ch 7 reaction mechanism and products
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
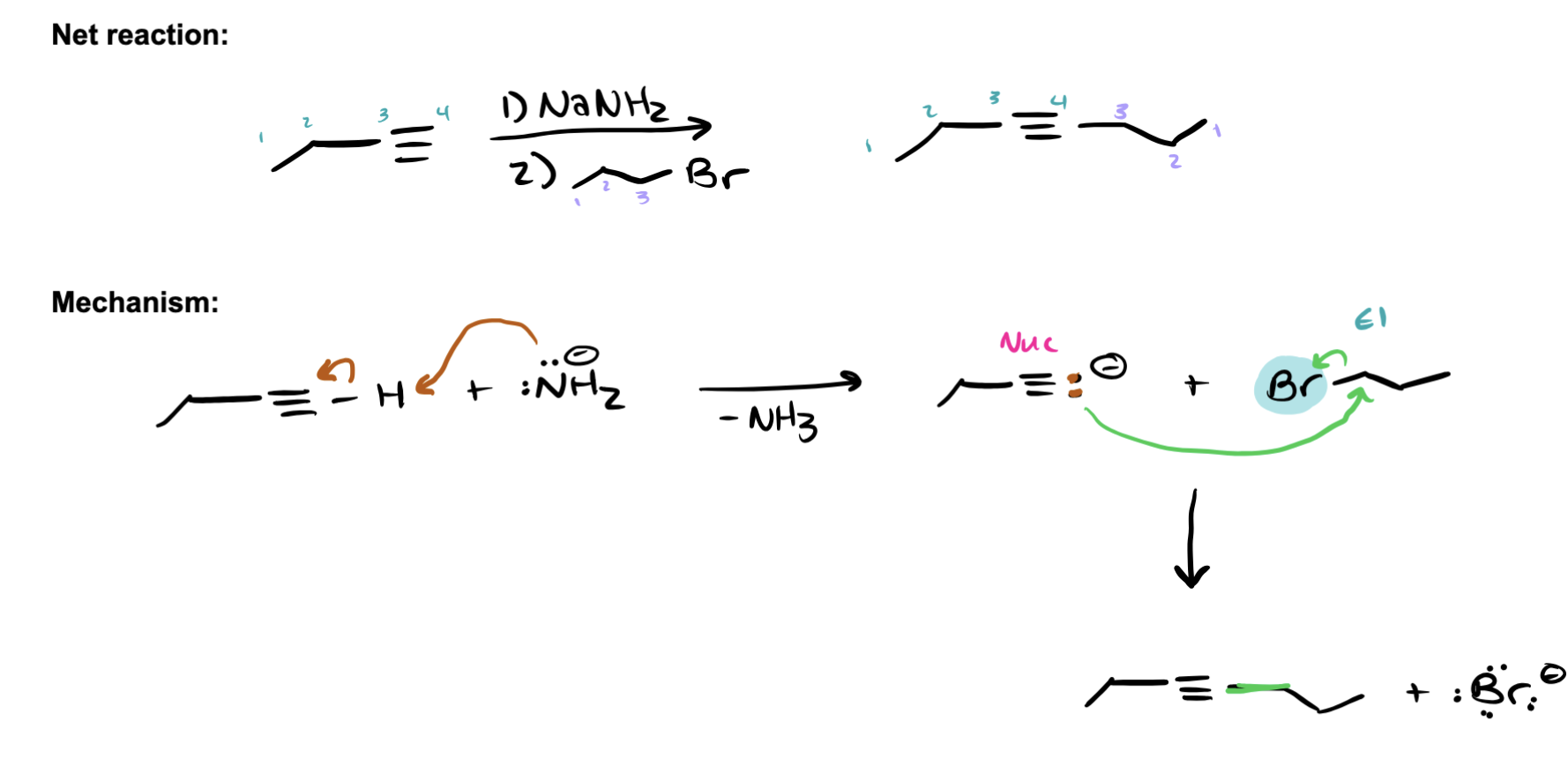
alkylation - name starting material, products, steps, ending result
starts: terminal alkyne products: alkyl halide (Br2) and strong base (NaNH2) STEP 1 (Carbocation): NaNH2 deproponates terminal alkyne forming carbocation STEP 2 (Nucelophile attack to form internal alkyne): Br2 (EL) is attacked by carbocation (Nucl) to form new bond and results in internal alkyne.
dehydrohalogenation to form internal alkyne -
STEPS: 1. Add Br2, NaNH2 attacks H, arrow moves to make double bond, kick off Br2 - repeat to make internal alyne
starts with alkane, add BR2 (ANTI or one pointing out one in), NaNH2 attacks H, electrons/arrow move to double bond, kicks BR2 off - do it again to get internal alkyne // 💥 ONE-LINE MEMORIZATION EXAM VERSION
Br₂ adds → base removes H + Br → alkene → base removes again → alkyne
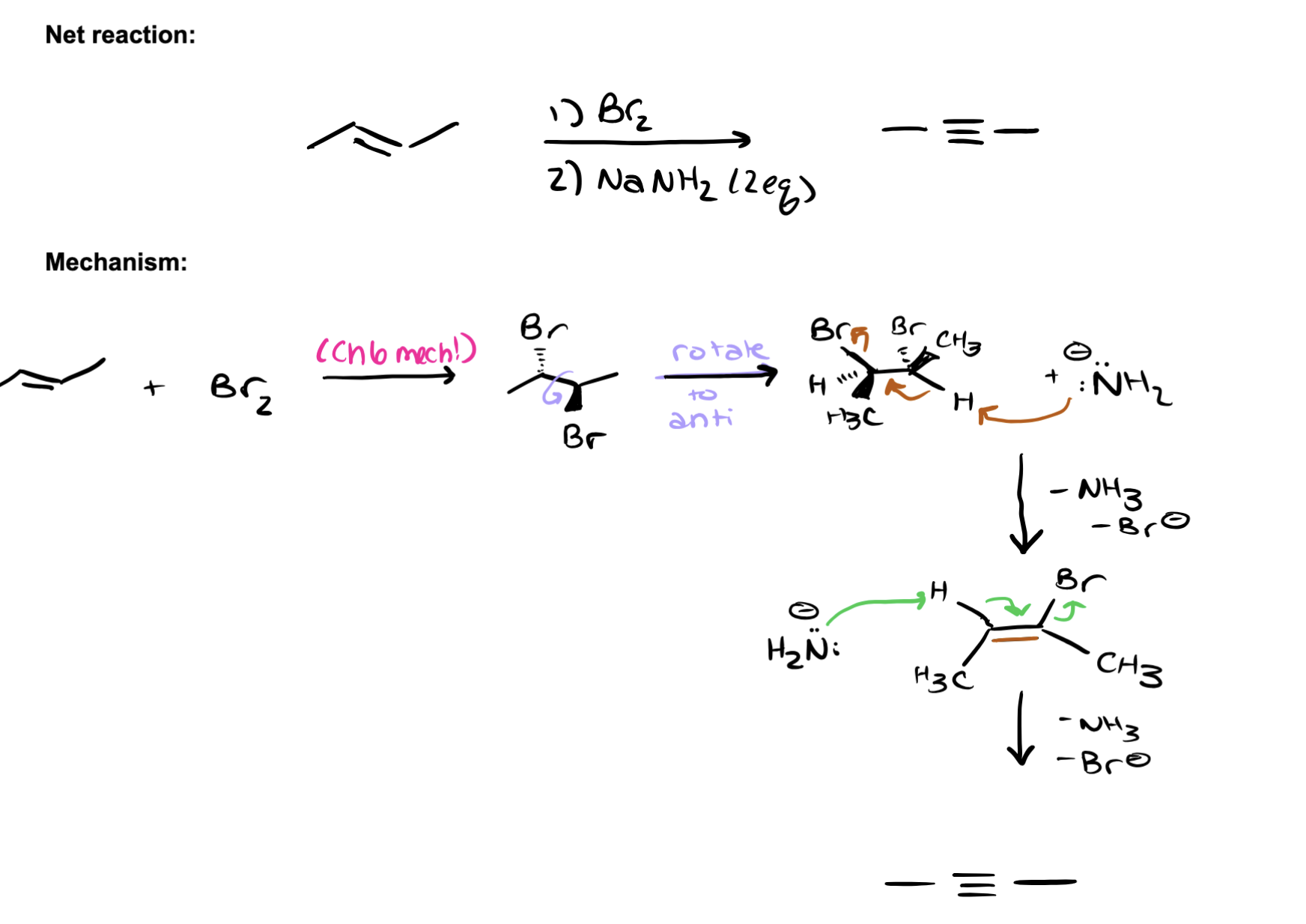
dehydrohalogenation
starts with internal alkene, add Br2 (anti), NaNH2 attacks H, forms double bond, kick off BR2 NaNH2 attacks another H, forms triple bond, kick off BR2, 3rd NH2 attacks H on newly formed internal alkyne to form carbocation, Since the newly formed alkyne is terminal, the strong base will deprotonate it (L). Therefore, a strong acid (hydronium) has to be added to reprotonate the alkyne in the final step.
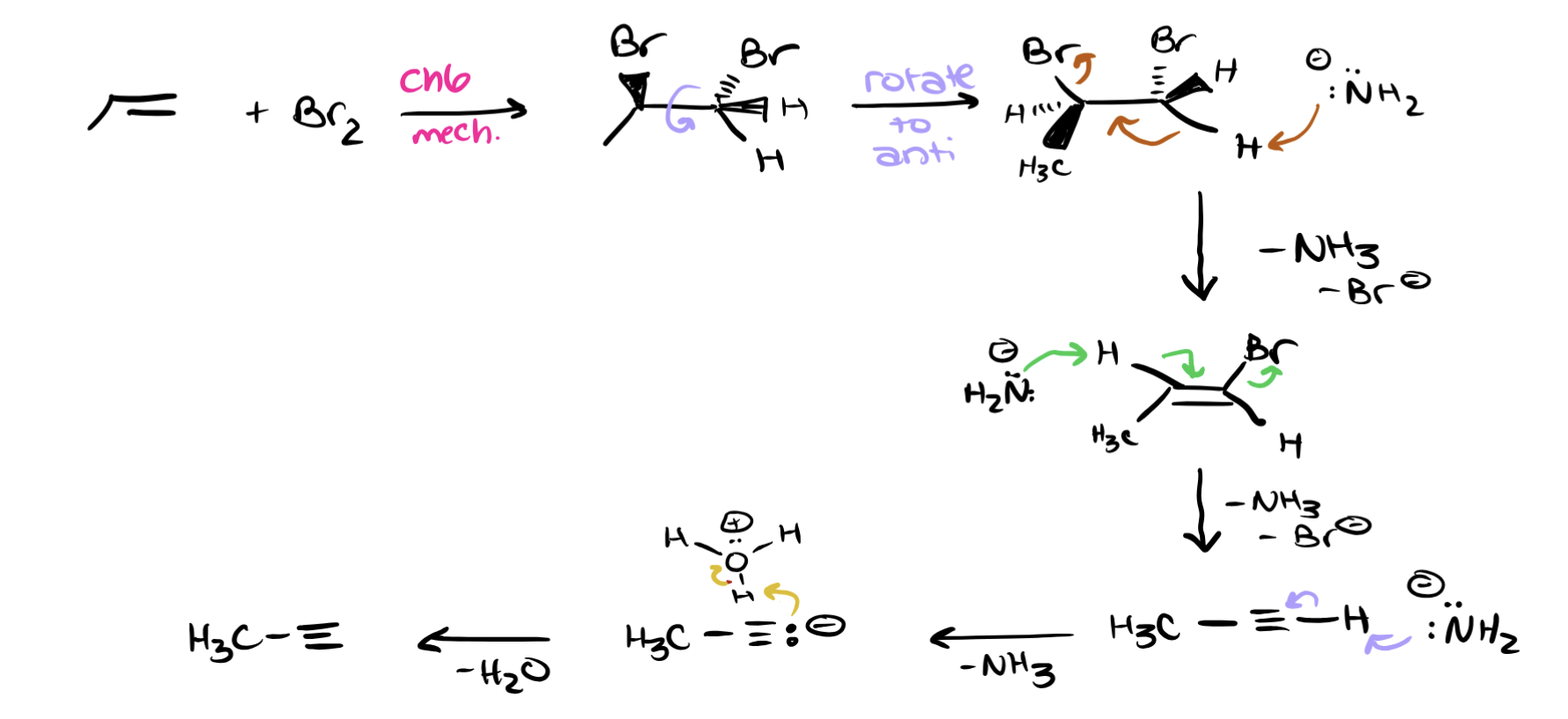
PRODUCTS: BR2, NaNH2 (3 eq), H3O, STEPS:
→ + Br₂ → 2 Br’s added
→ + NaNH₂ (twice) → triple bond
→ + NaNH₂ (again) → negative charge carbocation w/ CH3 attached to it
→ + H₃O⁺ → final neutral terminal alkyne is the product
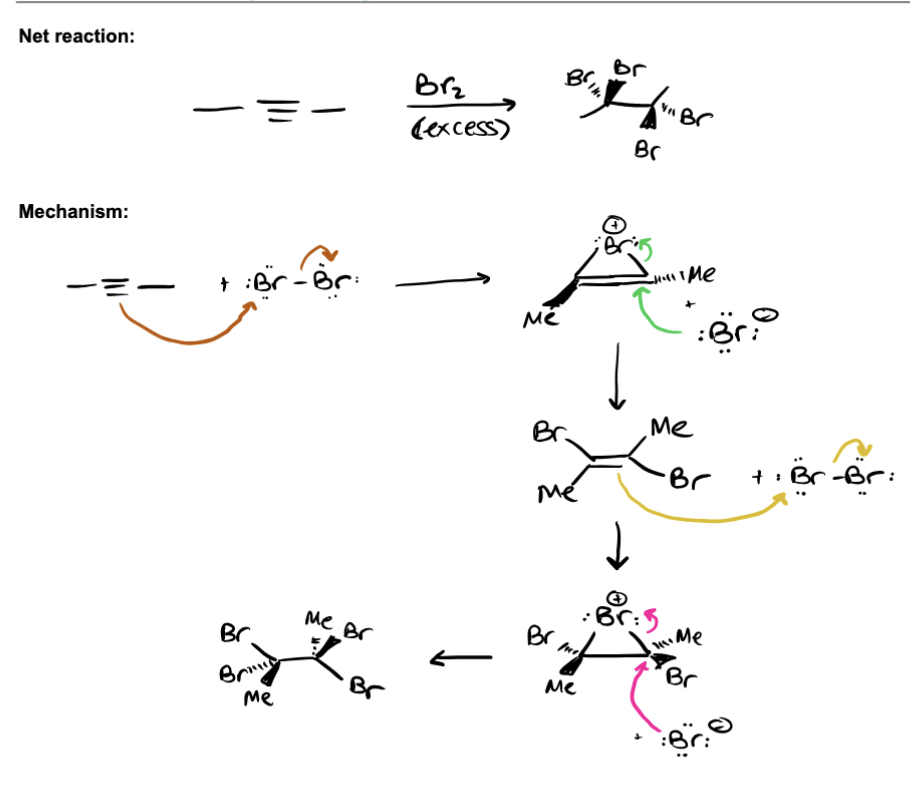
Alkyne Halogenation
MEMORIZE: alkyne attacks Br2, makes ALKENE bridge and Br anion, Br anion added to bridge anti fashion repeat until its an ALKANE with 4 Br ( tetra substituted alkane) STEPS: terminal alkyne (nucl) attacks the Br2, forms a bromonium bridge and Br anion, the Br anion attacks bridge forming a dibrominated alkene in ANTI fashion, the dibrominated alkene attacks another Br2 forming a bridge and Br anion, kicking off a Br, forming a bridge and Br anion, Br anion anion attacks bridge forming tetra substitued alkane
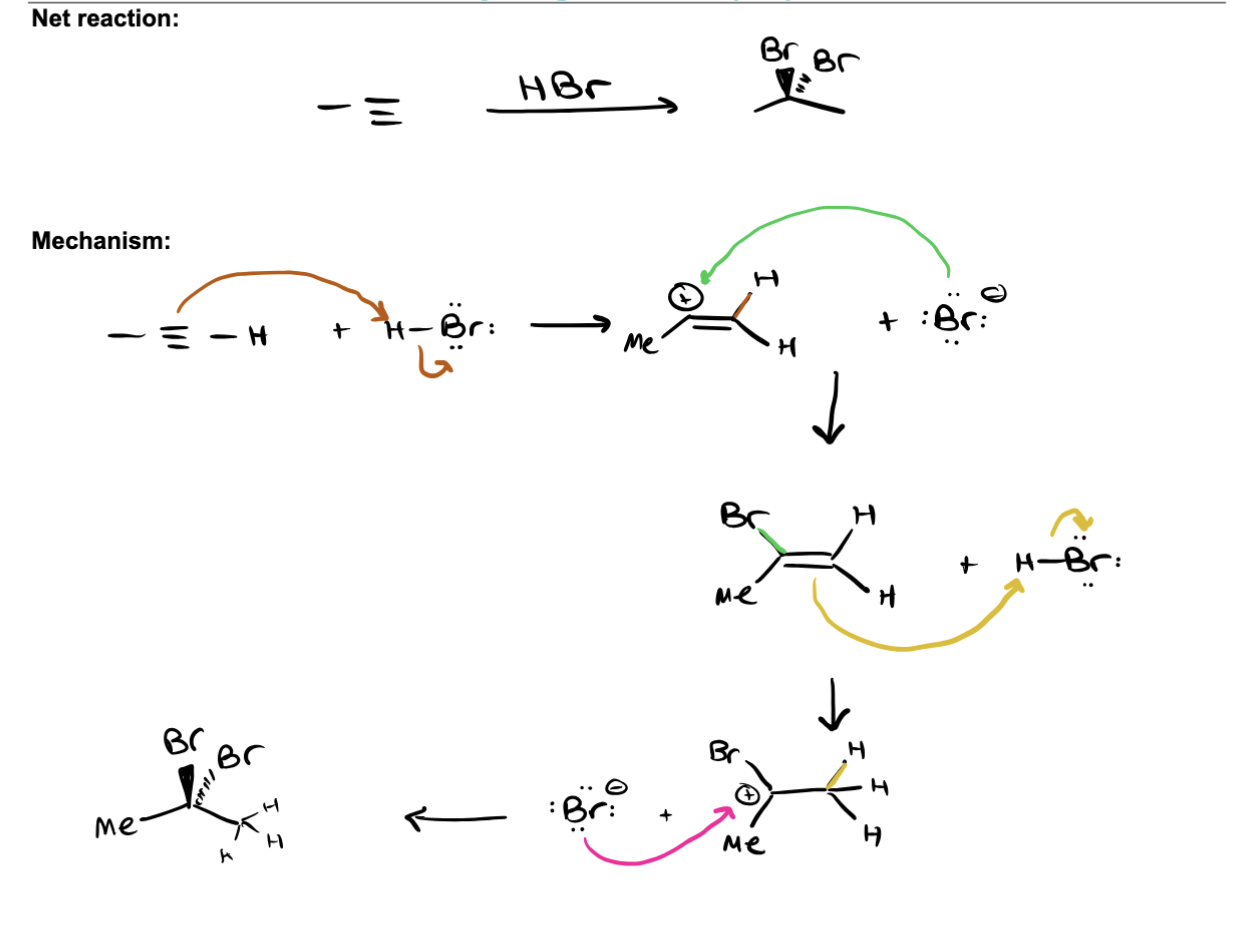
Addition of Hydrogen Halide to form Geminal Dihalide
PRODUCTS: HBR STEPS: ALKYNE ATTACKS H-BR MAKES ALKENE BOND WITH H ON LESS SUBSTITUED CARBON → BR ANION ATTACKS CARBOCATION → FORMS BROMINATED ALKENE → REPEATED UNTIL GEMINAL DIHALIDE ON ALKANE EXPLANATION: 2. alkyne attacks H on H-Br, forms alkene with carbocation (markiviok) and Br anion, Br anion attacks carbocation on alkene replacing it to form brominated ALKENE, then the ALKENE attacks another H on H-BR forming Br anion and alkane with carbocation, (H on less subsitiued AND hen final Br anikon attakcs carbocation forming geminal dihalid, where both Brs are on same carbon