4.5 Energy changes
1/31
Earn XP
Description and Tags
Completed flashcard set
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Why do chemical reactions often involve transfers of energy?
Due to the breaking and formation of bonds
Where do chemicals store their energy?
In the chemical bonds that hold their particles together
If the products of a reaction store more energy in their chemical bonds than the original reactants, where did that extra energy come from?
The energy must have been taken in from the surroundings during the reaction
What is an exothermic reaction?
An exothermic reaction is a reaction that transfers energy to the surroundings, so the temperature of the surroundings increase
As energy has been transferred to the surroundings, the product molecules must have less energy than the reactants, by the amount transferred
Give examples of exothermic reactions
Combustion
Many oxidation reactions
Neutralisation
Give everyday uses of exothermic reactions
Hand warmers
Self heating cans (eg. for coffee)
What is an endothermic reaction?
An endothermic reaction is a reaction that takes in energy from the surroundings, so the temperature of the surroundings decreases
As energy has been taken in from the surroundings, the product molecules must have more energy than the reactants, by the amount transferred
Give examples of endothermic reactions
Thermal decompositions
Reaction of citric acid and sodium hydrogencarbonate
Give everyday uses of endothermic reactions
Sports injury packs
What do reactant particles need to react?
Chemical reactions can occur only when reacting particles collide with each other with sufficient energy
What is activation energy?
The minimum amount of energy that particles need to react
What do reaction profiles show?
Reaction profiles are diagrams that show the relative energies of the reactants and products in a reaction.
It also shows the activation energy and the overall energy change of a reaction
Draw the reaction profile for an exothermic reaction
The energy of the products is less than the energy of the reactants as energy has been transferred to the surroundings
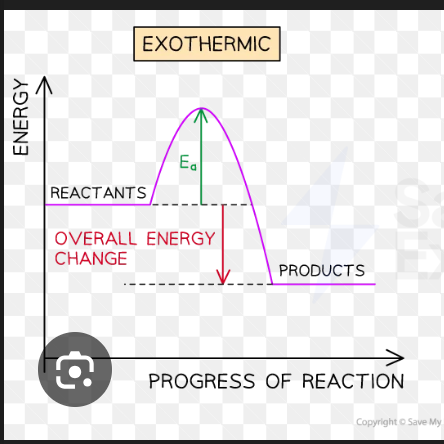
Energy changes are negative as the particles have lost energy.
Draw the reaction profile for an endothermic reaction
The energy of the products is greater than the energy of the reactants as energy has been taken in from the surroundings
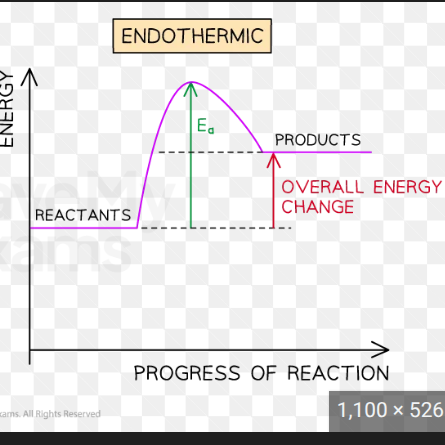
The overall energy change is positive as the particles have gained energy from the surroundings.
Why are there energy changes during reactions?
Energy must be supplied to break bonds in the reactants
Endothermic process
Energy is released when bonds in the products are formed
Exothermic process
What is the overall energy change of a chemical reaction?
Relate this to exothermic and endothermic reactions
The difference between the sum of the energy needed to break the bonds in the reactants and the sum of the energy released when bonds in the products are formed.
In an exothermic reaction, the energy released from forming new bonds is greater than the energy needed to break existing bonds
In an endothermic reaction, the energy needed to break existing bonds is greater than the energy released from forming new bonds
How do cells work?
A cell is made up of two different electrodes in contact with an electrolyte;
a chemical reaction between the electrolyte and the electrodes occurs on the surface of the electrode which creates a charge difference/ potential difference between the electrodes
If the electrodes are then connected by a wire, the charge difference/ potential difference can cause charge to flow and electricity to be produced
A cell contains chemicals which react to produce electricity
What does the voltage produced by a cell depend on?
Different metals will react differently with the same electrolyte; this is what creates the charge difference/ voltage of the cell.
Type of electrodes used
The greater the difference in reactivity of the two different metals used as electrodes, the greater the potential difference produced
Type of electrolyte
How can a simple cell be made?
A simple cell can be made by connecting two different metals in contact with an electrolyte
What are batteries?
Batteries consist of two or more cells connected in series to provide a greater voltage
Why can non-rechargeable cells and batteries only produce electricity for a certain period of time?
Because eventually one of the chemicals/ reactants will get used up in the reactions , so can no longer react to create a potential difference
Give an example of non-rechargeable batteries
Alkaline batteries are non-rechargeable
There is no way to reverse the reactions in these batteries, so they are non-rechargeable
What must be true for a cell to work?
The metals used as the electrodes must have different reactivities; the greater the difference in reactivity, the greater the potential difference produced.
Electrodes of the same type of metal have the same reactivity so do not produce a potential difference.
Why and how can rechargeable cells and batteries be recharged?
Because the chemical reactions in the cells can be reversed when an external electrical current is supplied
This means that the chemical reactions that create the charge difference at the two different electrodes can occur again and produce a potential difference again, causing charge to flow and thus producing electricity
What are alkaline batteries?
An example of non-rechargeable batteries
What is meant by electrochemical oxidation
A substance loses electrons at an electrode due to an electric current
This occurs within fuel cells to produce a potential difference
What are fuel cells?
A fuel cell is an electrical cell that is supplied by an external source of fuel (eg. hydrogen) and oxygen or air
The fuel is oxidised electrochemically (loses electrons at an electrode due to an electric current) within the fuel cell to produce a potential difference
There are different types of fuel cells using different fuels and different electrolytes
Explain how hydrogen fuel cells work
The hydrogen fuel cell has two electrodes which are in contact with an electrolyte
The hydrogen goes into the negative electrode compartment and the oxygen goes into the positive electrode compartment
At the negative electrode, the hydrogen is oxidised (loses electrons) to produce H+ ions
H2 → 2H+ + 2e-
Both electrodes are connected by a wire, and the electrons lost by the hydrogen pass through the wire to the positive electrode
The flow of electrons is the electric current
The hydrogen ions move through the electrolyte to the positive electrode
At the positive electrode, the hydrogen and electrons can react with the oxygen being entered into the positive electrode compartment to produce water
O2 + 4H+ + 4e- → 2H2O
The water leaves the fuel cell via the outlet
Write the half equations for the electrode reactions in the hydrogen fuel cell
Negative electrode:
Hydrogen is oxidised (loses electrons) to become a positive H+ ion
H2 → 2H+ + 2e-
The electrons move through the negative electrode through the external wire/ circuit to the positive electrode (this flow of electrons is the electricity produced)
Positive electrode
The hydrogen and electrons react with the oxygen entering the cell to form water
O2 + 4H+ + 4e-→2H2O
The water leaves the hydrogen fuel cell through an outlet
Give the names of the electrodes for electrolysis and in hydrogen fuel cells
Electrolysis:
Positive: anode
Negative: cathode
Hydrogen fuel cells:
Positive: cathode
Negative: anode
Give advantages of using hydrogen fuel cells in comparison to using rechargeable cells and batteries (in cars)
No toxic chemicals to dispose of at the end of the cell’s life; whereas rechargeable batteries are made of highly toxic chemicals
Hydrogen fuel cells take less time to refuel (whereas rechargeable cells take a long time to recharge)
Hydrogen fuel cells allow cars to travel further before refuelling than recharging rechargeable batteries (hydrogen has a greater energy density)
No loss of efficiency over time, whereas rechargeable cells lose efficiency
Give the disadvantages for using hydrogen fuel cells over rechargeable cells and batteries
Disadvantages:
Hydrogen is a gas, so takes up much larger amounts of space to store than rechargeable batteries
Hydrogen is explosive when mixed with air so is hard to store safely, is dangerous
Making hydrogen fuel requires energy, which usually comes from fossil fuels