Functional Groups and pKa Values
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Alkane
A saturated (only single bonds and maximum number of hydrogens) hydrocarbon with only single bonds between carbon atoms.
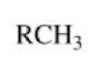
Alkene
An unsaturated hydrocarbon with at least one double bond between carbon atoms
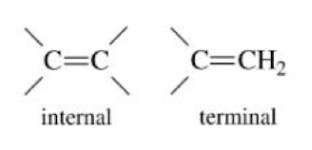
Alkyne
An unsaturated hydrocarbon containing at least one triple bond between carbon atoms.

Nitrile
A compound containing a cyano group (–C≡N) attached to a carbon atom (carbon triple bonded to a nitrogen)
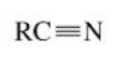
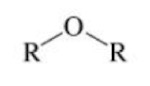
Ether
A class of organic compounds characterized by an oxygen atom single-bonded to two alkyl groups.
Arene
At least one aromatic ring (meaning it has delocalized electrons that makes it very stable). The electrons are shared evenly, but we draw this with alternating carbon double bonds
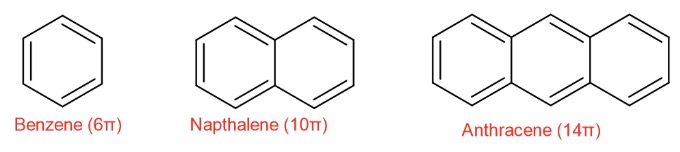
Carboxylic Acid
A compound with a carbonyl group (carbon double bonded to oxygen, C=O)
And a hydroxyl group (-OH) bonded to the same carbon
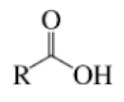
Ester
Compound with a carbonyl group
And an oxygen atom that is bonded to the same carbon and an alkyl group
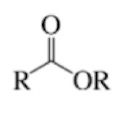
Amide
Carbonyl group
And the same carbon bonded to a nitrogen atom (the rest attached to nitrogen does not matter)
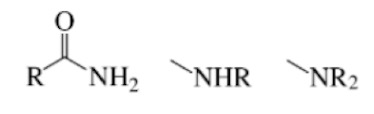
Ketone
Carbonyl group
The same carbon single bonded to two more carbon atoms
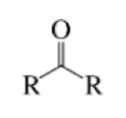
Alcohol
A hydroxyl functional group (-OH) bonded to a saturated (only single bonds and maximum hydrogens) carbon atom
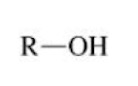
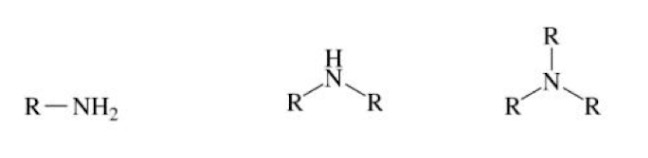
Amine
Ammonia NH3, but one of the hydrogen atoms is replaced by an alkyl group
Alkyl halide
A halogen (F, Cl, Br, I) single bonded to an sp3 hybridized carbon atom of an alkyl group
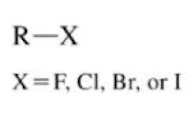
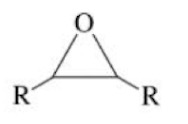
Epoxide
A 3 member ring containing one oxygen and two carbon atoms
Thiol
An alkyl group bonded to a sulfhydryl group (-SH) (similar to an alcohol group but with sulfur)
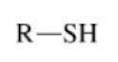
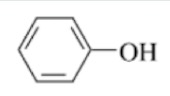
Phenol
A hydroxyl group directly bonded to an aromatic ring (evenly distributed electrons, alternating double bonds), not necessarily just one ring
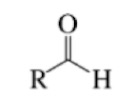
Aldehyde
Carbonyl group
The same carbon bonded to at least one hydrogen atom
HCl pKa
-7
Hydronium pKa
0
Carboxylic acid pKa
5
Ammonium (NH4+) pKa
9
Phenol pKa
10
Thiol pKa
11
Protonated amine and its pKa
An amine that has gained a proton (H+) on the nitrogen atom, number of R groups depends on the number in original amine (same number)
pKa is 11
Water pKa
14
Alcohol pKa
17
Alkyne pKa
26
Ammonia (NH3) pKa
36
Methane pKa
60
things to add
strong acids, types of aprotic and protic solvents,