CHM2046 Final (Pham notes)
1/22
Earn XP
Description and Tags
Currently, I am on the exam 1 material for these flashcards and will add exam 2,3, and 4 after my chicken sandwich
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
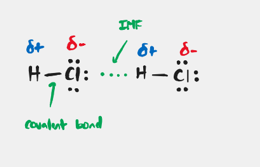
Intermolecular Forces (IMF)
The forces of attraction that occur between atoms and molecules.
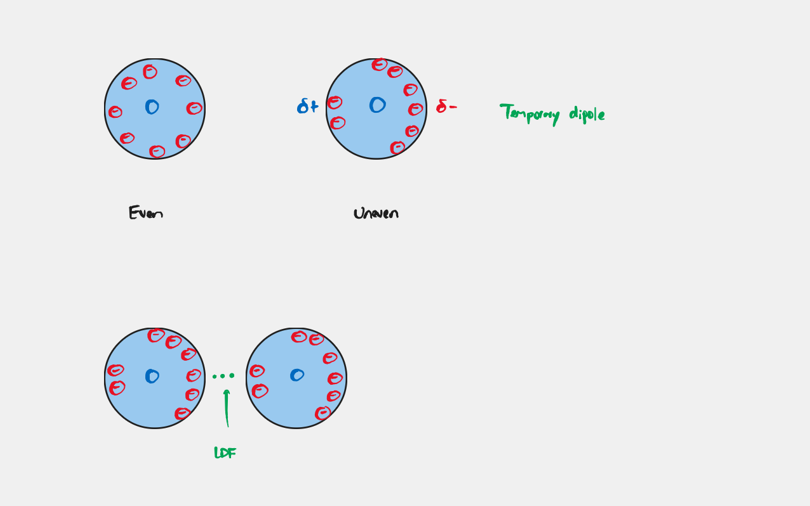
London Dispersion Force
Forces that are the result of fluctuations in the electron distribution within atoms and molecules. It is present in ALL atoms and molecules.
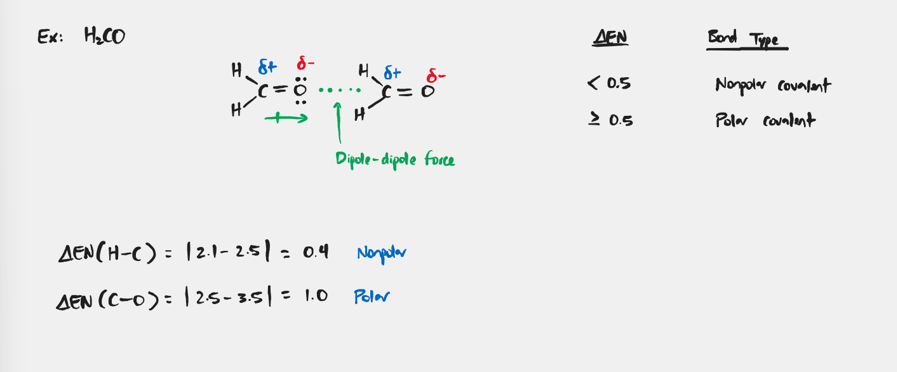
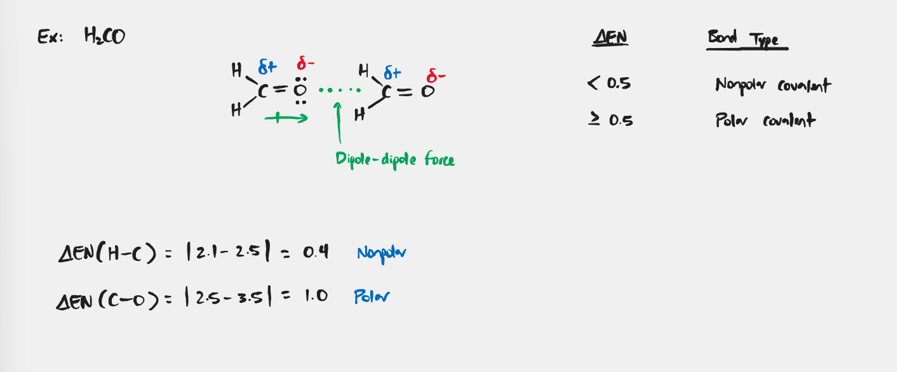
Dipole-Dipole Force
Forces that exist in all polar molecules as a result of uneven charge distribution.
Hydrogen Bonding Force
Forces that are present in all polar molecules that have an H atom bonded to small, highly EN atom (N,O, or F)
Ion-dipole Forces
Occurs when an ionic compound is mixed with a polar compound. It represents the force between an ion and the oppositely charged end of a polar molecule.
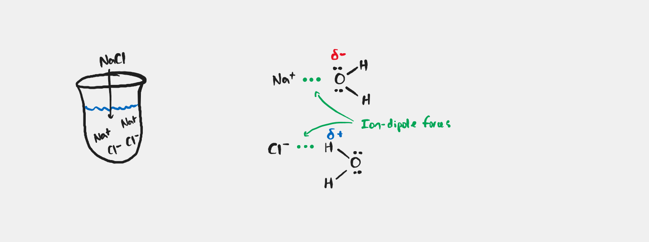
What happens to a strong IMF?
The stronger the IMF, the more energy that is required to break the interactions and the higher the bp.
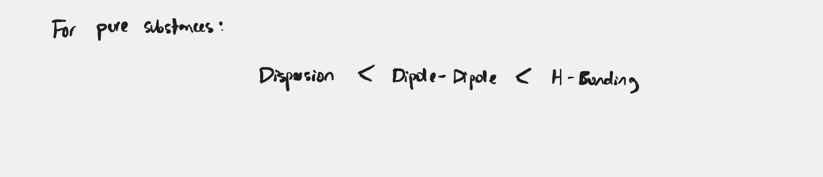
What do you consider when considering atoms that have the same type of predominant IMF?
The bp generally increases with increasing molar mass.

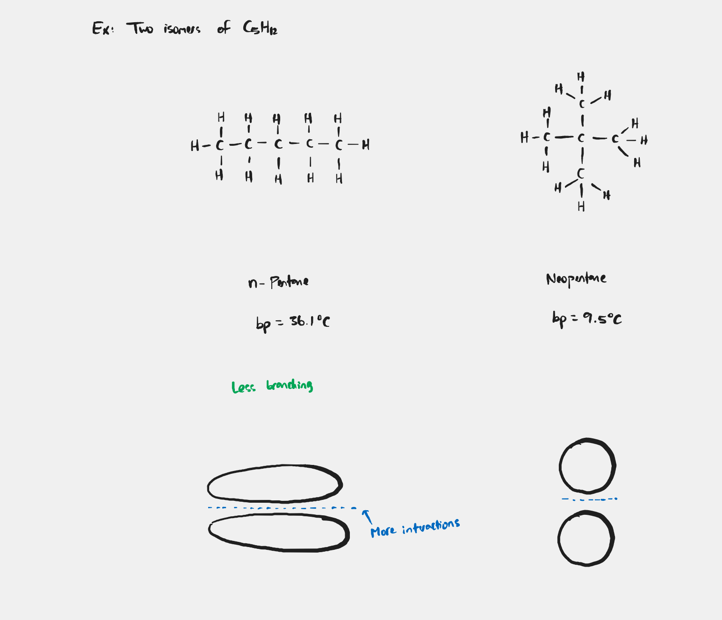
What happens when comparing molecules that have the same type of predominant IMF and the same molar mass?
Molecules that have a longer overall shape will have the higher bp.
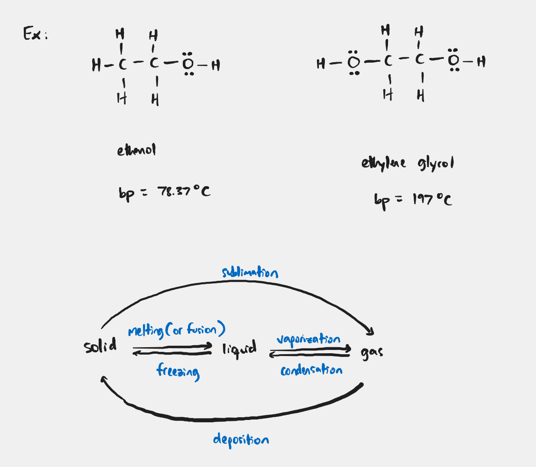
When considering molecules that H-bonding forces as their predominant IMF how does that effect bp?
The bp increases with increasing number of H-X (X = N, O, F) bonds.
Endothermic
Delta H is greater than 0
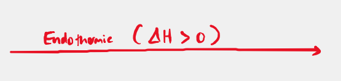
Exothermic
Delta H is less than 0
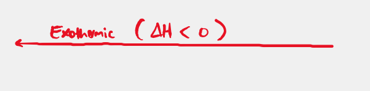
Surface Tension
The energy required to increase the surface area of a liquid by a unit amount. It is the tendency of liquids to minimize their surface area.
Viscosity
A measure of the resistance of liquid to flow.
Capillary Action
The ability of a liquid to flow against gravity up a narrow tube. Results from the combination of two forces (cohesive and adhesive)
Cohesive Forces
The attraction between individual molecules
Adhesive Forces
The attraction between the molecules and the surface.
How does surface tension, viscosity, and capillary action act with IMF?
Increases with increasing IMF.
Rate of Vaporization
Increases with increasing temperature
Increases with increasing surface area
Increases with decreasing IMF
Enthalpy of vaporization (delta H vap)
The amount of heat required to vaporize 1 mol of a liquid to a gas.