linewidths in spectroscopy
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
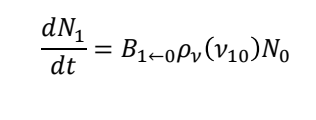
for an absorption from E0 to E1, what are all the terms?
N0 and N1 are populations in state 0 and state 1
B1←0 is the rate constant for absorption from state 0 to 1
p𝜈(𝜈10) is the radiation density at the precise frequency 𝜈10 needed to drive a transition from ground to excited state
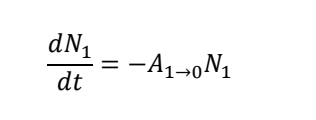
for an emission from E1 to E0, what are all the terms?
N1 is the populations in state 1
A1→0 is the rate constant for the spontaneous emission from state 1 to 0
show stimulated emission diagram from E1 to E0
hint : laser amplification
resulting photons are exactly in phase so they add
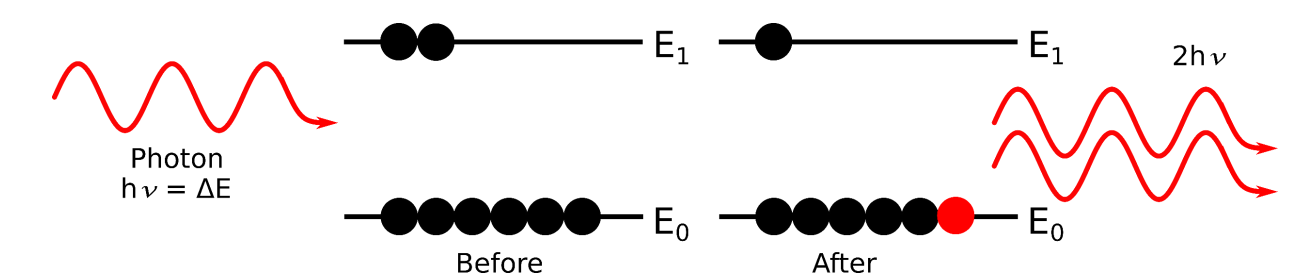
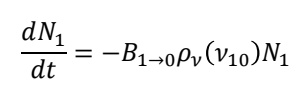
for stimulated emission from E1 to E0, what are all the terms?
N1 is the populations in state 1
B1→0 is the rate constant for the stimulated emission from state 1 to 0
p𝜈(𝜈10) is the radiation density at the precise frequency needed to drive a transition from excited to ground state
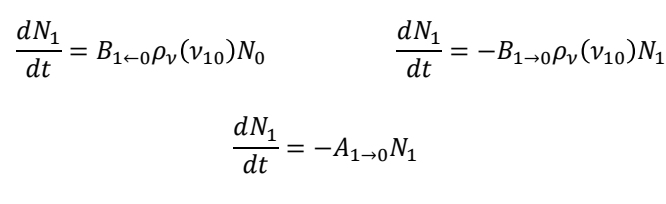
how are these three equations related to the lifetime?
a measurement of the absorption process can be used to determine the lifetime of the excited state
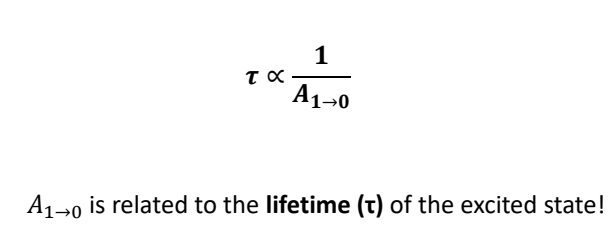
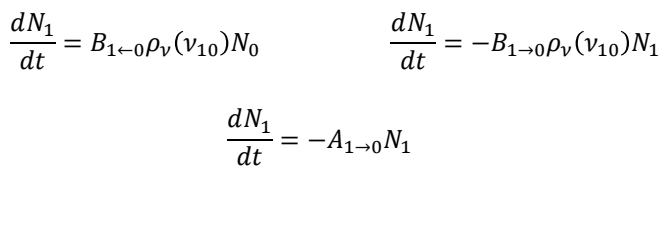
how are the Einstein coefficients related at equilibrium?

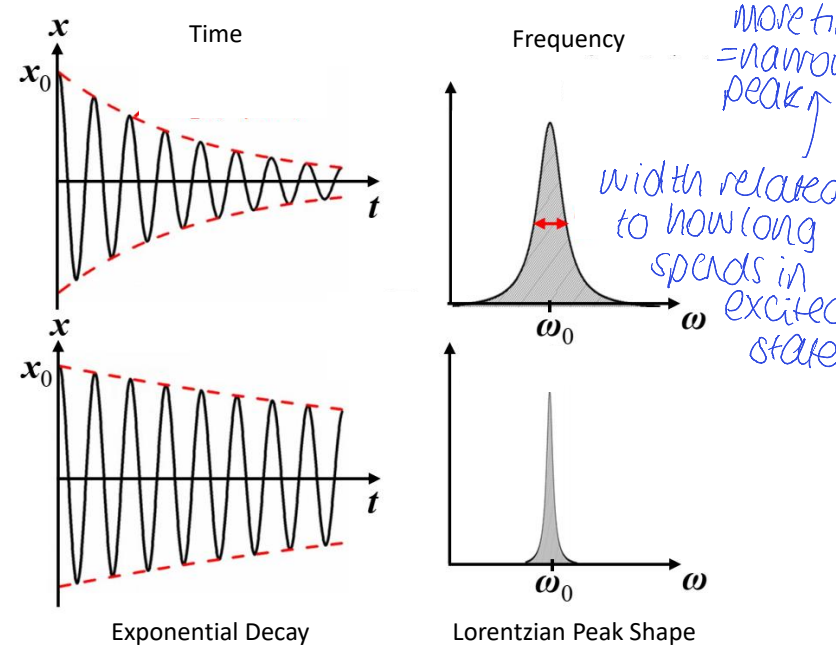
how is the width of a frequency peak related to the lifetime of excited state?
more time spent in excited state means a narrower peak
show time decay (for a greater vs smaller amount of time) vs resulting peak shapes
what is the name of this type of peal?
Lorentzian peak shape
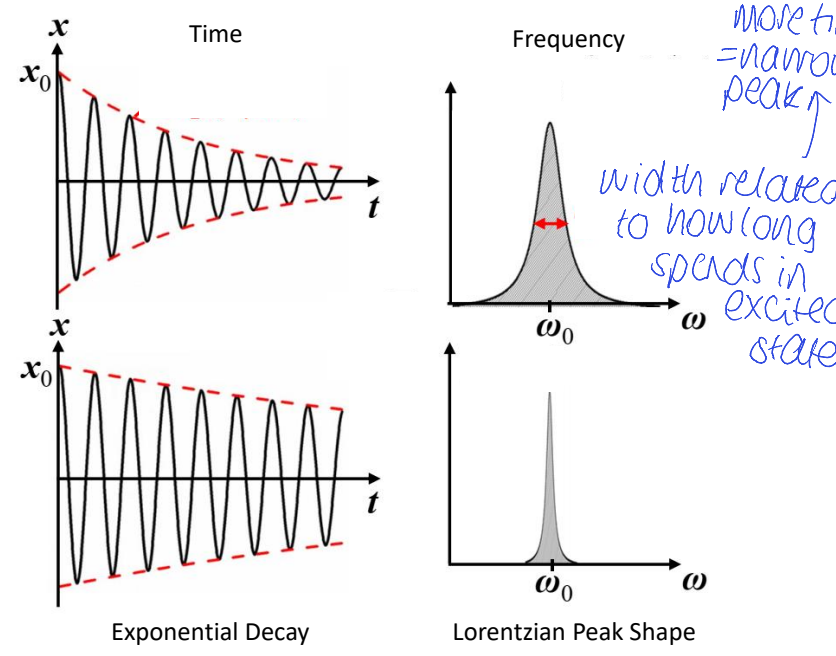
what is the time energy uncertainty principle?
what is the linewidth the same as?
what happens when the lifetime changes?
linewidth is that of a damped oscillator
homogenous broadening
show doppler broadening diagram

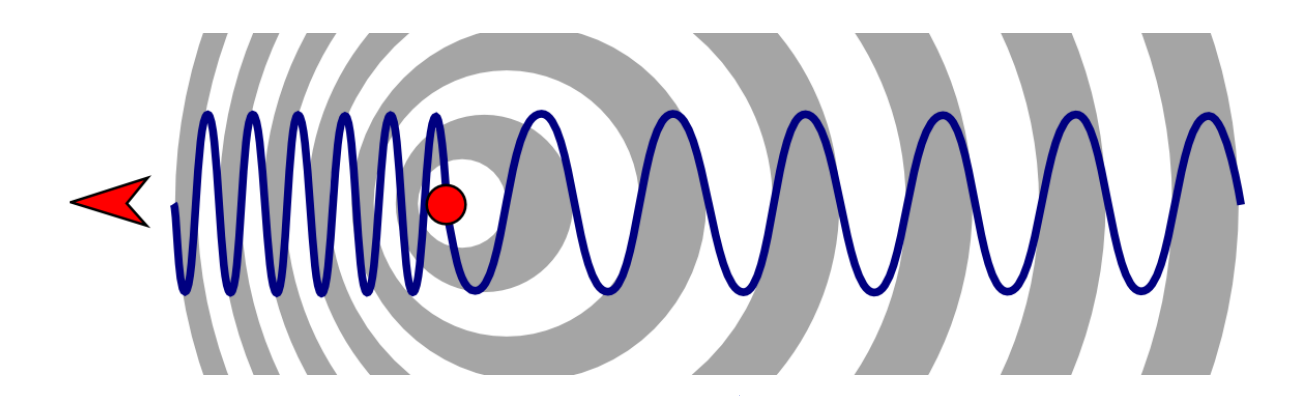
what is the equation for frequency of approaching?
s is velocity of molecule
c is speed of light
𝜈0 is vibrational frequency
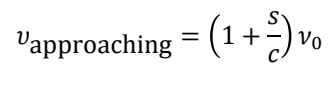
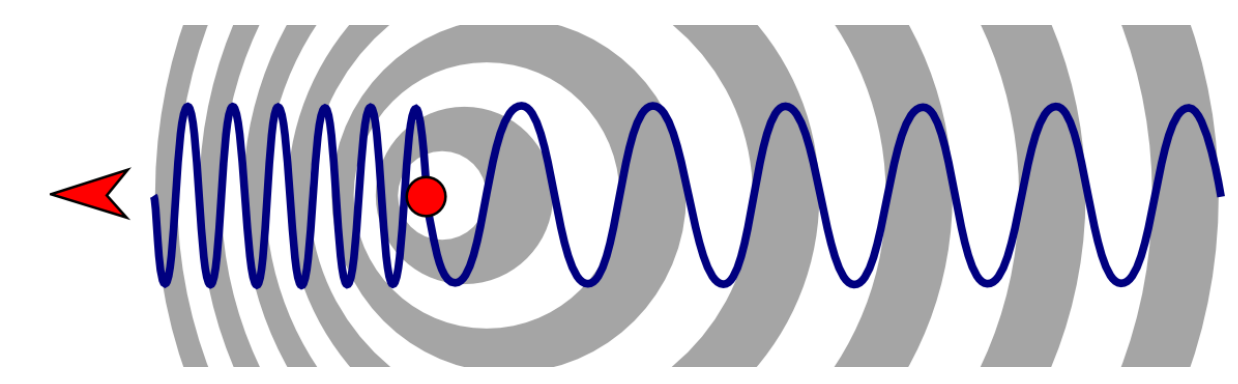
what is the equation for frequency of receding?
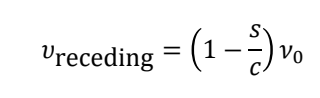
how is a spectra related to Lorentzian peaks?
it is a sum of homogenously broadened Lorentzian peaks
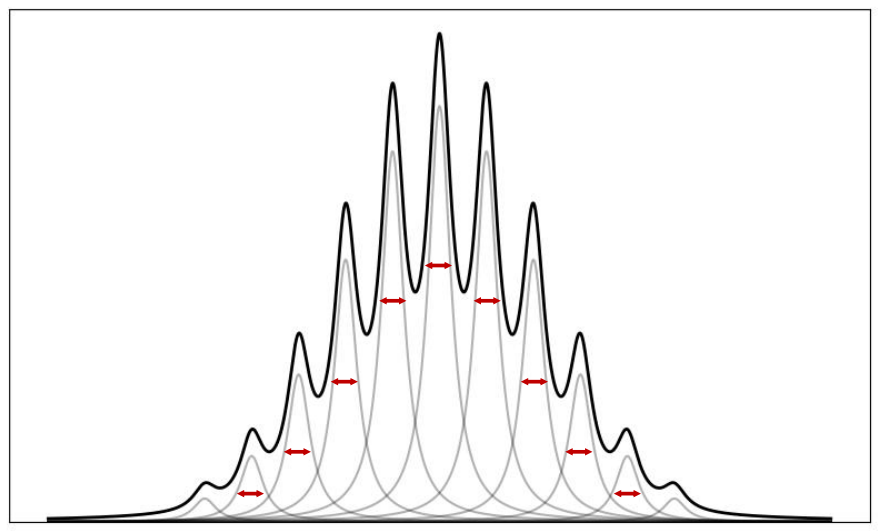
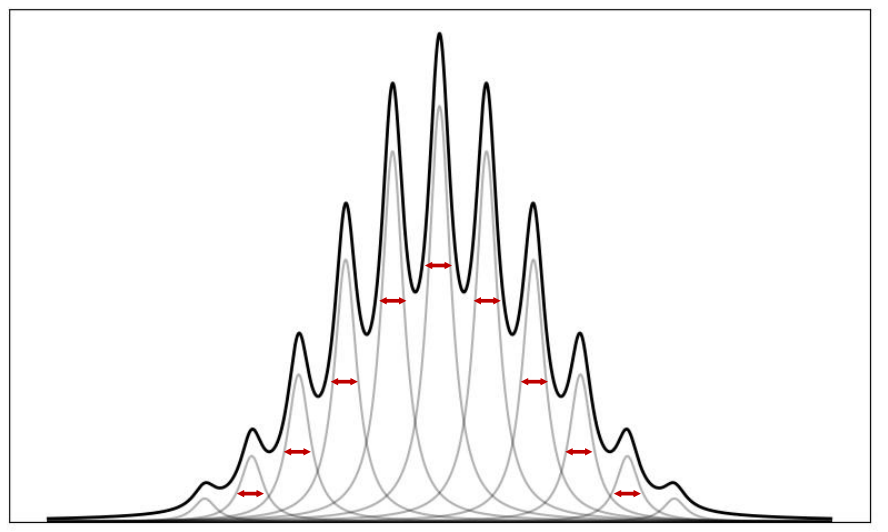
when is the spectra Lorentzian?
when there is a series of molecule travelling slightly faster/slower
gives normal distribution
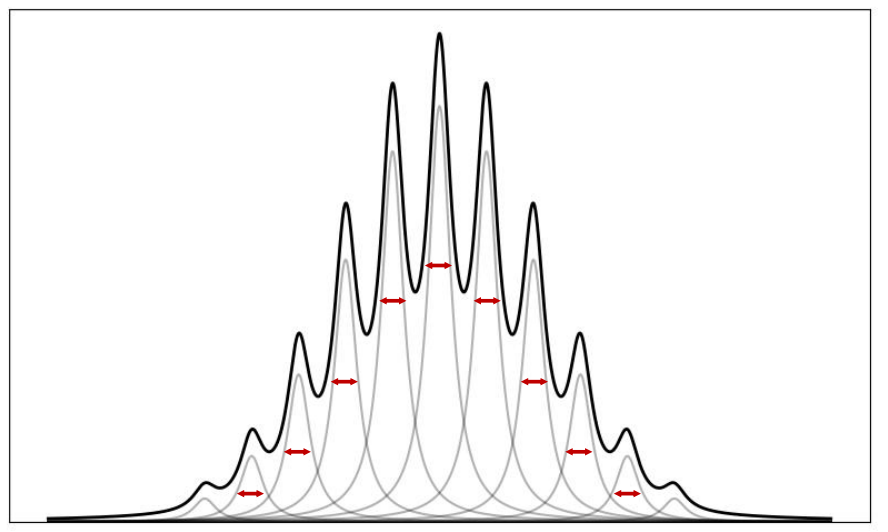
when does the spectra become Gaussian?
when the transition frequency varies because of many small independent perturbations
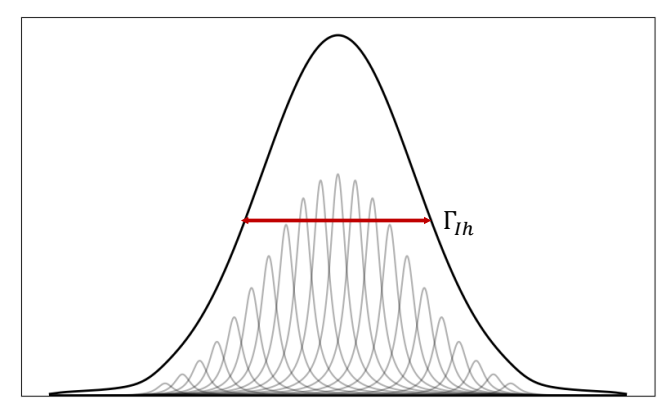
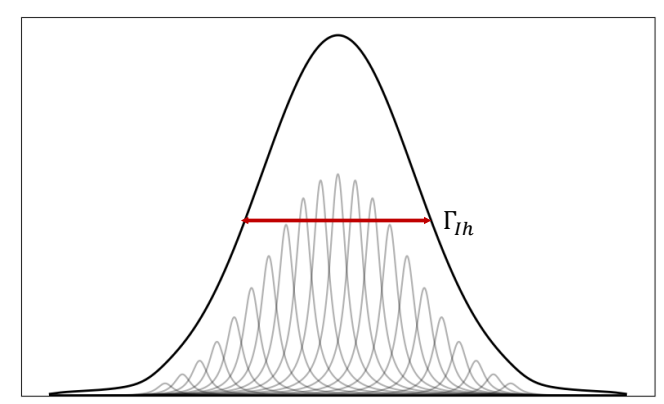
how is the Doppler effect an example of this? when is this dominant?
inhomogeneous broadening
dominant at low P, high T and high 𝜈
any time the transition frequency varies because of many small independent perturbations, peak shape becomes Gaussian
in gases, what is the relation between collision lifetime and frequency?
as collision frequency increases, time in excited state decreases

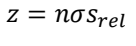
what are the terms?

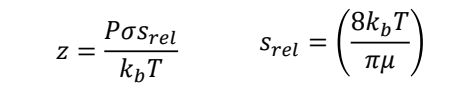
when is pressure broadening in a gas significant? what is the spectra peak shape?
significant at high P, low T, low 𝜈
example of homogenous broadening, linewidth is that of damped oscillator = Lorentzian peak shape