronal cell shrinkage); Perivascular haemorrhage in cerebral cortex
How is carbon monoxide poisoning treated?
Breathing pure oxygen or hyperbaric oxygen therapy
What is the difference between ionising and non-ionising agents
Ionizing is extremely harmful due to it's ability to displace electrons from atoms
How does UV cause pathology?
UVA rays penetrate deeper into tissue layers of eyes and skin than UVB (more energy). Dimer formation is induced because nucleotides are chromophores which can cause frameshift or point mutation
What are the 3 most commonly caused cancers by UV radiation?
Melanoma (high survival)
Basal cell carcinoma (fleshy bumps)
Squamous cell carcinoma (red scaly, can spread)
How can UV radiation increase infection?
Compromises epidermal barrier function so bacteria and allergens can penetrate
How can immune function relate to UV caused cancer?
UV-induced immune suppression of adaptive immunity has shown to contribute to skin cancer development
How can UV radiation direct increase immune system functions?
Can activate Treg cells and modulation of antigen presenting cells; can increase B cell activation (IL-10 increase) and suppress dendritic cell function
How can UV radiation decrease immune function?
Can reduce the sensitisation to antigens and recall immune responses (fewer memory cells); can reactivate viral infections
Outline the steps of UV radiation inducing immune suppression
Keratinocytes (when damaged by UV) produce and release immunomodulators which stimulate langerhans cells, platelet activating factors and mast cells -> increased IL-10 and IL-4
What is the impact of UV radiation on autoimmunity?
Can exacerbate due to damage of cellular components that are engulfed by macrophages and induce autoantibody development
How does UVR impact dermatomyostisis (autoimmune disease)
UVR induced immune suppression leads to an inability for the immune system to target and eliminate autoantibodies
How can UV radiation be used to treat autoimmune diseases?
At specific wavelengths it can suppress neuroinflammation in patients with clinically-isolated syndrome
What is the effect of methylmercury on bodily function?
90% absorbed by GI tract = precipitation of proteins in intestinal cells; cleared by kidneys = renal damage; sensitive developing brain = loss of function
Describe the effect of methylmercury on cellular function
Strong affinity to sulfhydryl groups (particularly cysteine which is abundant in proteins throughout the body); so function of any cellular/subcellular structure may be impacted
What are the main cellular impacts due to methylmercury?
Damage organelles, free radical generation, impaired cell membrane integrity, immune dysfunction neurotransmitter disruption, inhibit enzyme function, impaired protein synthesis, impaired cell metabolism
What are the key steps of neurotoxicity?
High concentrations of glutamate at the synaptic cleft acts as a toxin (increased Ca+ influx) and occurs because methylmercury inhibits glutamate uptake
How is mercury poisoning treated?
Chelation because chelating agents bind to sulfhydryl groups that will bind to mercury
How does lead cause blood abnormalities?
It has an affinity for sulfhydryl groups and interferes with 2 enzymes important for haem synthesis. SO ZPP is formed instead of haem which decreases iron incorporation (anaemia)
How does lead poisoning specifically affect red blood cells?
Inhibits sodium and potassium dependent ATPases which increases RBC fragility, causing haemolysis
How are the cells morphologically affected by lead poisoning?
Microcystic, hypochromic anaemia with distinct punctuate basophilic stippling of red cells
What neurotoxicity does lead poisoning cause?
More likely in children as blood-brain barrier is less defined.
Changes to CNS include brain oedema, demyelination in cerebrum and necrosis of neurons.
In adults it can affect PNS resulting in loss of motor function (footdrop)
How does lead poisoning cause neurotoxicity?
Lead mimics calcium and blocks the neuron, weaker and fewer and inappropriate signals
What happens to asbestos fibres once they are inhaled?
Alveolar macrophages attempt to remove the asbestos fibres via phagocytosis but is unsuccessful with larger fibres = frustrated phagocytosis and triggering of inflammatory mediators and ROS production
How can asbestos fibres be measured?
They accumulate iron on their surface - ferruginous bodies. This also leads to ROS production.
Name the diseases caused by asbestos
Asbestosis
Mesothelioma
Lung adenocarcinoma (cancer)
What is asbestosis?
Interstitial fibrosis that worsens dyspnea, cough and pulmonary hypertension
What is mesothelioma?
Cancer of the mesothelial cells that form the lining layers of the viscera, perisistant inflammation leading to DNA damage
What is lung adenocarcinoma?
5 fold higher risk of lung cancer due to asbestos exposure
How does smoking contribute to the development of cancer?
Carcinogens in tobacco smoke leads to metabolic activation, DNA adducts and therefore miscoding leading to mutations in multiple genes
what are the issues of nicotine (the main chemical in tobacco)?
Highly toxic, highly addictive and a stimulant
How does smoking tobacco contribute to the development of pulmonary disease?
Chronic pulmonary obstructive disease (COPD) is limited airflow due to exposure to noxious particles/gas
What is emphysema pathogenesis?
Permanent enlargement of the alveoli/terminal bronchioles due to parenchymal destruction by oxidative stress, inflammation and protease imbalance
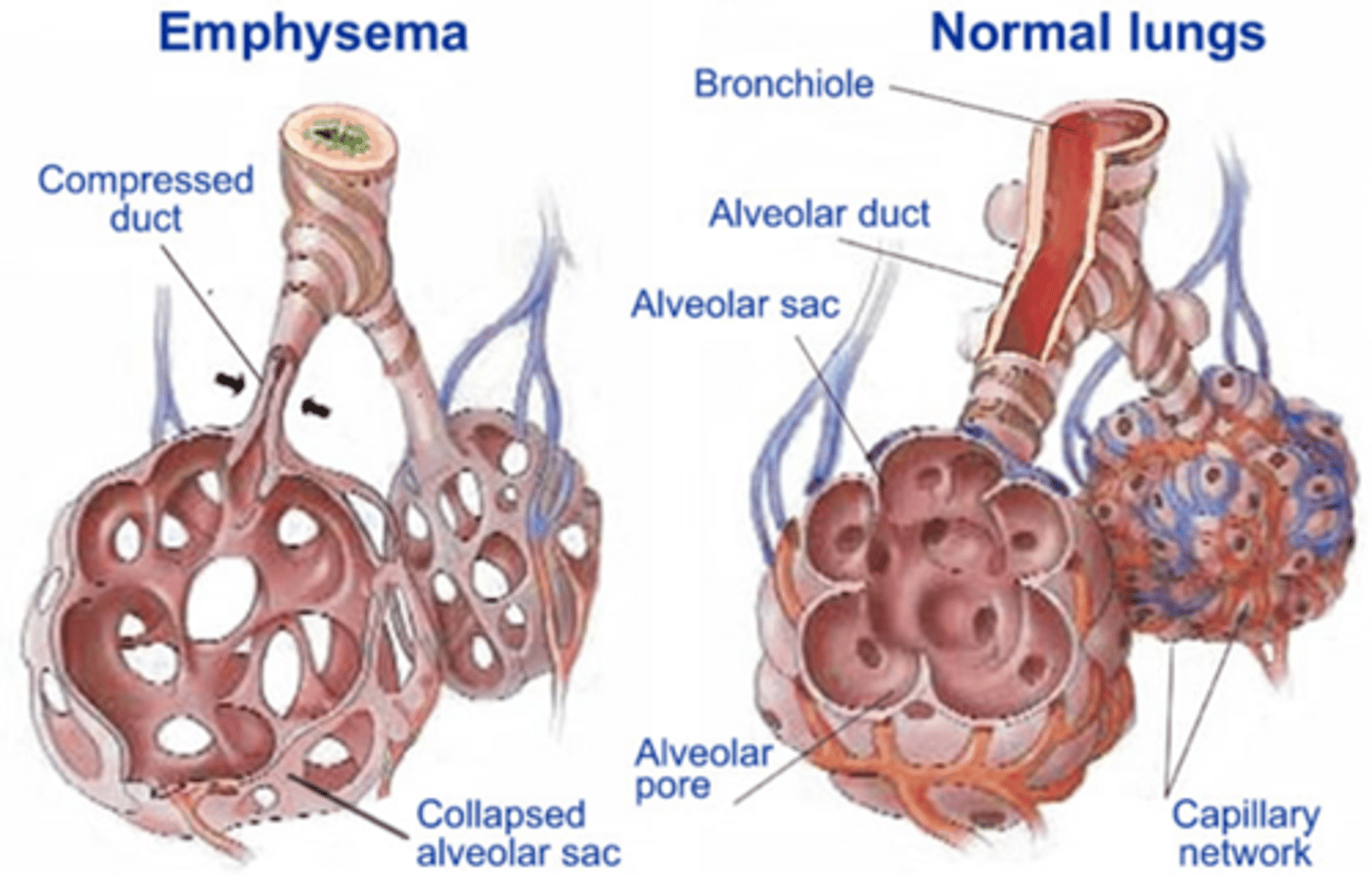
What is chronic bronchitis?
Hypersecretion of mucus because smoking increases number of goblet cells, smooth muscle hypertrophy and inflammation of bronchi/bronchioles
How does smoking contribute to cardiovascular disease?
Increases the risk of atheroclerosis, coronary heart disease, stroke, peripheral arterial disease, abdominal aortic aneurysm
What is atherosclerosis pathogenesis?
Increases platelet aggregation, decreased myocardial oxygen supply, decreased threshold for ventricular fibrillation
How does carbon monoxide from smoking affect pregnancy?
Forms carboxyhaemoglobin which results in impaired oxygen delivery to myometrium and placenta = fetal growth restriction and preterm birth
How do carcinogens from smoking affect pregnancy?
Act as teratogens causing potential birth defects and oxidative stress
How does nicotine from smoking affect pregnancy?
Direct effect on fetal and placental vasculature and disrupts fetal brain function/development
How many deaths are estimated to be caused by alcohol consumption?
WHO - 2.6 million (smoking is 8 million)
Outline the toxic effects of ethanol metabolism
Metabolised to acetaldehyde (toxic and carcinogenic), inflammation generates ROS leading to DNA adducts and fibrogenesis
What are the 3 different outcomes in the liver as a result of alcoholic liver disease?
Alcoholic fatty liver (hepatocytes contain lipid); Alcoholic micronodular cirrhosis (fibrous); hepatocellular carcinoma (thicker wall of trabeculae)
How does alcohol contribute to cancer development?
Acetaldehyde (produced from ethanol oxidation) generates ROS which impairs DNA repair and causes cell injury
What is the impact of alcohol on the brain (short term)?
Dopamine release
Impaired speech
Impaired motor and decision-making
Impaired memory creation
What are the impacts of alcohol on CNS (long term)?
Damage to white and grey matter = deficits in cognitive function. Alter adolescent brain development, contributes to dementia, neuroinflammation/nutritional deficiencies, reduced synaptic complexity decreasing brain volume
What are the effects of alcohol during pregnancy?
Crosses the placenta and can cause FASD which causes neurodevelopmental impairment
What are the mechanisms of alcohol teratogenesis?
Exposure during first-trimester organogenesis may cause brain, craniofacial, skeletal and internal organ dysmorphology
Describe what damage paracetamol causes
Overdose/repeat can lead to hepatotoxicity (acute liver failure) and death
How does paracetamol cause liver damage?
Breaks down into NAPQI which causes liver damage (oxidative stress, heptocyte necrosis etc.) but when in normal amounts it is metabolised into nontoxic metabolites. Liver damage can be repaired by release of DAMPs, immune activation and removal of cell debris
Explain the concept of nutrition
Necessary for normal body function and cannot be synthesised by the human body, so they must be consumed in food
What are the positive roles of nutrition across the lifespan?
Optimise overall wellbeing, prevent deficiencies, prevent loss of weight/muscle in illness and aged, maximise effectiveness of medicine, reduce inflammation, regulate gene expression
What is obesity?
A chronic complex disease defined by excessive fat deposits that can lead to diabetes, heart disease, cancer, quality of life
What are the common causes of malnutrition?
Poverty, Ignorance (of nutritional needs), Chronic excess alcohol use, acute and chronic illness (increased BMR), Dietary restriction
List the major sources of iodine
Seafood, iodised salt
What are the physiological roles of iodine for maintaining health?
Required for synthesis of thyroid hormones which regulate body function
Outline the consequences of iodine deficiency
Preventable brain damage and intellectual disability, chronic = goitre (thyroid gland enlargement)
What are the major sources of vitamin D?
Ergocalciferol (D2) from plants and fortified food; ;Cholecalciferol (D3) in animal tissue and sunlight
What are the physiological roles of vitamin D for maintaining health?
Helps kidneys control calcium levels (bone health), neurodevelopment, immunomodulation, regulation of cell growth and differentiation
What is the optimal dose of vitamin D?
50-70 nmol/L per day
What diseases are correlated with vitamin D deficiency?
Osteoporosis, rickets and osteomalacia
What are the major sources of vitamin A?
Animal foods, Carotenes (carrots, sweet pots, spinach); Breast milk
What is the physiological role of vitamin A?
Retinol/retinoic acid: Growth and development, immune system maintenance, good vision, skin support
What clinical complications does vitamin A deficiency lead to?
Premature death, increased infection, Ocular manifestations (eye disease)
What are the key sources of folate?
It is water-soluble vitamin B. From green leafy veg, fruits, cereals, grains, nuts, meats. Folic acid is the synthetic form.
What diseases does folate acid deficiency lead to?
Anencephaly (absence of parts of brain/skull); Spina bifida (defect in spinal column); encephalocele (protrusion of brain tissue through skull defect)
What benefits does folate acid fortification and supplementation bring?
Enhance cell proliferation and neural closure (or epigenetic regulation), decreased risk of recurrent neural tube defects when taken pre-conception
Name two common nutritional deficiences
Iron
Vitamin D
What are the two present forms of iron
Haem (easily absorbed)
Non-haem (less easily absorbed)
Why is iron important?
Critical part of haemoglobin
Outline iron homeostasis
It is absorbed into blood via the small intestine, in blood it binds to transferrin and is transported to the bone marrow (for haemoglobin and RBC synthesis); muscle (for myoglobin); liver (surplus iron forms ferritin)
What are the different ways in which iron can be clinically measured
Ferritin
Plasma iron concentration (reflects recent dietary intake)
Transferrin concentration (capacity of protein to transport iron, higher in iron deficiency)
Transferrin saturation (%)
What are the common causes of iron deficiency anaemia
Physiological demand, pathological (inadequate dietary intake, blood loss); malabsorption (coeliac disease)
What is the underlying cause of vitamin D deficiency
Lack of sun exposure
If a 60 year old woman (post-menopausal) has good iron diet intake but has iron deficiency anaemia, what may likely be the case?
She may have bowel cancer - causing gastrointestinal blood loss
What is the clinical manifestation of vitamin D deficiency
Rickets in children/bone deficiency; bone pain/fatigue/impaired wound healing/muscle weakness in adults
What clinical investigations are useful for diagnosis of vitamin D deficiency
25(OH)D test, Blood test, serum calcium
Mild vitamin deficiency = 40nmol/L