Alkenes ✅
1/43
Earn XP
Description and Tags
need to do flashcards on polymers
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
are alkenes saturated or unsaturated
unsaturated
give the general formula for alkenes
CnH2n
for a carbon chain length longer than three carbons, what must you do
state the position of the double bond
e.g. but-1-ene vs but-2-ene
what type of bonds attach hydrogens to carbons in an alkene
sigma bonds
what how is a pi bond formed
formed by the sideway overlap of adjacent p orbitals above and below the bonding C atoms
why does a pi bond mean that C=C double bonds can’t rotate
overlap of 2 p-orbitals forms an orbital with a cloud of electron density above and below the single bond
called a pi orbital
meaning C=C bonds can’t rotate
a C=C is comprised of a … bond surrounded by a … bond
sigma
pi
what is the shape around carbon atoms in an alkene and what is the bond angle
trigonal planar
120 ( 3 bonding regions and 0 lone pairs )
define stereoisomerism
compounds with the same structural formula but a different arrangement of atoms in space
give another name for E/Z isomerism
geometric isomerism
read page 6 in alkenes booklet that talks you through E/Z isomerism and naming
okay
define electrophile
electron pair acceptor
what type of reactions do alkanes undergo and what type of reactions are these most commonly
addition
electrophilic addition
Addition of Hydrogen
name for this reaction
reagents
conditions
draw out the displayed equation for reaction of ethene + H2
what happens in this reaction
when is it done- example
hydrogenation
H2
Nickel catalyst
okay
unsaturated alkene becomes saturated
used in manufacture of margarine

Addition of halogens
name for this reaction
reagents
conditions
draw out the displayed equation for reaction of ethene + Br2
when is addition of bromine carried out
when is addition of iodide carried out
halogenation
Cl2 Br2 I2
room temp
okay
laboratory test for the presence of double bonds
to test for unsaturation in vegetable oils

describe the chemical test for alkenes
add bromine water
shake
orange → colourless
Addition of hydrogen halides
name for this reaction
reagents
conditions
draw out the displayed equation for reaction of ethene + HCl
when is addition of bromine carried out
when is addition of iodide carried out
halogenation
HCl HBr or HI
room temp

order the first 4 hydrogen halides in order or decreasing bond enthalpy
H-F, H-Cl, H-Br, H-I
Reaction with Steam
name for this reaction
reagents
conditions
draw out the displayed equation for reaction of ethene + H2O
when is this reaction carried out
hydration
H2O (g) (temp>100*C)
Phosphoric acid H3PO4 catalyst
okay
addition of steam across a double bond is used in industrial manufacture of alcohols

Reaction with concentrated H2SO4
reagents
conditions
draw out the displayed equation for reaction of ethene + H2SO4
name the product formed
what can the sulphuric acid be used as
what is the electrophile in this reaction
H2SO4
room temp
okay
ethylhydrogensulphate
a catalyst for the addition of water
partially positively charged H atom in sulphuric acid molecule

why do addition reactions happen with alkenes
pi bond is easily broken
so atoms can be added
what type of product is formed in an addition reaction
saturated
what happens when an unsymmetrical alkene goes under an addition reaction with hydrogen halides or other non symmetrical molecules
2 products are formed
1 minor and 1 major
the hydrogen becomes attached to the carbon with the mot hydrogen attached to it already
therefore that will produce the major product
draw the mechanism for the electrophilic addition reaction of ethene (symmetrical molecule) with a symmetrical molecule e.g. Br2
okay

draw the mechanism for the electrophilic addition reaction of ethene (symmetrical molecule) with an unsymmetrical molecule e.g. HBr
okay

Unsymmetrical alkenes
what is formed when unsymmetrical alkenes undergo reactions with hydrogen halides and other non symmetrical molecules
why does this happen
e.g. draw the mechanisms with propene with hydrogen bromide
major and minor product is formed
major product is formed from carbocation with more alkyl groups attached which is therefore more stable carbocation ( always state classification of carbocation for major and minor products in exam )
remember: when drawing mechanism for major product, H attaches to the C either side of double bond which has the most H’s attached to it already!
What is a primary carbocation
Carbocation bonded to 1 C atom

What is a secondary carbocation
Carbocation bonded to 2 C atoms

What is a tertiary carbocation
Carbocation bonded to 3 C atoms

define addition polymerisation
reaction in which many monomers containing at least one C=C double bond form long chains of polymers as the only product
define term polymer
long chain molecule that is made up of many repeating units
steps for drawing repeat unit of polymer when given monomer
double bond → single bond
brackets and n
extending bonds either side out of brackets
yes
draw the displayed formula for the repeat unit of poly(ethene)
okay
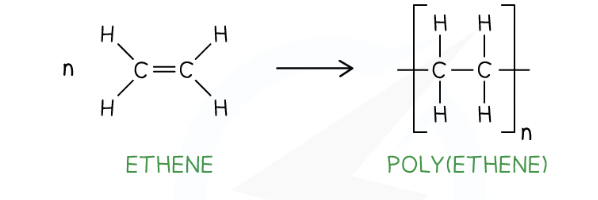
draw the displayed formula for the repeat unit of poly(chloroethene)
okay
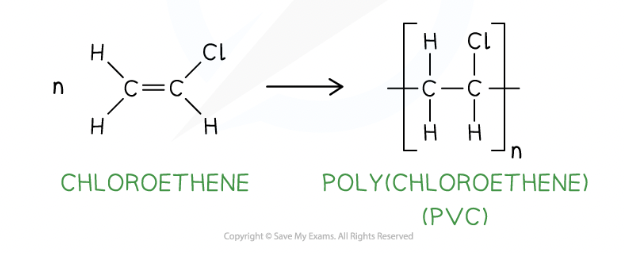
how does adding a plasticiser change a polymer
makes it more flexible
by preventing the polymer chains from being close to one another
this disrupts the van der Waal forces between the chains, making them weaker
so chains slide over each other easily
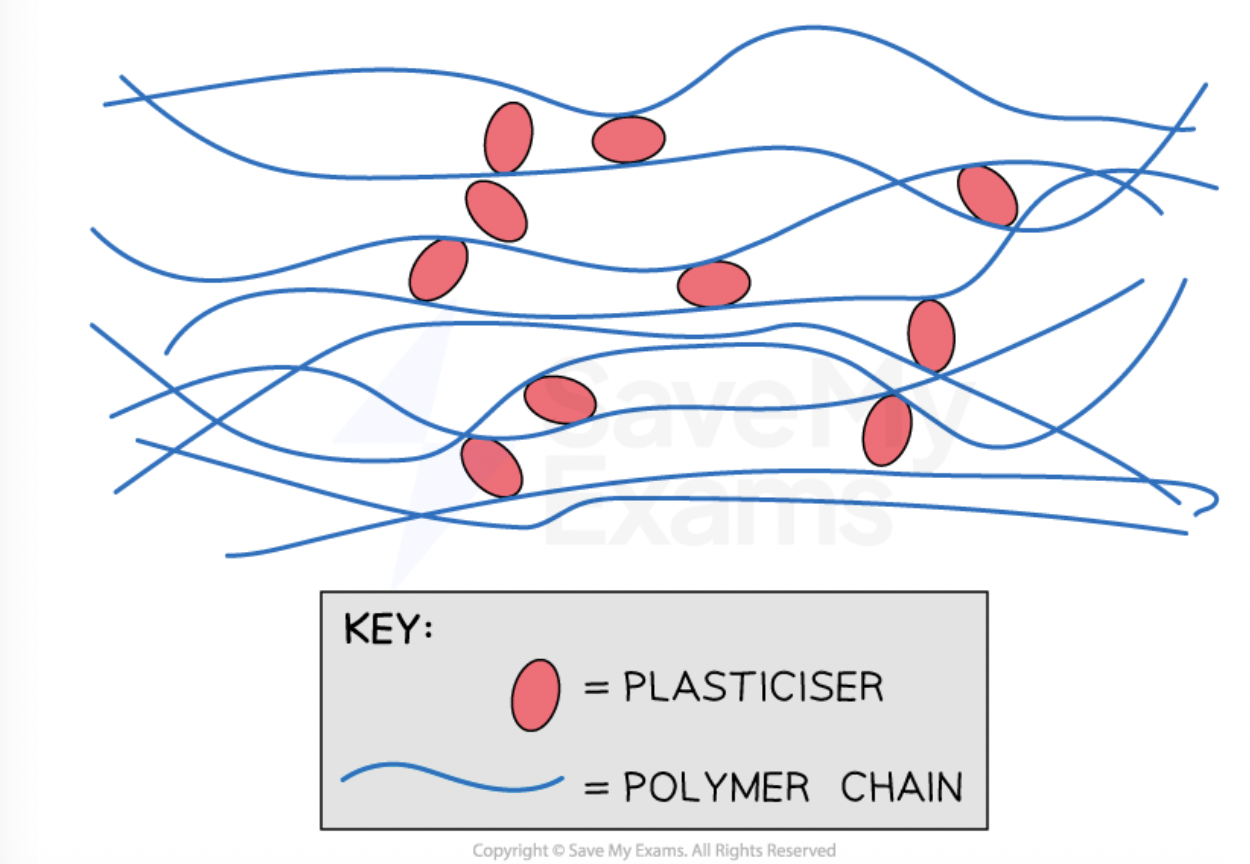
what can PVC be used for
before adding plasticiser
after adding plasticiser
making drainpipes
make pool liners
explain why poly(alkenes) are not biodegradable
inert and very unreactive
made up of strong C-C bonds
no C=C double bonds
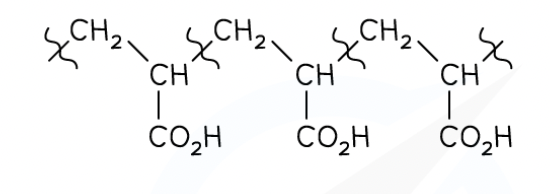
deduce the monomer of this addition polymer
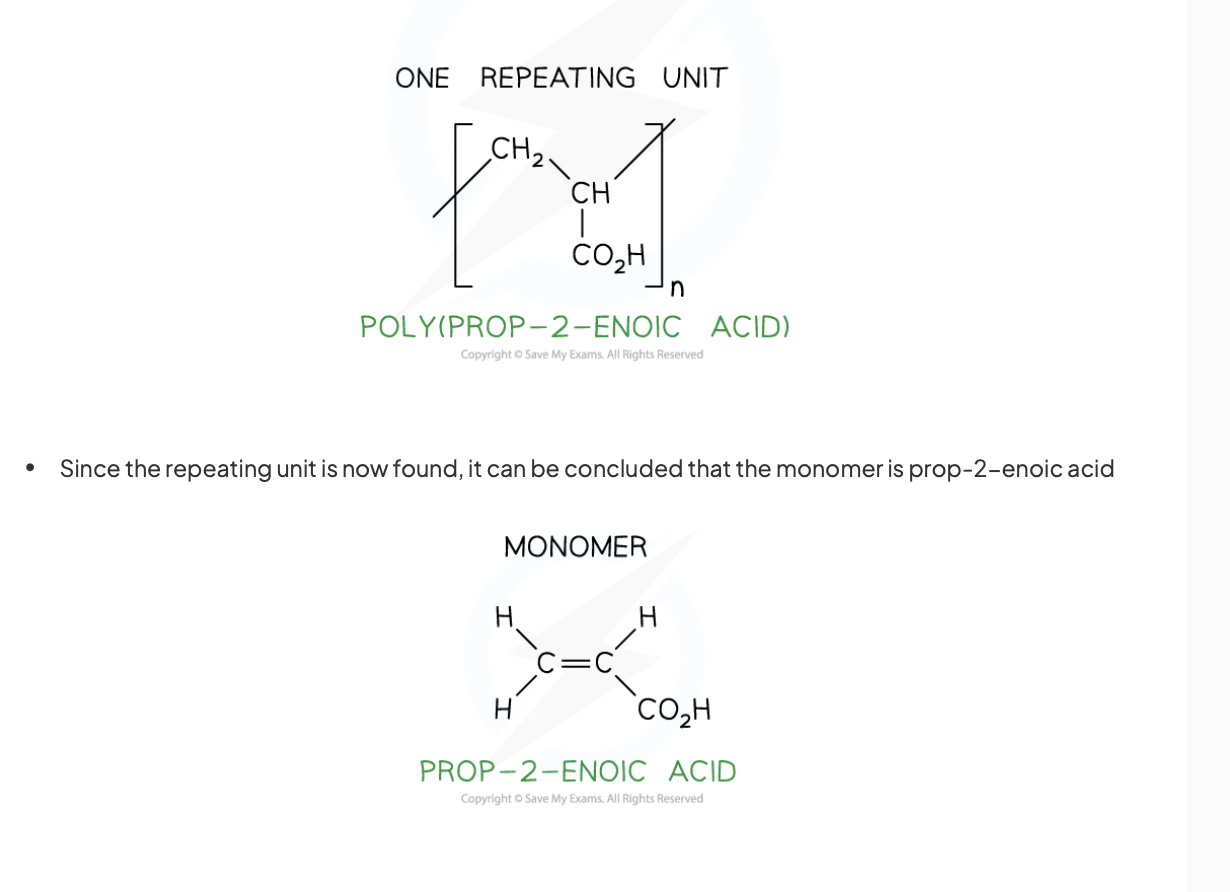
how many carbons does a repeating unit have in the main carbon polymer chain
2
why is the disposal of polymers a challenge
due to their:
unreactivity
non-biodegradability
harmful combustion products when burnt
name 2 types of recycling to help reduce pollution by plastics
mechanical recycling
feedstock recycling
how does mechanical recycling act as a solution to pollution by plastics
different types of plastic are separated and washed thoroughly
ground up into small pellets
pellets are heated until they melt, then remoulded so that they can be used again
all without breaking down its polymer structure
primary method for processing plastic waste
how does feedstock recycling act as a solution to pollution by plastics
Heating the plastic to a high enough temperature that the polymer bonds break, and monomers are formed
The monomers formed are then used to produce new plastics
describe an issue with these methods of recycling plastics
Some plastics, like poly(propene), can only be heated and reused a number of times
Each time the plastic is heated, some of the key chains break
This means that over time, the properties of the plastic are degraded