Formulas
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
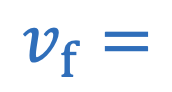
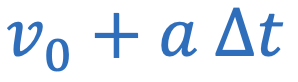
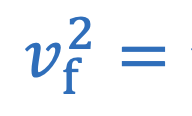
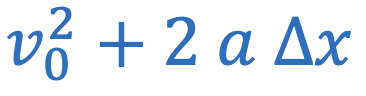
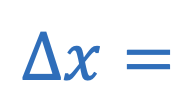
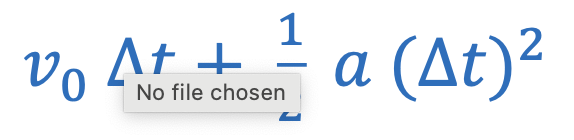
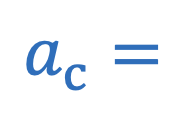
Centripetal acceleration
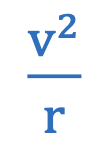
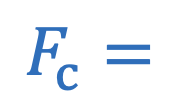
Centripetal force
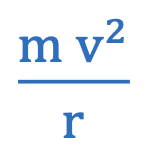
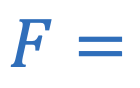



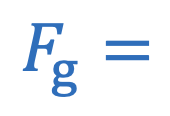
Universal law of gravitation
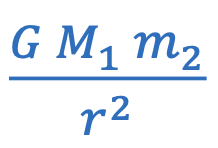
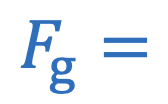
Force of gravity
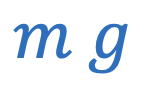
Torque
r = Distance from pivot point to force of application
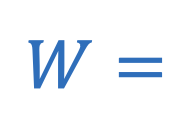
Work (kinematics)

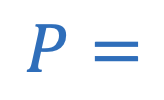
Power
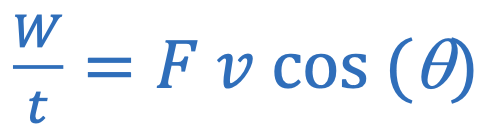
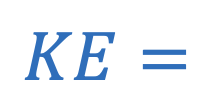
Kinetc energy
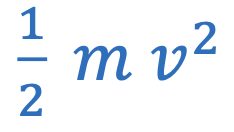
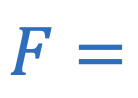
Hook’s law
Force required to compress a string
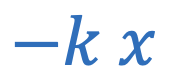
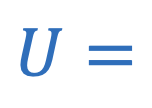
Elastic potential energy
k = Spring constant
x = Displacement
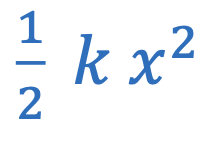
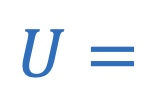
Gravitational potential energy
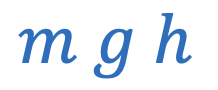
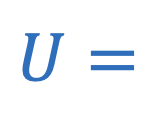
Gravitational potential energy between two objects
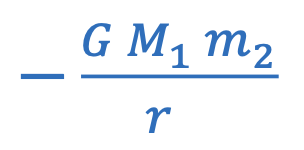
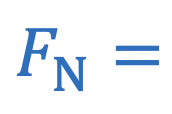
Normal force on inclined plane


Force of friction on inclined plane

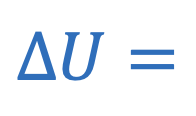
Change in internal energy
Q = Heat
W = Work
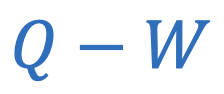
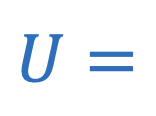
Internal energy of gas
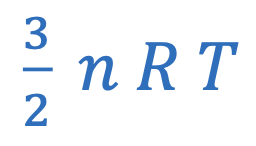
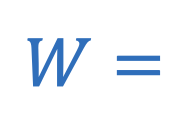
Work (thermodynamics)

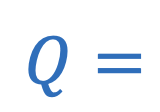
Heat
c = Specific heat constant
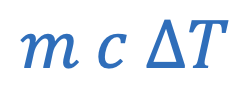
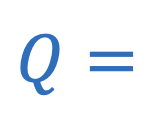
Heat energy required or released during phase change
HL = Latent heat coefficient

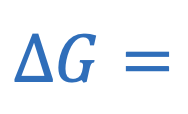
Gibb’s free energy
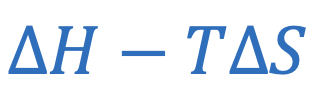

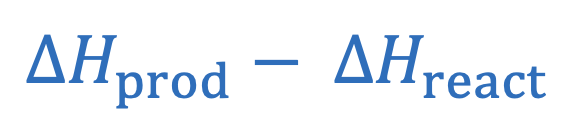
Ideal gas law
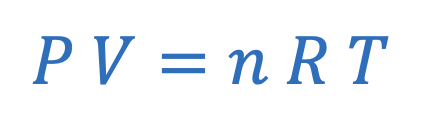
Boyle’s law
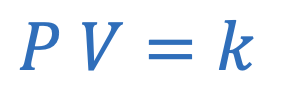
Gay-Lussac’s law
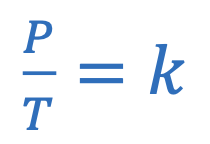
Charle’s law
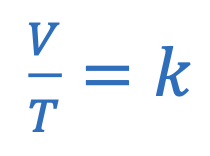
Avogadro’s law
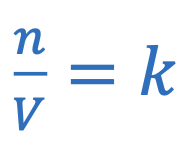
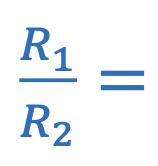
M = Molar mass
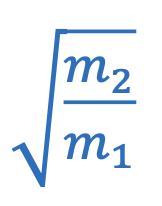
Concentration of dissolved gas
C = (Kh)(Pgas)
Kh = Solubility coefficient
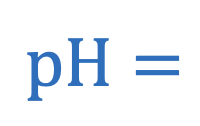
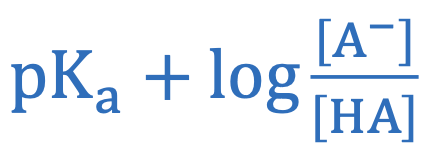
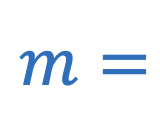
Molality
mol of solute / kg of solvent
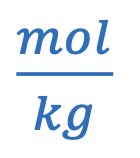
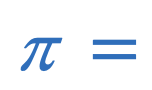
Osmotic pressure
i = Number of particles
M = Molarity
R = Ideal gas constant 0.08
T = Temperature (K)

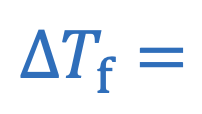
Freezing point depression
i = Number of particles
m = Molality
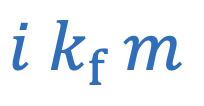
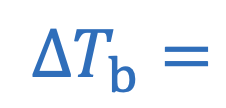
Boiling point elevation
i = Number of particles
m = Molality
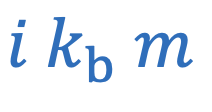
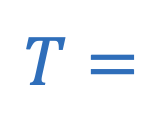
Period
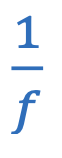
Snell’s law
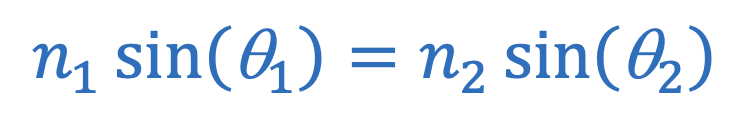
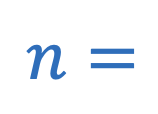
Index of refraction
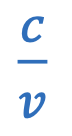
Energy of photon
h = Plank’s constant = 6.626×10-34
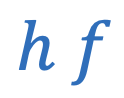
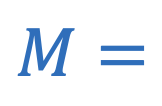
Magnification
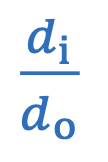
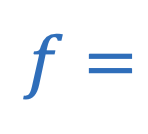
Focal length
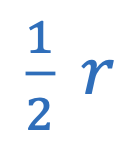
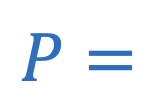
Power of lens
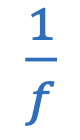
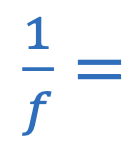
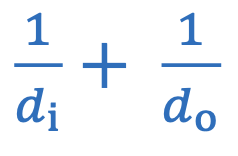
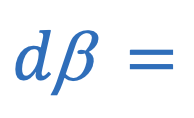
Decibels
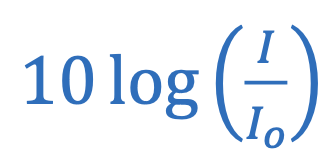
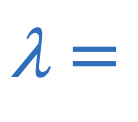
Wavelength of fixed string or open pipe
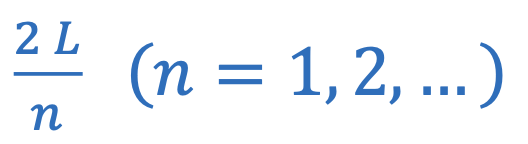
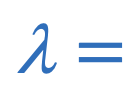
Wavelegnth of closed pipe
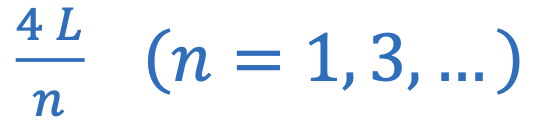
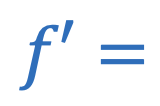
Doppler effect
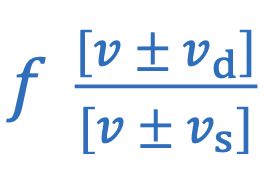
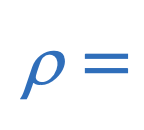
Density
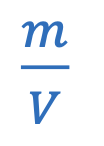
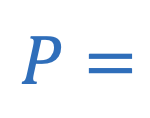
Pressure
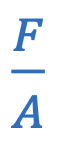
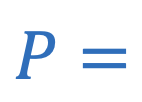
Absolute pressure
pgh = Hydrostatic pressure
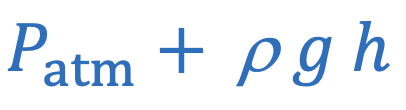
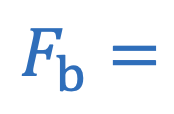
Buoyant force
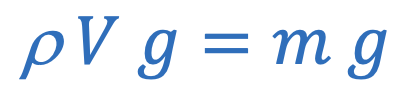
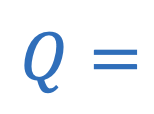
Flow rate
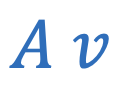
Bernoulli’s equation
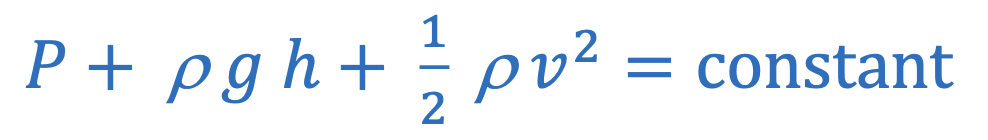
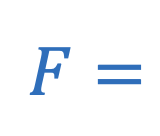
Coulomb’s law
Electrostatic force
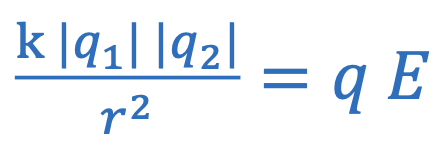
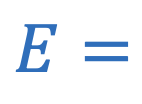
Electric field strength
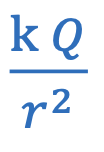
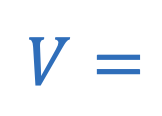
Electric potential
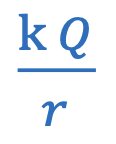

Electric potential energy
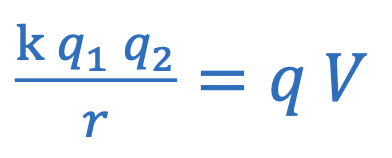
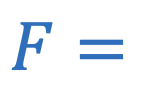
Magnetic force (charge)
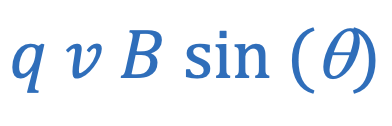
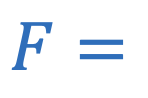
Magnetic force (current)
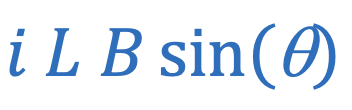
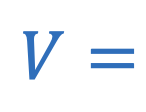
Ohm’s law

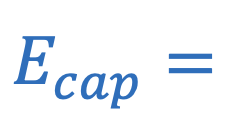
Electric field strength of parallel plate capacitor
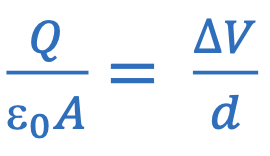
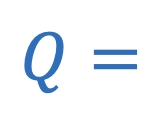
Charge
C = Capacitance

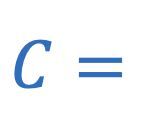
Capacitance
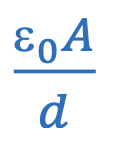
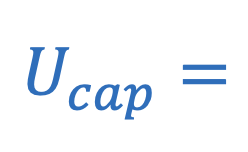
Electric potential energy in capacitor

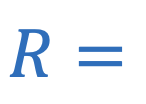
Resistance
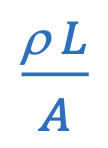
Avogadro’s number
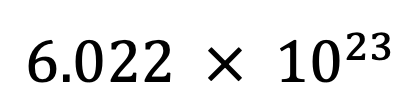
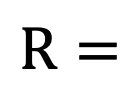
Gas constant
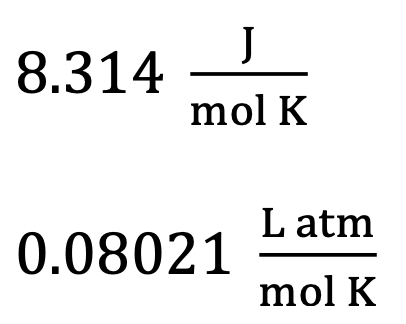
Planck’s constant
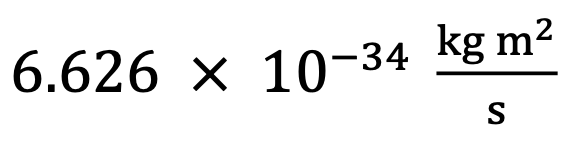
Density of water
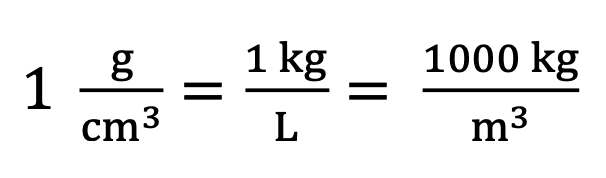
Speed of light
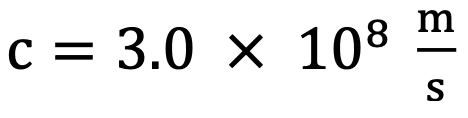
Faraday’s constant
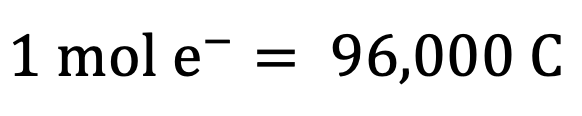
Units of Newton
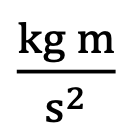
Units of Joule
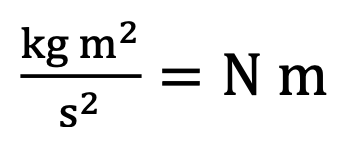
Units of Pascal
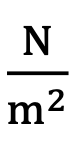
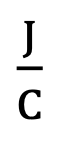
Units of Volt
Units of Amp
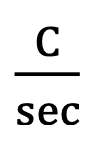
Units of Watt
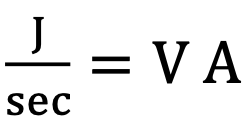
Units of Ohm
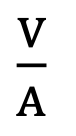
Units of Farad
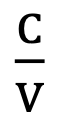
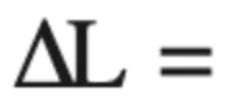
Thermal expansion
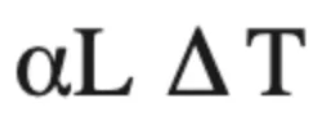
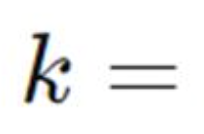
Arrhenius equation
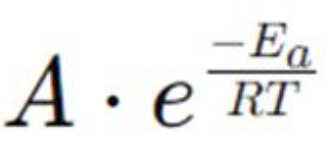

q = Heat of reaction'
n = Moles
