chemistry
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
what is an objective in a lab report?
Objectives outline the general framework of the research direction and define what the researcher aims to achieve or explain.
what is a hypothesis in a lab report?
Hypotheses, on the other hand, represent the expectations or preliminary answers that the researcher formulates based on these objectives, to be verified later through experimental or statistical means.

what is Ea?
Activation energy the amount of energy difference between the reactant and the height of the activation complex

What is EPac?
Potential of the activated complex from zero to the height of the activation complex

what is EPr and HR or EPP snd HP
EPR id the potential energy of the reactants
HR is the potential energy of the reactants
EPP id the potential energy of the products from zero to the the hightest value of the products
HP is the potential energy of the products from zero
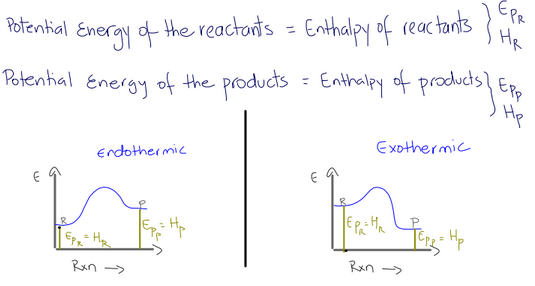
what is Denla H
the difference between the height of the recants and products
what is specific heat capacity
the amount(quantity) of heat involved when you raised a substance’s temp by 1 degree C
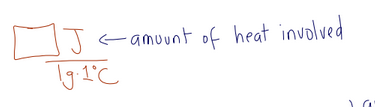
what is the specific heat capacity of water
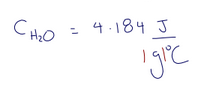
what are the two types of dissolution?
exothermic and endothermic reactions
what are the properties of gases
expansion with increased pressure and
Contraction when temp goes down volume decreases
Compression when volume does down pressure increases
how much is 1 atm
101.3 kpa or 760 mm Hg
What is considered room temperature
25 C
What is stp
standard temperature and standard temperature 0 C and 101.3 kpa
what is the conversion formula for converting atm to kpa
KPA= (101.3 KPA)(x mmg)
_________________________
160 KPa
what is the converson from kelvin(K)
0 C is 273 K
what is knetic energy of gas particles
kenetic energy(Ek) is related to the temperture of the gas
2 gas particles move freely to another space avalible.
when the space increases gas spreads
the force of attraction between gas particles is not strong enough to give gas a defined shape
gas particles are ina a continious rapid motions
they vibrate, translate and rotate
the collide constantly with one another and the walls of the container
what is avogadro’s hypothesis
if gasses have the same volume and pressure they will contain the same amout of mols
true or false pressure and volume have an inverse relationship
true pressure and volume have an inverse relationship
what is the formula for pressure
p(pressure)=F (force)
______________________
Area
what is the units of area
m²
what unit is force
Newtin (N)
what is Boyle’s law
(P1)(V1)=(P2)(V2)
P1=(V2)(P2)
_____
V1
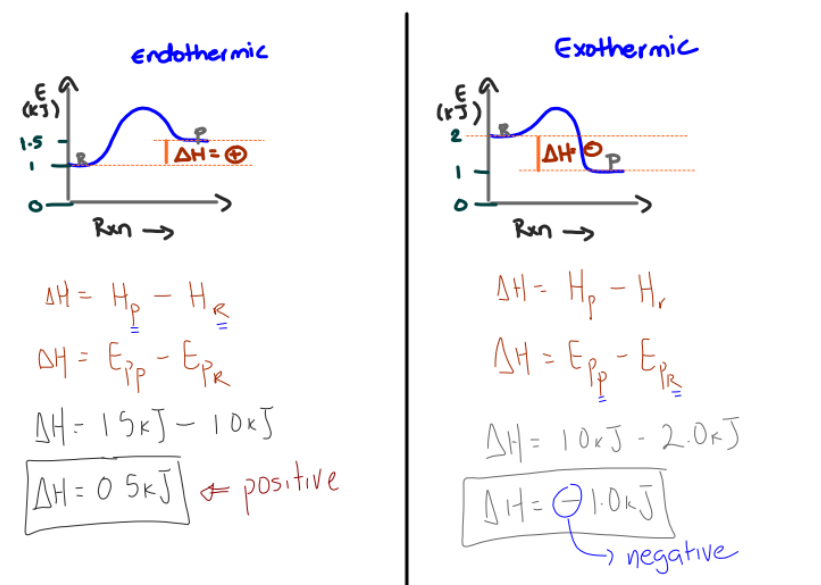
What is the empathy change formula
Delta H= EPP-EpR
Hp is the rmpathy of the product
HR is empathy of the reactants
What are the characteristics of an endo and exo thermic reaction just by looking at the reaction
For endo
the value of delta H is positive
Heat is absorbed with the reactants
For exo
The calue of delta H is negative
Reactants release the heat
What is guy- lussac’s law
Pi P2
— = —
T1 T2
Temp is only in Kelvin
Preasure have to be the same unit either kpk or atm
What is the general gas law
P1V1. P2V2
—- = ——
T1. T2
How do you solve for general gas law
T1= p1 v1 T2
———
P2 V2
what is dalton’s law
Px Nx
__ =__
P total N total
it’s for a mix gas in the same container
how to solve
P(pressure of partial)=(partiame number of mols)(Total pressure)
=========================
Total mols
what are the conditions you need for charle’s law
for charle’s law pressure and amount of mols must be the same
temp in kelvim
if Keq is greater than 1 is the products or reactants favored?
products
what is avagadro’s hypothesis
if the question states all the gases have the same pressure they have equal volume
what is ideal gas law
P(V)=n(R=(8.31 Kpa)(Volume in L))(T)
(mol(Kelvin )
to solve for V or P divide the rest of the formula with it
What is effects chemical rection
Nature of the reactant
Concentration of a reactant
what is a dynamic equilibrium
a closed system(closed container where nothing can enter or exit
a continuation
when asking for ksp is the reactant a aquious, solid or a liquid
solid