Main Group Systems
1/104
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
105 Terms
How do you calculate the metal oxidation state?
O.S. = Charge on complex - sum of the charges of the ligands.
How do you calculate the metal d-electron count?
d electrons = group number - O.S.
How do you calculate total valence electron count?
Valence electrons = d-electron count + electrons donated by ligands + number of M-M bonds
What are the principles of molecular orbital theory?
Orbitals must be of appropriate symmetry.
Orbitals must overlap.
Orbitals should be of similar energy.
Interaction energy is stronger for orbitals with good overlap and close in energy.
What does the molecular orbital diagram look like for a general transition metal complex with n-ligands?
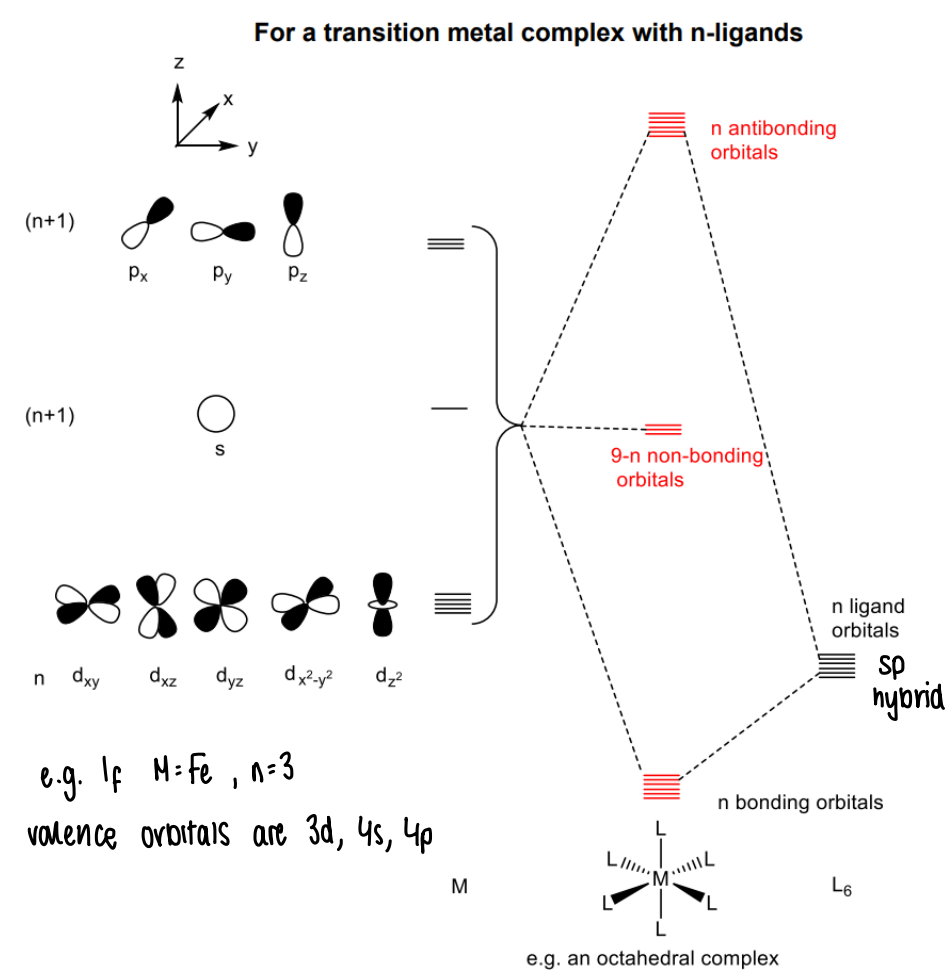
How does bonding to a ligand affect the orbitals?
It disrupts the degeneracy of the orbitals, the extent of which is dependent upon interactions between orbitals.
Stronger interactions means lower energy bonding MOs.
What is needed for M-L multiple bonds to form?
They contain a sigma-bond and one or two pi-bonds.
For pi donation to occur there must be an empty metal d orbital to accept the electrons.
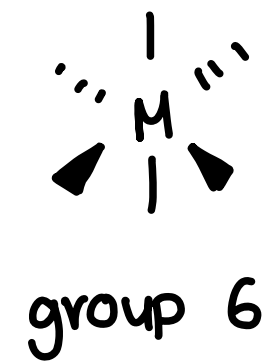
What is the coordination number of group 6 metals and how does it bond to other metals?
Forms a simple octahedral complex, does not bond to other metals.
Forms M(CO)6
What is the coordination number of group 7 metals and how does it bond to other metals?
Can bond to one other metal.
As only sigma bonds are involved, the staggered conformation gives the least steric clash.
Forms M2(CO)10
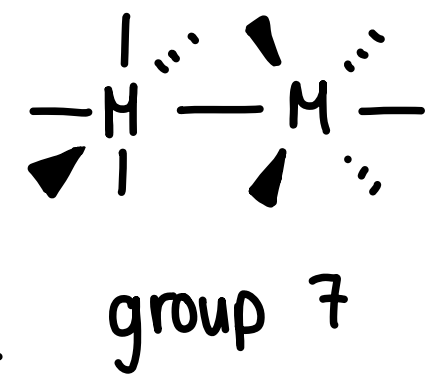
What is the coordination number of group 8 metals and how does it bond to other metals?
Bonds to 3 other metals.
Forms M3(CO)12
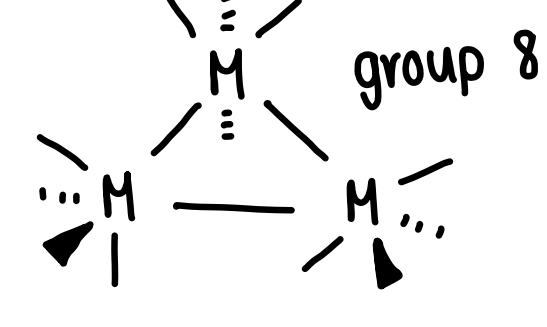
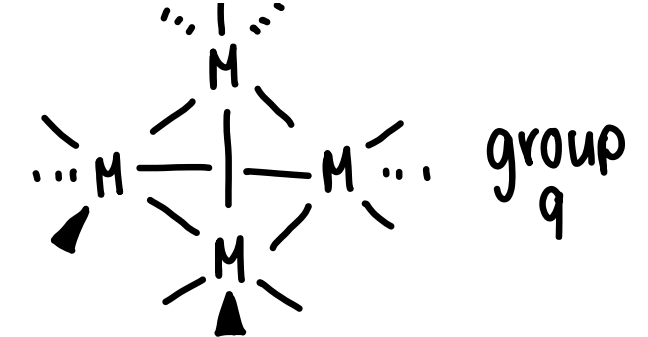
What is the coordination number of group 9 metals and how does it bond to other metals?
Bonds to 4 other metals.
Forms M4(CO)12
What is the coordination number of group 10 metals and how does it bond to other metals?
The metal is too small to fit 6 groups around, therefore forms a tetrahedral complex.
Forms M(CO)4

Why do metals form 18 electron complexes?
This maximises orbital overlap and minimises steric clash.
How do metals form carbonyl complexes?
React the metal with excess CO at 200 atm.
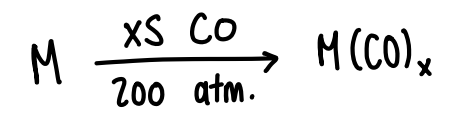
How do d-orbitals interact with one another?
The least overlap gives the weakest bonding interaction, therefore they have higher energy.
Orbitals which can overlap the strongest give the strongest bonding interaction (such as dz2)
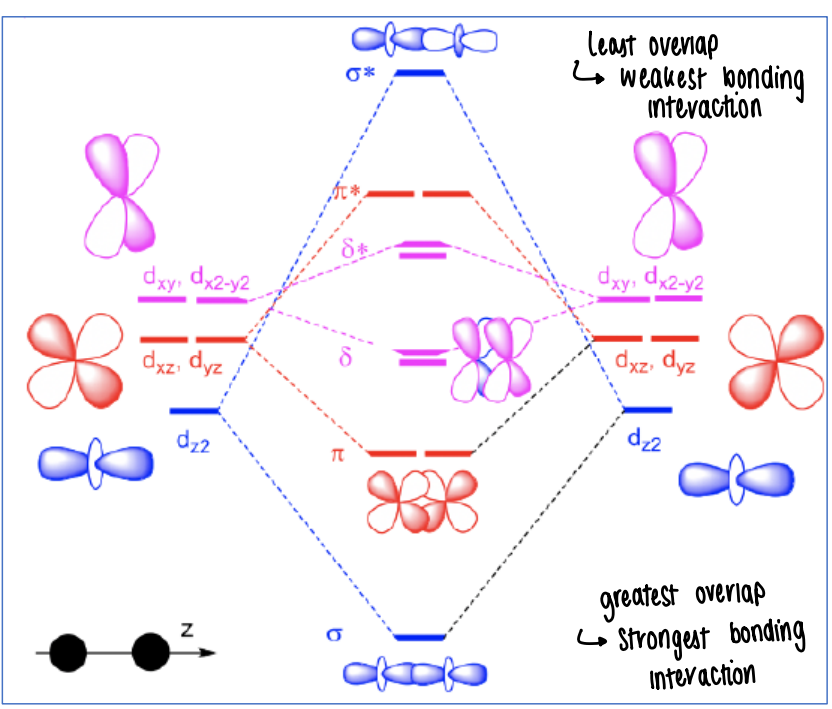
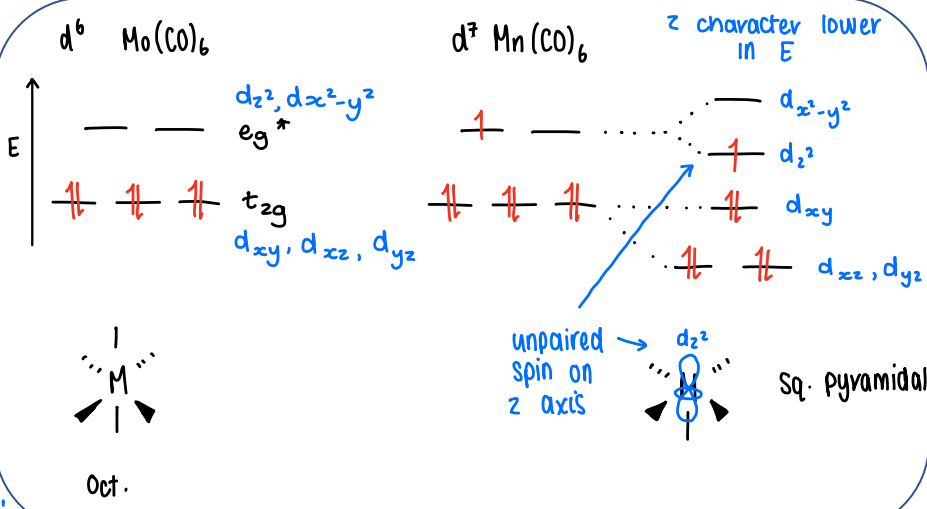
How does loss of a ligand affect the energy of the d-orbitals?
As the dz2 orbitals point straight towards the ligands, when a ligand is lost in the z direction this causes the dz2 orbital to become less antibonding, therefore it falls in energy.
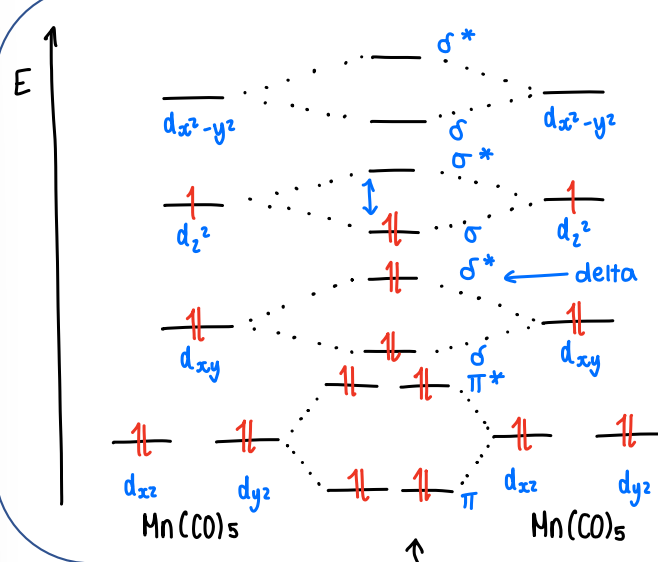
What is the molecular orbital diagram for the bonding of two metal complexes (e.g. Mn(CO)5)?
Each complex has one unpaired electron to donate to the other, forming a MO.
How is bond order calculated?
BO= Bonding pairs - antibonding pairs.
What is the trend in BDE of metal complexes as you go down the group?
The BDE gets stronger as you go down. This is due to the greater energy to promote an electron from the sigma bonding orbital to the sigma antibonding orbital.
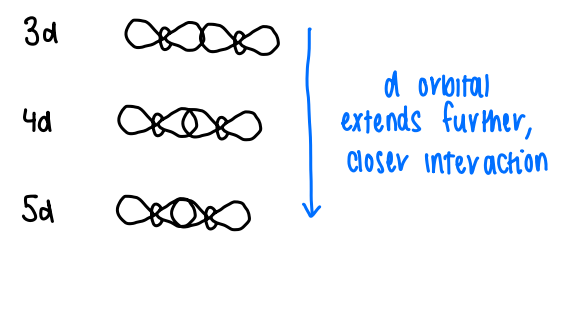
What is the trend in bond strength to transition metals down a group?
As you go down the group, the bonds tend to get stronger. This is due to the d orbitals extending further as you go from 3d to 5d, allowing a closer interaction to ligands.
This increases dz2 overlap, therefore the sigma bonds are more stable.
What bonding orbitals are formed when metals quadruple bond to one another?
The formation of quadruple bonds decreases the bond distance from around 3 A (single bond) to 2 A.
The sigma, pi and delta bonding orbitals are all occupied.
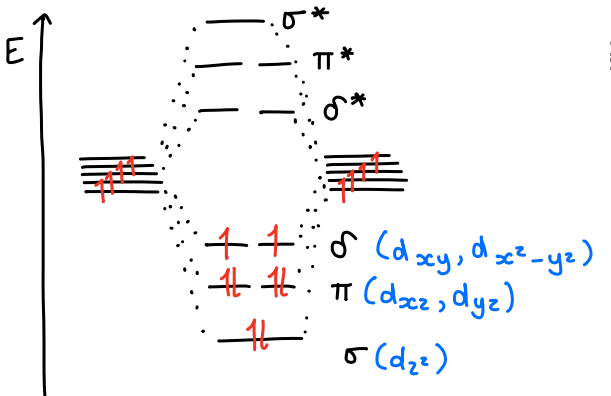
How do delta bonds compare to pi bonds?
They are generally weaker due to poorer overlap between precursor orbitals.
What is the trend in bond order of M2 dimers?
The bond order increases, and effective bond order (calculated by quantum mechanics) increases.
The dimers are in the 0 O.S.

What does the molecular orbital diagram of a M2 transition metal (d6) dimer look like?
The p orbitals are too high in energy so do not interact.
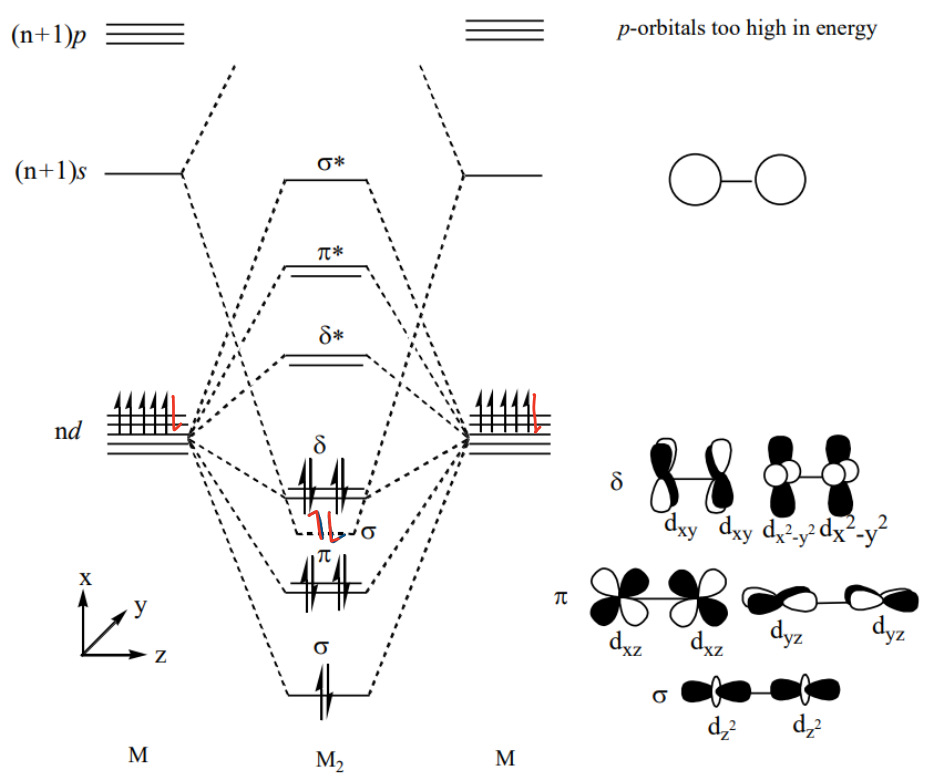
What bond order is expected in metal complexes?
A bond order less than 5 as the metal d orbitals are required for the M-L bonding to occur.
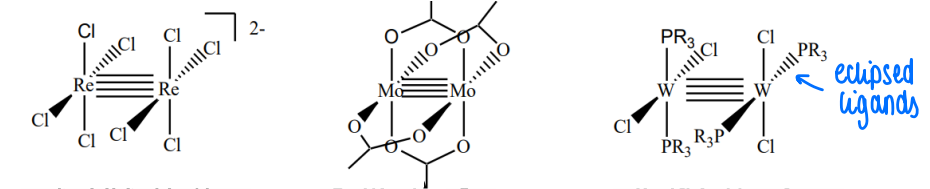
What are M2X8 complexes and what causes the arrangement of ligands?
d4 metal form quadruple bonds to another metal. The ligands form a square planar geometry around the metal, forming an eclipsed structure.
Eclipsed gives the orientation that allows best overlap of the d orbitals (although it causes unfavourable sterics).
What is the molecular orbital diagram for a square planar complex?
The dz2 orbitals form the sigma bonds, the dxz and dyz form pi orbitals, the dxy orbitals form delta bonds (they have 2 nodes, so have 4 overlapping orbitals).
The dx2-y2 orbitals are primarily involved in bonding to ligands.
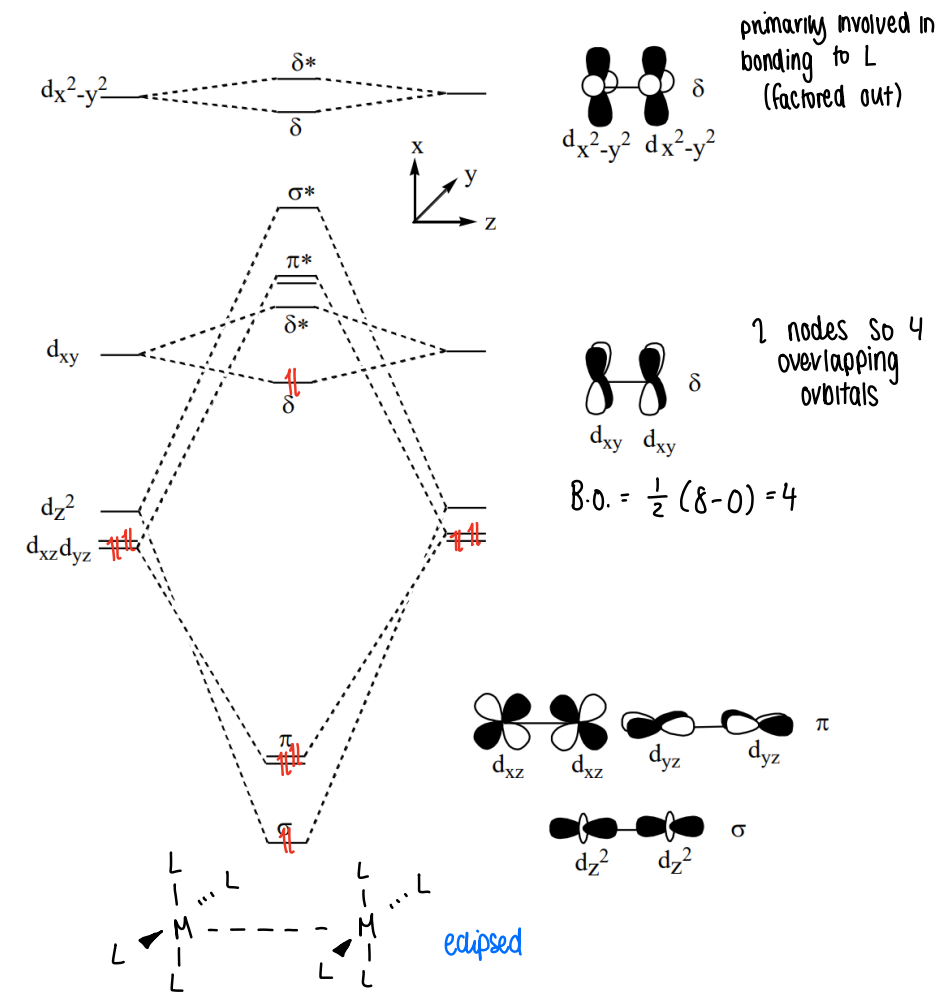
Why is an eclipsed conformation most common?
In most complexes, the eclipsed conformation is needed for overlap of the delta orbitals.
However, in some complexes such as Os2Cl82-, there are two extra electrons which go into the delta* orbital, giving a bond order of 0 therefore the complex can be either eclipsed or staggered.
When are M2X6 and M2X9 structures formed?
d3 metals form triple bonds to one another.
This can give bridged (for 9 ligands) or non-bridged (eclipsed or staggered) conformations.
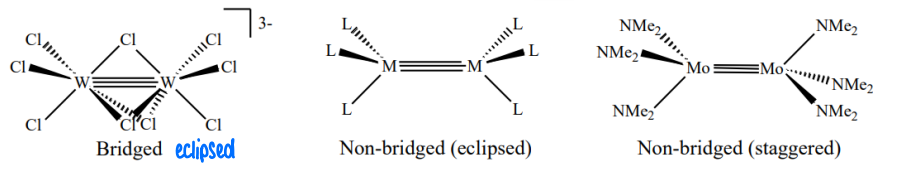
What does the molecular orbital diagram look like for a transition metal M2X6 complex?
The dxz and dyz orbitals become hybrids as they are mixed with dx2-y2 and dxy orbitals respectively. This causes tilting which causes the pi and pi* orbitals to have some delta character.
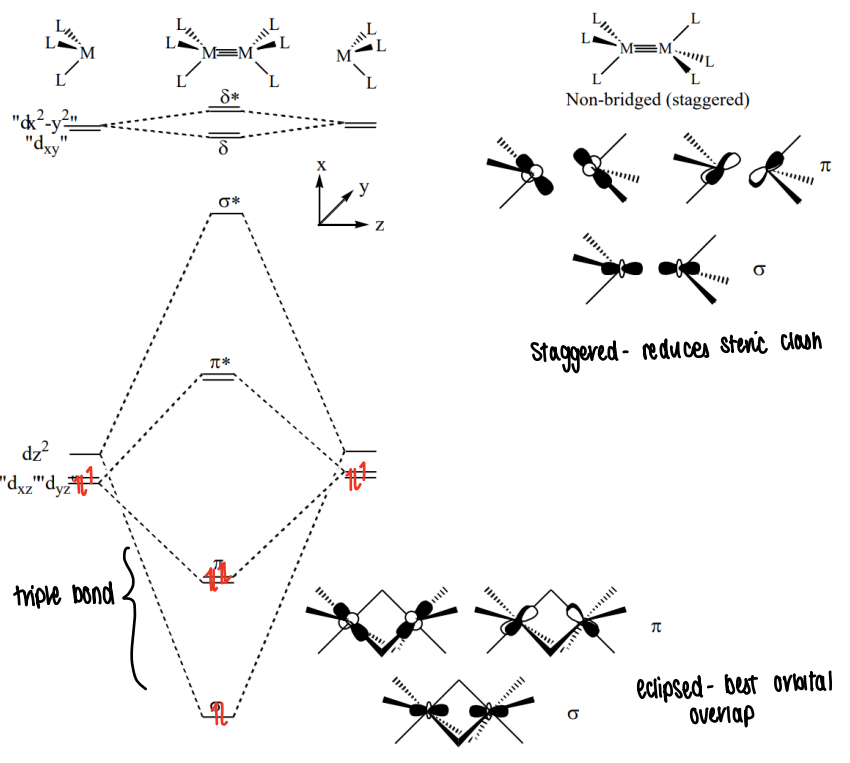
Do M2X6 complexes prefer eclipsed or staggered?
The eclipsed conformation gives the best orbital overlap with ligands, however the staggered conformation reduces the steric clash.
The staggered conformation is mostly seen.
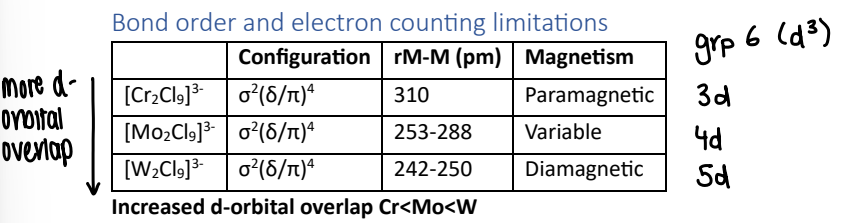
How does d-electron overlap affect magnetism?
Complexes with all electrons paired up should be diamagnetic, however the 3d orbital doesnt extend out far enough for significant orbital overlap, therefore there is little covalency and so the M-M bonds are weak and so the electrons remain unpaired.
If 5d orbitals are involved, the orbital extends further so the electrons are paired.
For 4d orbitals, the M-M distance is dependent upon the ction present in the structure. They are variable.
How do you form a quintuple bond?
A quintuple bond can be formed by reduction of [Cr(µCl)Ar’]2 with slight excess of potassium graphite, producing a very short Cr-Cr bond length.
![<p>A quintuple bond can be formed by reduction of [Cr(µCl)Ar’]2 with slight excess of potassium graphite, producing a very short Cr-Cr bond length.</p>](https://assets.knowt.com/user-attachments/2abbc9a0-63f9-4e24-b486-9d0d2fa48634.png)
What does the molecular orbital diagram look like for quintuple bonded complexes?
There are 9 valence orbitals.
The ligand forms bonds with 4s orbitals, however the 4p are too high in energy. This leaves the 3d orbitals available for bonding to the metal.
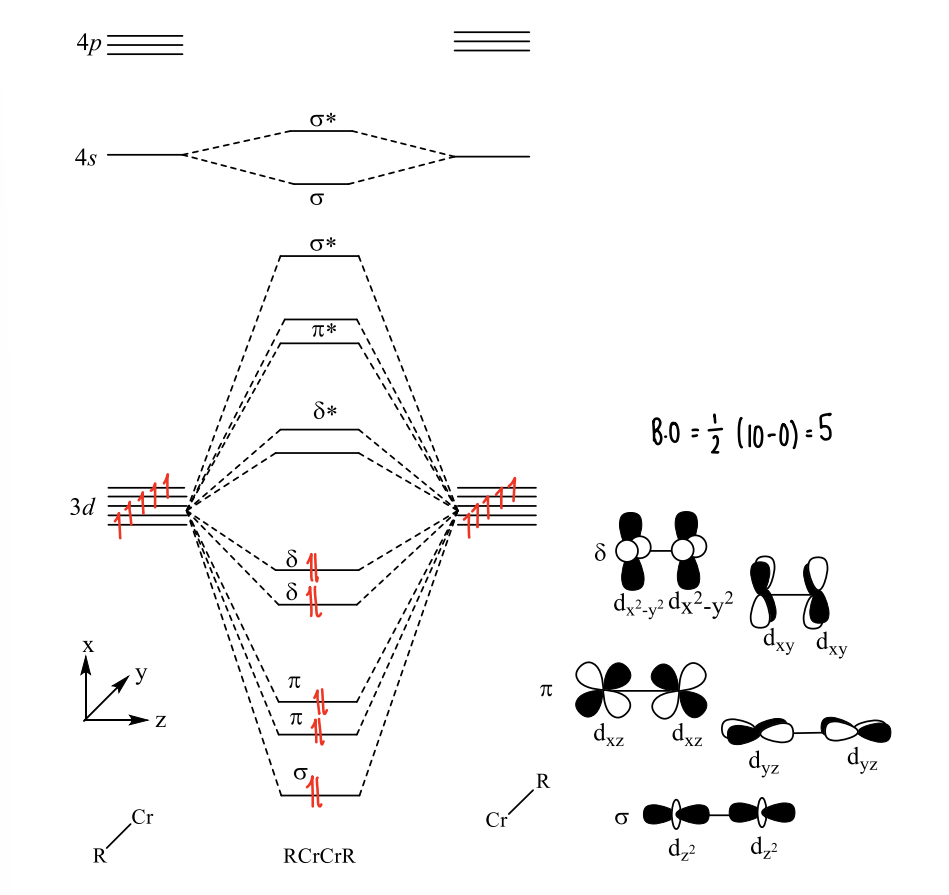
What is the geometry of a quintuple bond?
A linear geometry is expected, however a trans bent geometry is observed, suggesting orbital mixing occurs.
What is the general mechanism for catalytic cyclotrimerisation?
Occurs via a [2+2+2] cycloaddition, proceeding via a metallacyclopentadiene intermediate.
![<p>Occurs via a [2+2+2] cycloaddition, proceeding via a metallacyclopentadiene intermediate.</p>](https://assets.knowt.com/user-attachments/6ea80322-b788-4f4e-84cf-053b61510bdb.png)
How do you isolate intermediates?
Via stoichiometric reactions, forming the intermediate only.
What is salt metathesis?
A metal salt is reacted with desired ligands, a new Mo complex is formed as the two ligands attach, removing KCl.
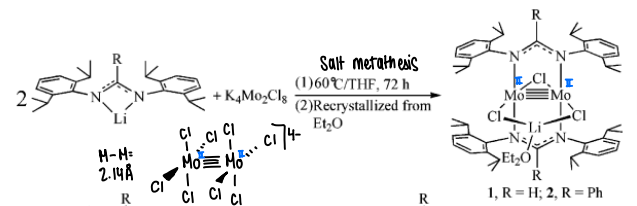
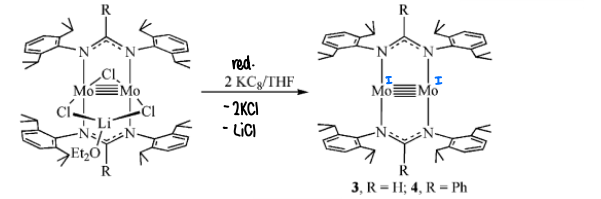
How is a quadruple bond reduced into a quintuple bond?
Reducing using 2 equivalents of KC8 in THF.
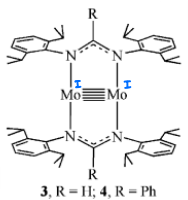
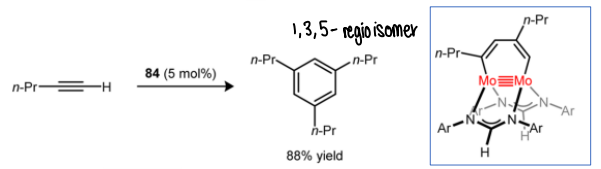
How does this Mo complex catalyse the cyclotrimerisation of an alkyne? And what intermediate forms?
Selectively forms one regioisomer.
What oxidation state do the group 1 elements majorly form?
+1
What oxidation state do the group 2 elements majorly form?
+2
What is the inert pair effect?
In heavier elements, the s electrons don’t want to bond so stay ‘inert’, this is due to the poorer shielding from the d and f electrons. The s electrons are therefore held tightly by the nucleus, making lower O.S. states more stable.
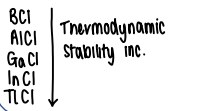
How does the inert pair effect influence group 13 XCl compounds?
As you go down, the inert pair gets stronger therefore the elements are more stable in the +1 state, increasing thermodynamic stability of the compounds.
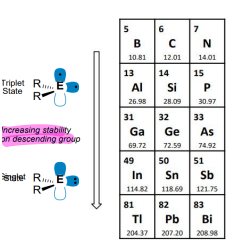
What is the trend in electronic ground state (singlet or triplet) as you descend a group?
The preferred state changes from triplet to singlet, the singlet is more stable due to the lower O.S..
The electrons in the singlet state have a greater ionisation energy.
What is the double bond rule?
It was believed that multiple bonds involving an elements with a principal quantum number greater than 2 could not exist.
Why are sigma-bonded main-group polymers stronger than double bonded polymers?
Pi bonding in heavier main group elements is weaker than sigma bonding.
The sigma bonded polymers are therefore more stable than double bonded, which involves a sigma and pi bond.
It can be seen that two sigma bonds are thermodynamically favoured over a sigma and pi bond.
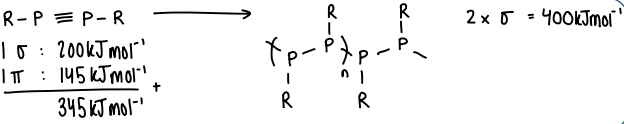
How are main group double bonded complexes stabilised?
Large ligands can be used to provide kinetic stabilisation.
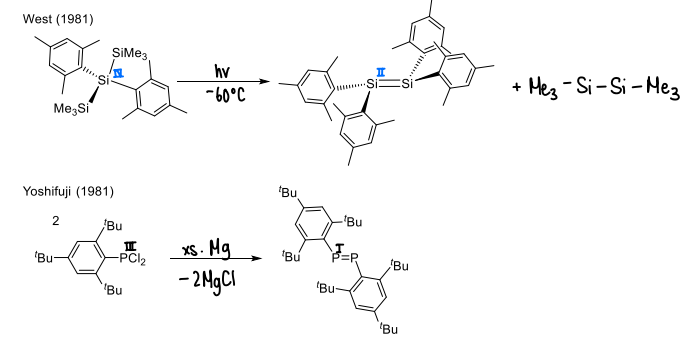
What is the geometry of a main-group double bond?
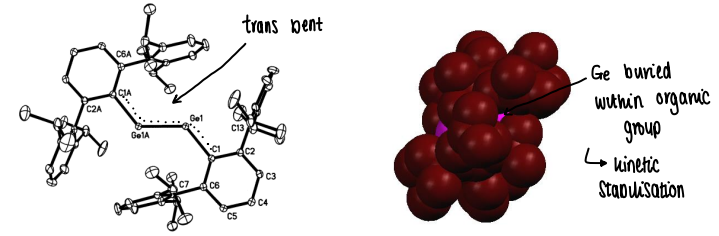
Apart from carbon, all main group double bond compounds have a trans-bent structure.
Distortion in planarity increases as the group descends.
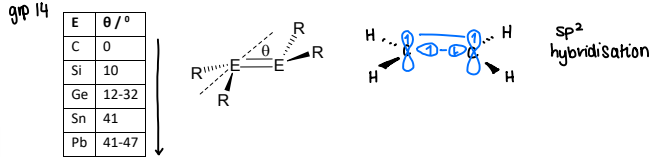
Why does a trans-bent structure form in main group compounds?
Due to the interelectronic repulsion between the non-bonding lone pairs.
There is increased non-bonding electron density on the heavier main group elements.
Why does the energy gap between the p and n orbitals increase as we go down the group?
The E gap increases as the singlet state starts to dominate, therefore lowering the E of the n orbital as the s electrons become tightly held by the nucleus. Both electrons are then held in the n orbital, whereas in a triplet state (with a smaller energy gap) the electrons are unpaired in the p and n orbitals.
This therefore decreases the gap between the pi and sigma* orbitals.
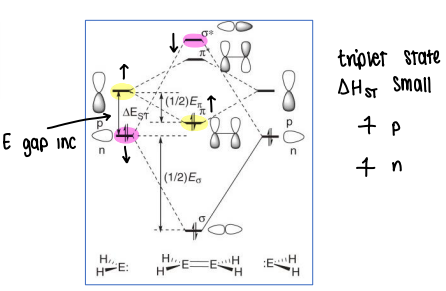
Why does deviation in planarity of main group compounds increase as the group descends?
As we go down the group, the energy gap between the pi and sigma* MOs decrease, allowing more mixing of orbitals to occur and so a greater deviation in planarity occurs.
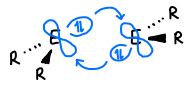
How does mixing of the orbitals occur in main group compounds?
As the pi and sigma* orbitals get closer in energy, they are able to mix and form two new orbitals (n+ and n*).
This gives the orbital more s character and the energy of the filled orbitals is lowered by mixing, stabilising the structure.
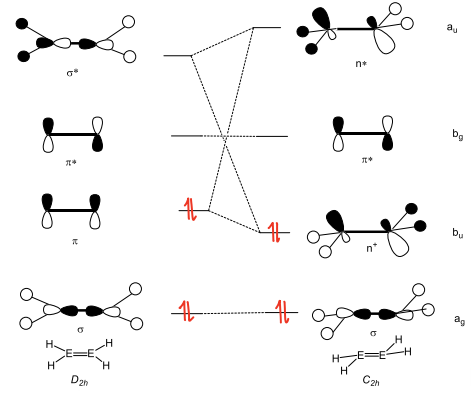
Why do carbon based compounds not have deviation of planarity?
In C=C compounds, the energy gap between the pi and sigma* orbitals is too large and so no mixing occurs.
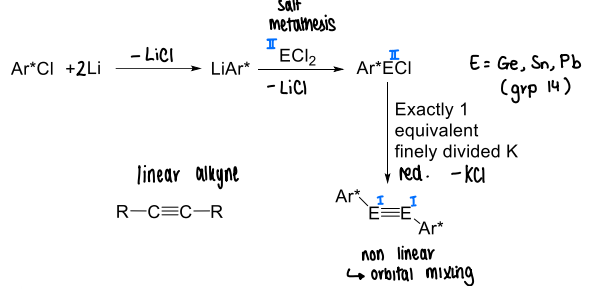
How do you form main group analogues of alkynes?
Via lithiation, salt metathesis, and reduction.
A very sterically demanding substituent is required.
Why do sterically bulky substituents stabilise main group triple bonds?
The main group elements is buried within the organic group, providing kinetic stabilisation.
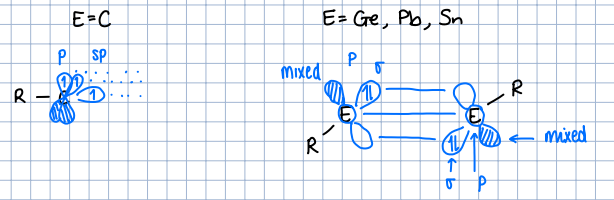
How do carbon triple bonds compare to other main group triple bonds?
Carbon has 2 sp and 1 p orbital which engage in bonding.
Other main group elements have a sigma, p and mixed orbital that engages in bonding.
What is the trend in E-E bond length down the group?
The E-E bond distance increases, however the Si-Si bond distance is shorter than the Si=Si bond.
The Pb-Pb bond is also longer than expected.
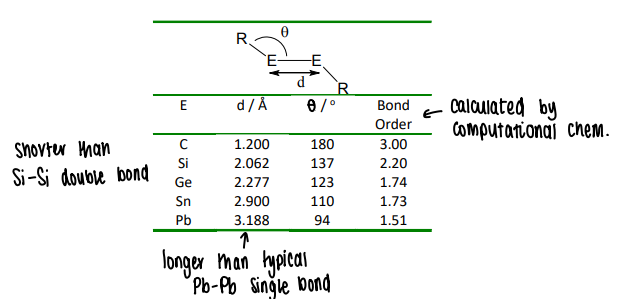
What happens to heavier E-E compounds in solution?
Heavier elements have weaker E-E bonds and have a higher stability of the singlet state, and so often dissociate into monomeric counterparts in solution.
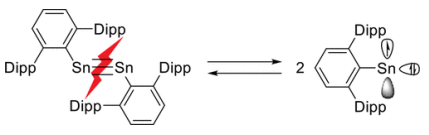
Why are main group elements able to activate small molecules?
They have energetically accessible frontier orbitals.
Why are main group compounds unable to act as redox based catalysts?
They can readily react in oxidative addition reactions, however reductive elimination is challenging due to the stability of the En+2 O.S.
Once the +2 O.S. is achieved, the complex wont want to reductively eliminate.
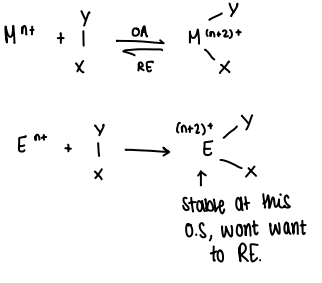
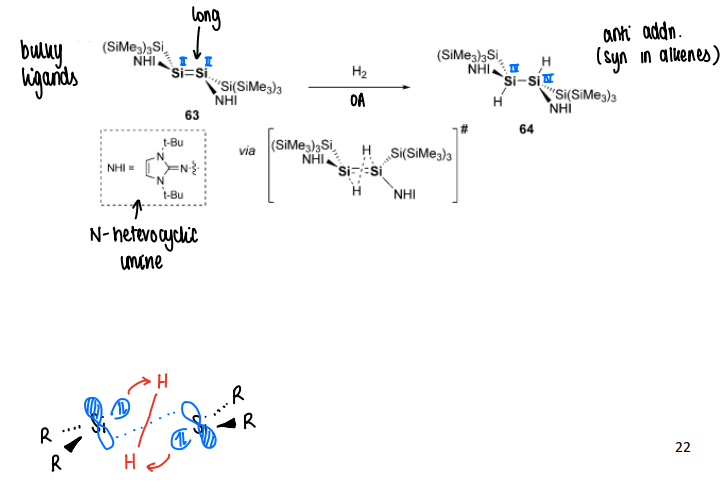
How do double bonded main group complexes activate dihydrogen?
Oxidative addition of H2 occurs onto the double bond (concerted anti addition occurs).
The double bond is highly trans-bent and twisted with a long double bond. The H2 forms a staggered arrangement which preorganises the central E-E for the anti-addition.
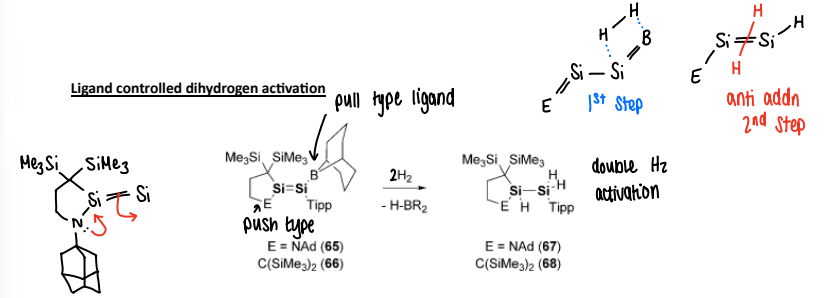
How does ligand controlled dihydrogen activation occur in main group complexes?
There is ‘push-pull’ ligands that enhances the rate of reaction. The NAd ligand acts as a push, with the boryl group acting as the pull.
The lone pair from the N is donated into the N-Si bond and the Si=Si break into a single Si-Si bond. The dihydrogen can then coordinate to the metal and the boryl group is cleaved, reforming the Si=Si bond.
The second equivalent of H2 than undergoes anti-addition to the Si=Si bond.
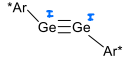
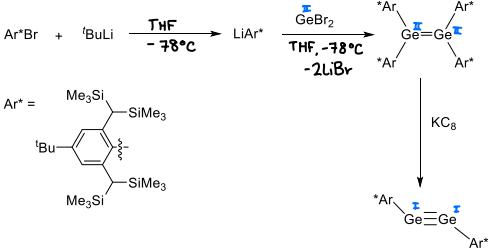
How is a digermyne complex formed?
The ligand (Ar*Br) is lithiated.
GeBr2 is reacted with LiAr* in THF at -78 degrees celsius, forming a Ge=Ge complex.
KC8 is then added forming the digermyne via reduction.
The bulky Ar* ligand is required.
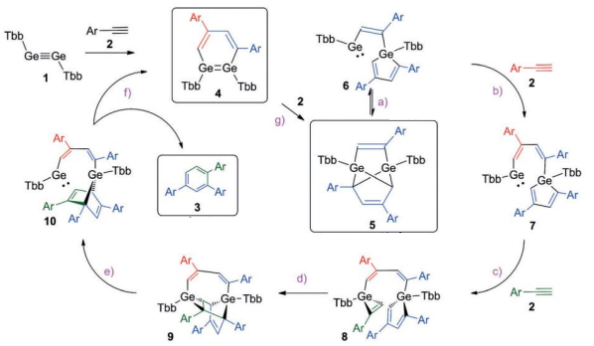
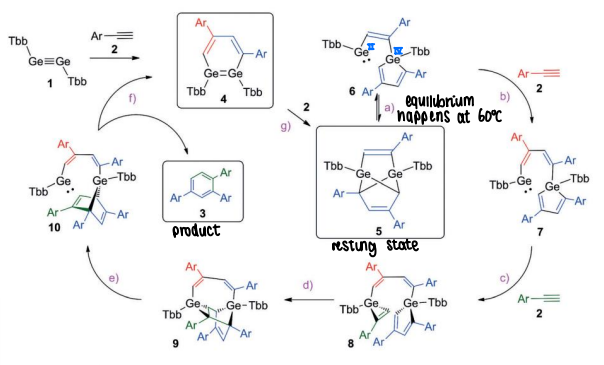
How does digermyne catalyse cyclotrimerisation?
The digermyne reacts with the alkyne, forming compound 5 which acts as the catalyst resting state.
This then forms compound 6 which has a low valent Ge(II) centre with a vacant coordination site that activates a substrate to push the cycle forward.
What is needed to form group 13 multiple bond compounds?
The group 13 compounds are hindered from forming multiple bonds by their electron deficient nature and weak E-E bond energies.
Using Lewis basic ligands (e.g. NHCs or phosphines) allow the development of boron multiple bonds.
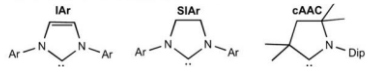
What are the differences between phosphines and NHCs as ligands?
Phosphines:
substituents oriented away from the metal centre, so minimal steric shielding.
Altering the substituents changes the steric and electronic properties.
Weaker sigma-donors, and pi-backbonding can occur from the metal to ligand.
NHCs:
Substituents oriented toward the metal.
R groups closer to the lone pair control sterics, whereas the R groups at the back control electronics.
Stronger sigma-donor and has little/no pi-backbonding.

How does the ligands affect the frequency of C=O IR data?
A ligand that acts as a strong sigma donor means there is greater backbonding occurring from the metal centre into the CO pi* orbitals, therefore weakening the CO bonding.
The wavenumber therefore decreases.
NHCs therefore have lower CO wavenumber values.
What is a comproportionation reaction?
Two molecules containing an element in different O.S. combine into one compound with a middle ground O.S.
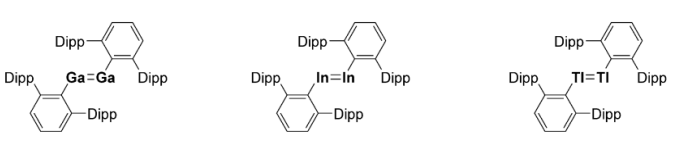
What is required to stabilise group 13 complexes as the group descends?
The stability of the lone pair increases, therefore Lewis base stabilisation is no longer required.
Trans-bent double bonds can be stabilised using sterically bulky ligands instead, such as terphenyl ligands.
How do NHC-B≡B-NHC complexes activate small molecules (H2, CO)?
There is no H2 activation, however CO coupling occurs.
CO is able to accept electrons into its pi* orbital.
The complex is able to donate electrons to H2, but cannot accept electrons back so H2 is not split.
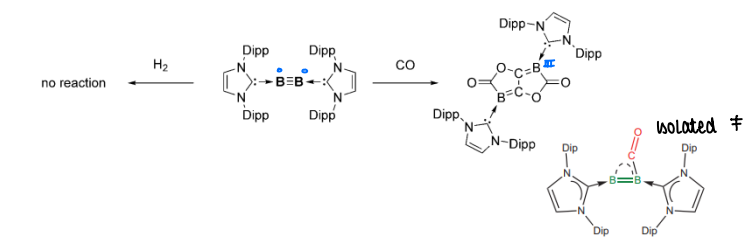
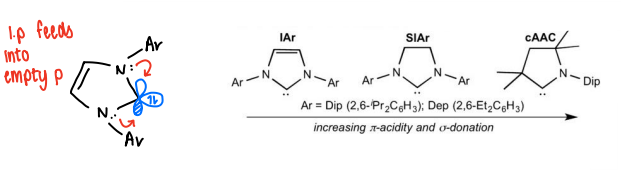
How does changing the NHC ligand affect reactivity of the B≡B complexes?
In NHC, the lone pairs from the nitrogens feed into the empty p orbital. NHCs can be altered to increase backbonding from the boron to lower its electron density.
By removing the double bond so no delocalisation occurs, it can be made a stronger sigma donor, therefore increasing backbonding and so weakening the B-B bond.
By removing a nitrogen from NHC, only one side has a lone pair to donate into the pi orbital of the carbene, therefore the p orbital is more available for backbonding so stabilises the electron rich boron.
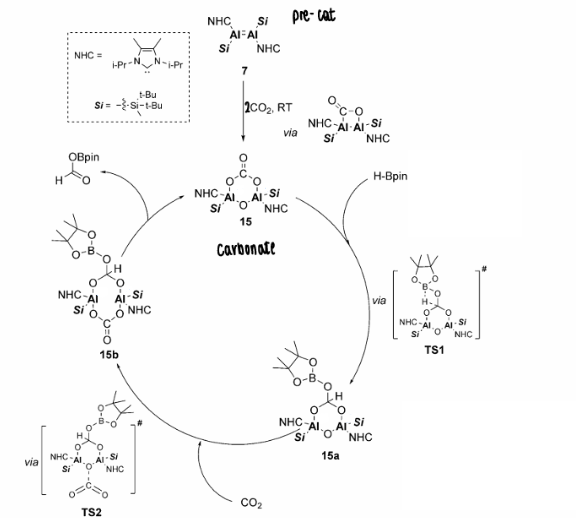
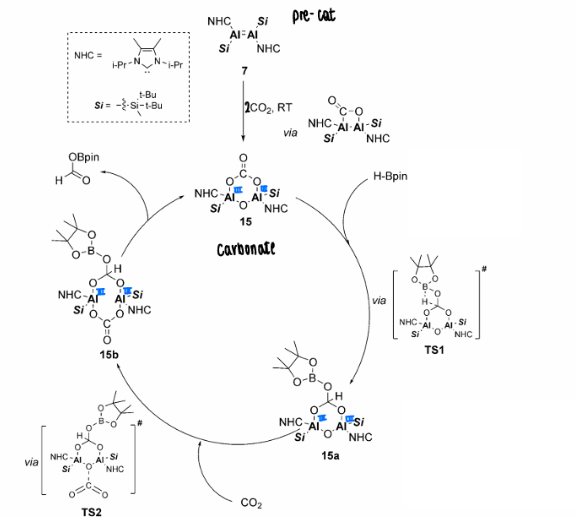
How does dialumene reduce CO2 catalytically?
The dialumene was found to fixate CO2, which formed a carbonyl in the absence of further CO2, or a carbonate in the presence of further CO2.
The carbonate formation is important in the catalytic reduction of CO2, which goes on to react in the catalytic cycle to form a carboxylic acid.
What are ‘low valent’ compounds?
Refers to an element having a low oxidation state and/or a low coordination number.
Tend to be reactive due to strong propensity towards a more stable higher O.S or C.N.
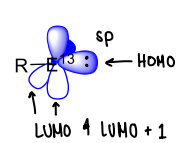
What are the properties of group 13 carbenes?
Form singlet state carbenes, with the HOMO containing the non bonding lone pair with sp character.
Contains two vacant p-orbitals that are very electron deficient.
Requires Lewis base stabilisation to be isolated.
Has one R group.
How do group 14 elements form carbenes?
They form singlet state carbenes with two R groups.
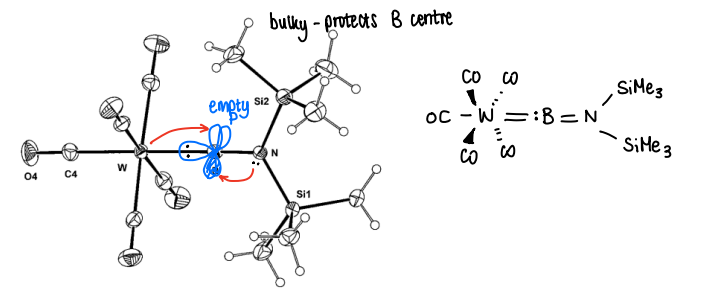
How are group 13 carbenes stabilised by metals and ligands?
Stabilisation is provided by back donation from metals into empty p-orbitals, as well as donation from the lone pair from coordinated nitrogens donated into the empty p
The bulky groups also protect the boron centre.
How are borylenes stabilised by lewis bases?
The boron is electropositive and so does not like to be electron rich.
cAAC (a carbene) can be used as the boron is able to donate its lone pair into the empty p orbitals. the cAAC ligand is more electrophilic so pulls electron density from boron.
Borylenes can be trapped by two coordinating cAAC ligands, forming an sp2 hybridised trigonal planar structure.
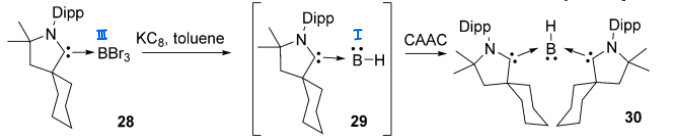
What are the differences between a metal N2 complex and a borylene N2 complex?
Metal complex:
Sigma donation from the N2 sp orbital into the empty metal d orbital.
Backdonation from the filled d orbital into the N2 pi* orbital.
Borylene complex:
Trigonal planar structure around the B.
Sigma donation occurs from the N2 sp to the boron empty sp2.
The lone pair in the p orbital is backdonated to the N2 pi*.
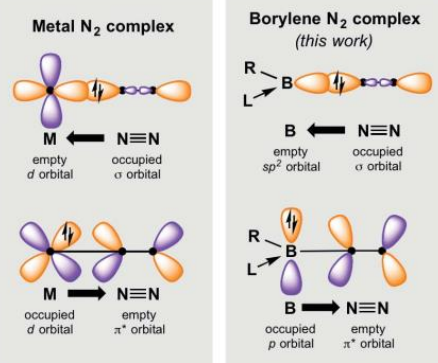
What is the geometry around the parent borylene (R-B)
sp hybridised with linear geometry.
How do boranes form radical anions?
Boranes are reduced.
How do boranes form radical cations?
Via chemical oxidation.
What are alumylenes?
structures containing an aluminium atom which acts as a singlet carbene.
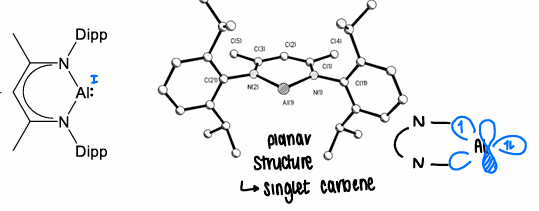
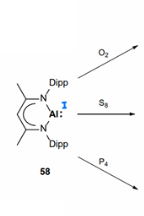
How do alumylenes undergo small molecule activation?
The are able to activate small molecules such as O2, S8 and P4, causing partial reduction of the small molecule.
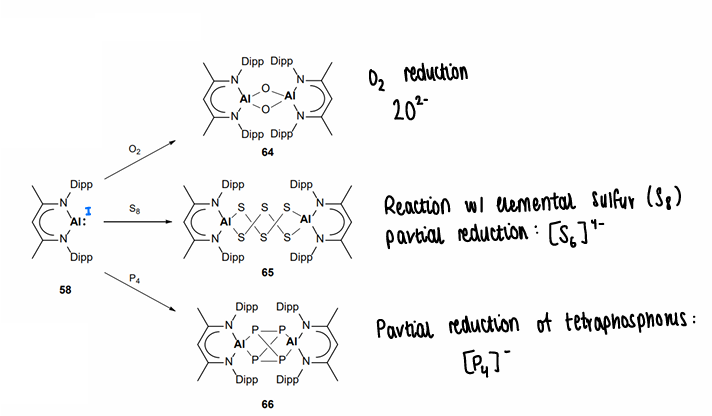
What causes the difference in reactivity of other group 13 elements compared to Al and B?
Descending down the group, the stability of the lone pair increases therefore the reactivity of Ga(I), In(I) and Tl(I) are more driven by basicity (or nucleophilicity) of the main group centre.
Compared to the acidic (or electrophilic) B and Al.
What kind of complexes do group 14 elements form?
They form R2E complexes (tetrylenes), such as carbenes.
How do TMs activate dihydrogen?
Electrophilic activation forms the hydride, followed by H+ transfer.
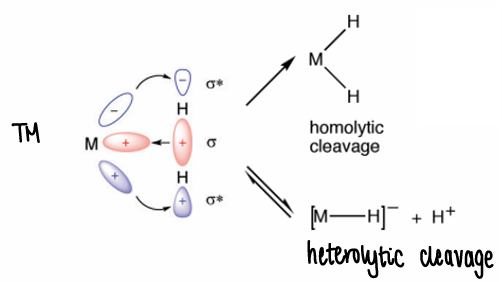
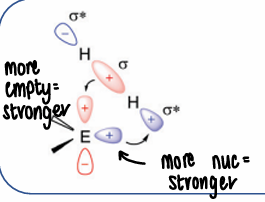
How do main groups activate dihydrogen (orbital diagram)?
The main group donates the lone pair to the dihydrogen sigma* and accepts electrons from the dihydrogen. This occurs via a more nucleophilic pathway.
The more nucleophilic the lone pair, the stronger the interaction.
The more empty the p orbitals, the stronger the interaction.
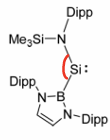
Why do silylenes require sterically demanding groups?
Silylenes tend to undergo dimerisation to form multiple bonds, the sterically large group prevents this by changing the bite angle, this changes the p and n energy gap.
Small bite angle= large energy gap.
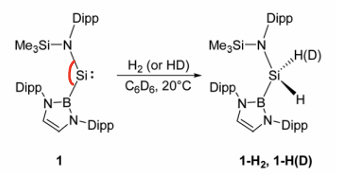
How do silylenes activate dihydrogen?
The dihydrogen bonds to the Si cis.
The wider bond angles decreases the singlet-triplet gap.
Carbenes have a smaller singlet-triplet gap than heavier main group elements, therefore can activate dihydrogen.
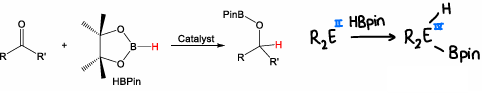
What is hydroboration?
The selective and efficient conversion of carbonyl compounds to alcohols by addition of the boron compound to the double bond.
Can also occur across main group polar bonds.
What are the properties of p-block anions?
As most low valent species have a lone pair and an empty p orbital they can be described as ambiphilic.
The negative charge can be increased on the central element which enhances nucleophilic character.
How do you stabilise the negative charge of group 13 anions?
Use an electron withdrawing group.
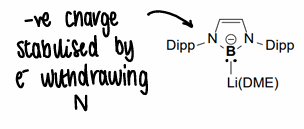
Why are crowns used for group 13 anions?
Binding the crown to the cation prevents the lone pairs on the anion interacting with the cation.
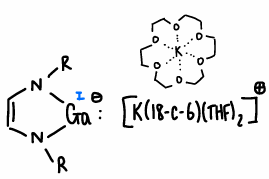
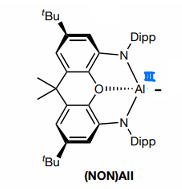
What are aluminyles?
Aluminium bonded to a NON group. Contains a negative charge.
What are p-block cations?
Positively charged species with enhanced electrophilic character, they have 6 valence electrons therefore are lewis acidic.
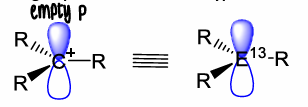
How do you form R2E+ cations?
The removal of a R- group generates R2E+. This has two vacant p orbitals and so is highly reactive.
Often found in combination with a lewis base to aid stability.
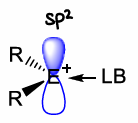
Why is the choice of lewis base important for stabilisation of R2E+?
Short range coulombic interactions lead to electron precise bonds that quench the cationic reactivity. A LB that is too strong will cause quenching.
Weakly coordinating anions provide long range electrostatic interactions to maintain the vacant coordination site.
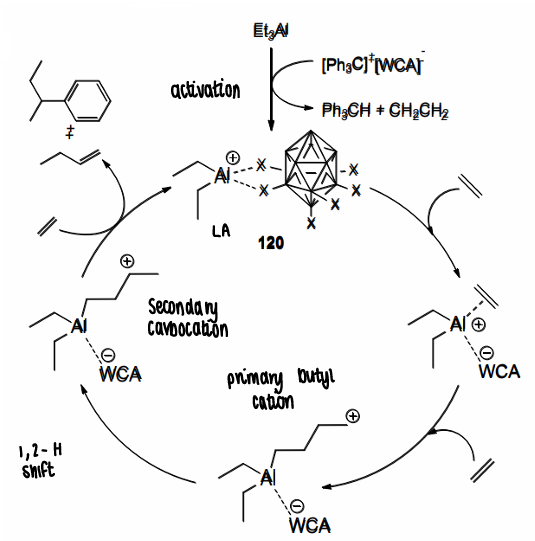
What are alumeniums?
A aluminium cation containing compound.
Can be formed via removal of an R group, such as by activation by a carborane.
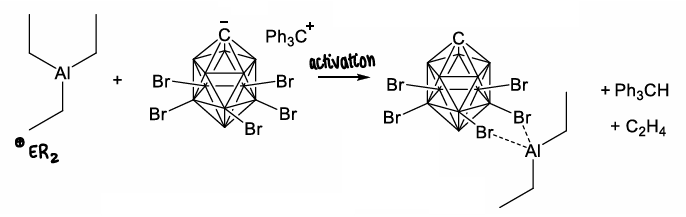
How are alumeniums used for catalysis?
They are able to catalyse ethylene oligomerisation.