Enzymes, Vitamins, and Proteins: Key Concepts and Classifications
1/177
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
178 Terms
What are proteins primarily composed of?
Chains of amino acids.
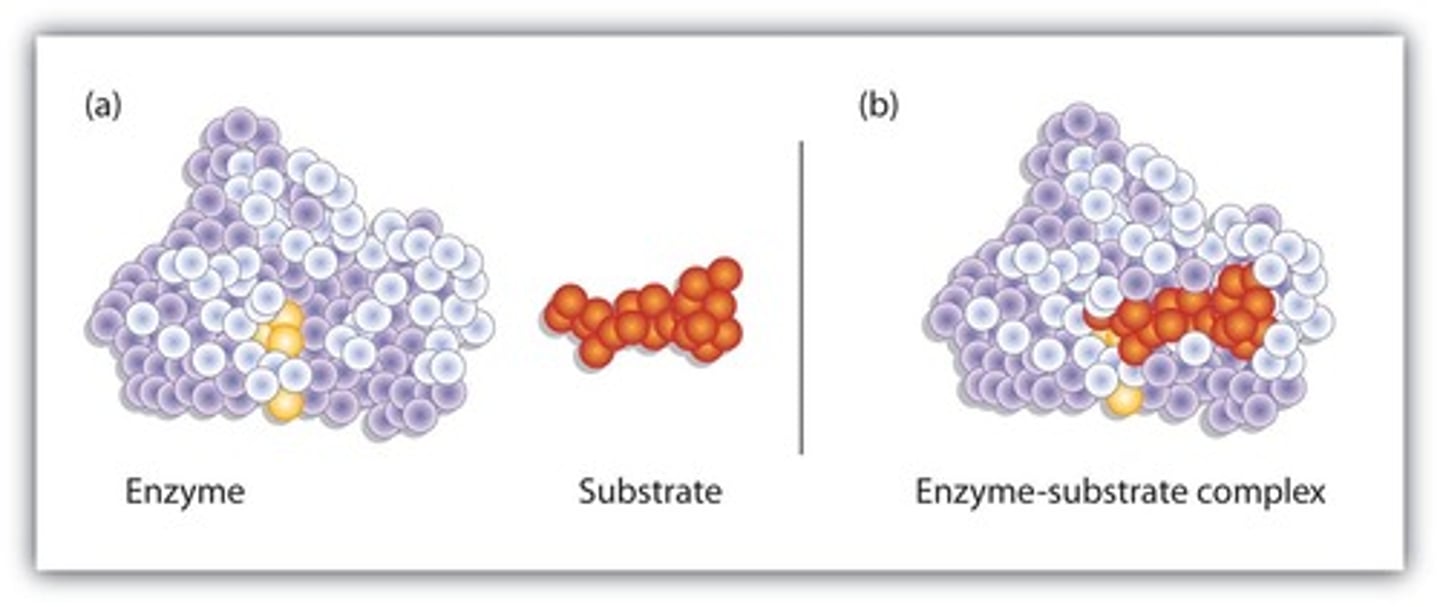
What is the mass range of proteins?
From several thousand to several million daltons (Da).
What elements do all proteins contain?
Carbon, hydrogen, oxygen, nitrogen, and sulfur.
What is an enzyme?
A biological catalyst that increases the rate of a chemical reaction without being changed or consumed.
What is the active site of an enzyme?
The specific region where a substrate binds to an enzyme.
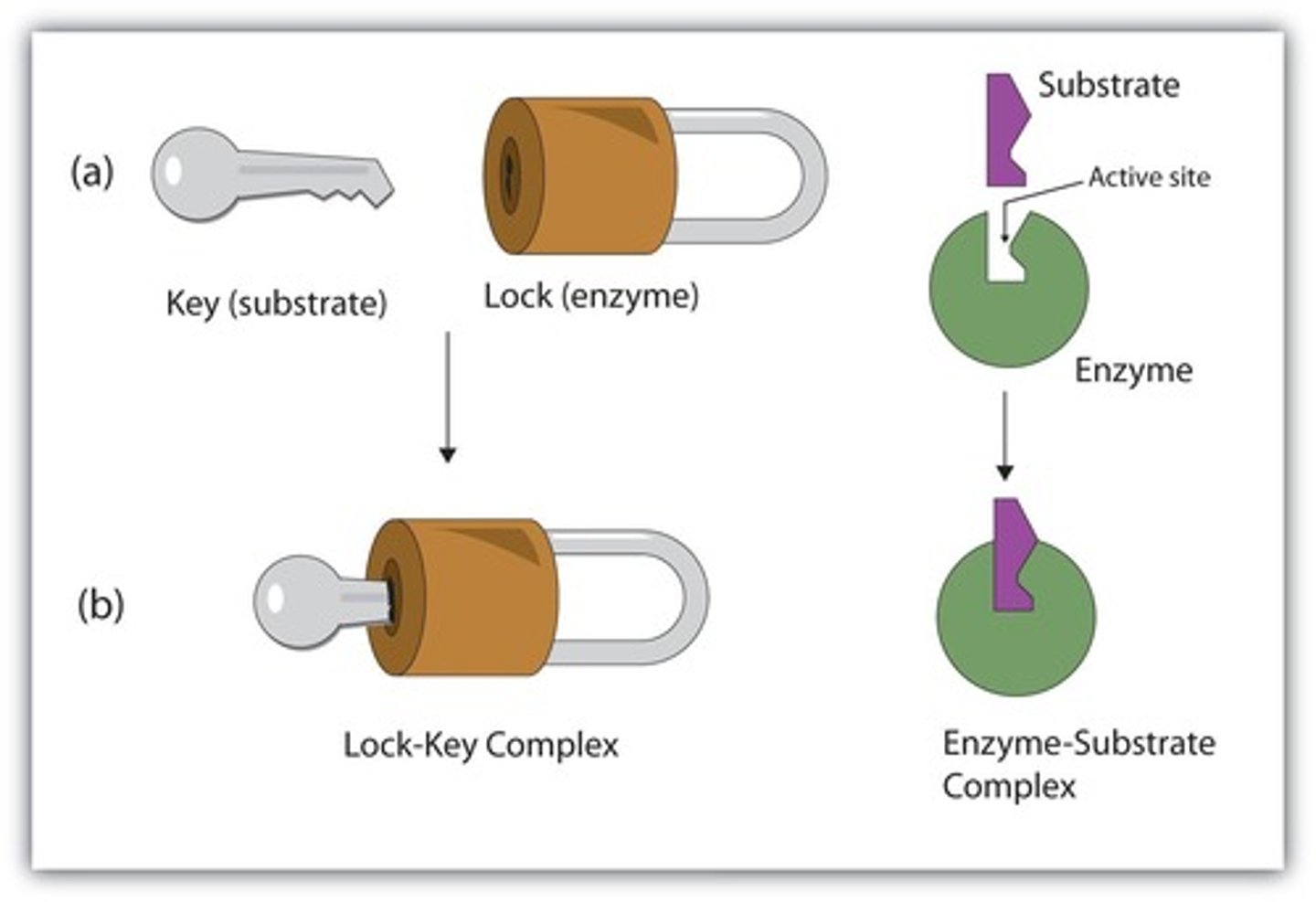
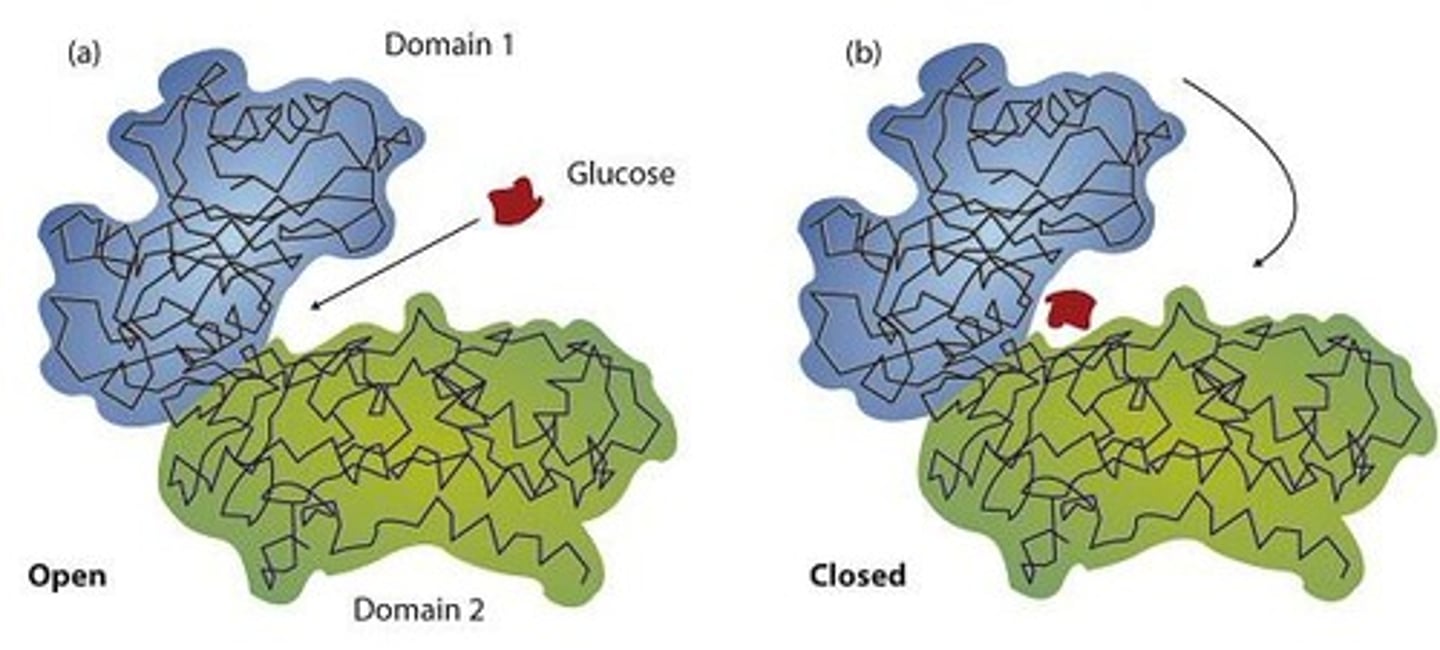
What model describes the conformational change of an enzyme upon substrate binding?
The induced-fit model.
How does substrate concentration affect enzyme activity initially?
An increase in substrate concentration increases the reaction rate.
What happens to enzyme activity at high substrate concentrations?
The reaction rate levels off as the enzyme becomes saturated.
How does temperature affect enzyme activity?
At low temperatures, an increase in temperature increases activity; at high temperatures, the enzyme may denature.
What are the two categories of enzyme inhibitors?
Irreversible inhibitors and reversible inhibitors.
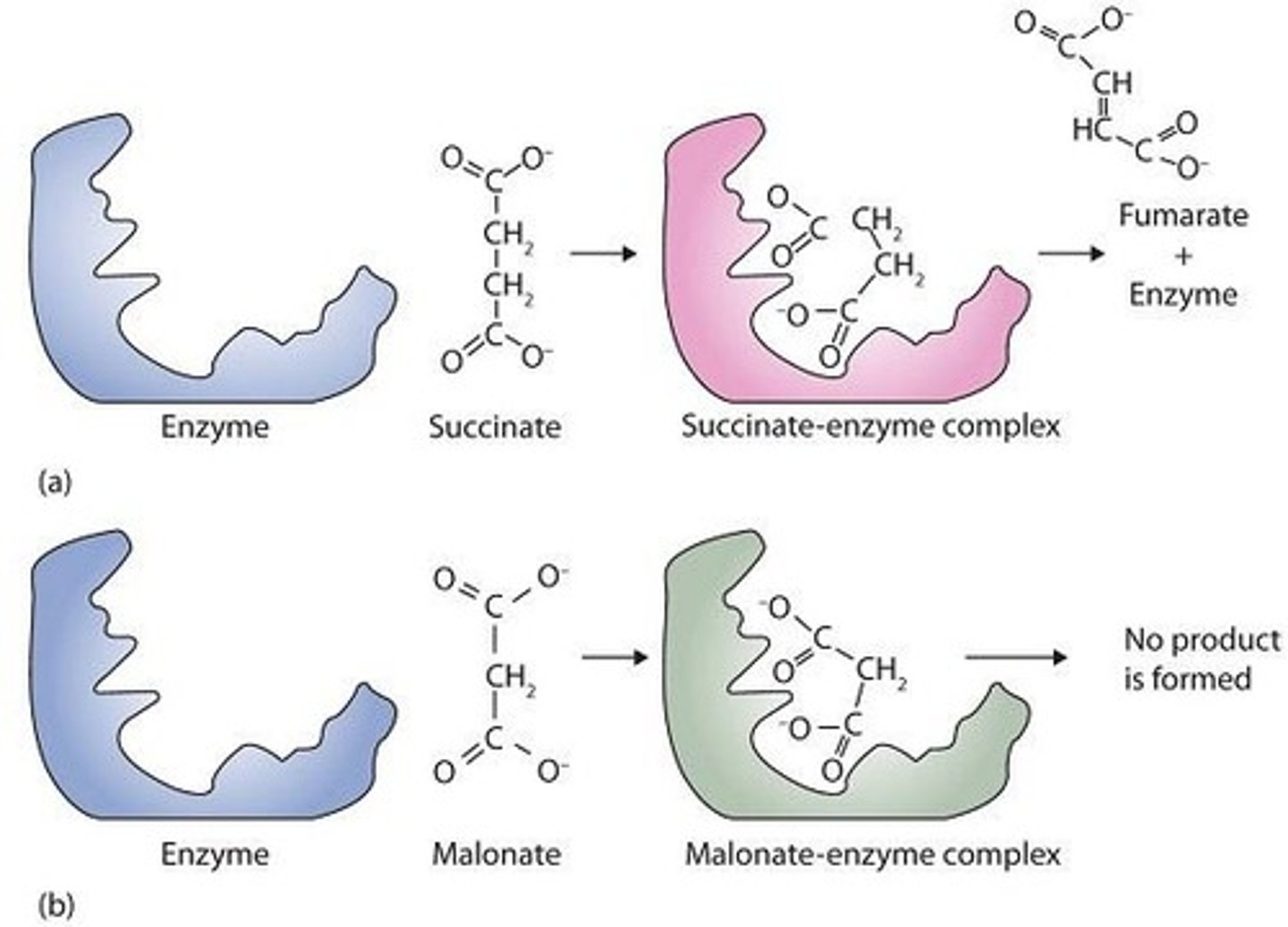
What is a competitive inhibitor?
An inhibitor that competes with the substrate for binding at the active site.
What is a noncompetitive inhibitor?
An inhibitor that binds at a site distinct from the active site.
What are vitamins?
Organic compounds essential in very small amounts for normal metabolism.
What are the two broad categories of vitamins?
Fat-soluble vitamins and water-soluble vitamins.
What role do water-soluble vitamins play in enzyme function?
They are needed for the formation of coenzymes, which are required for some enzymes' catalytic activity.
What is the significance of enzyme specificity?
Each enzyme catalyzes only one type of reaction in only one compound or a group of structurally related compounds.
What is the systematic naming convention for enzymes?
Enzymes are assigned a four-digit number and a name ending in -ase based on their substrate.
What does the first digit in an enzyme classification number indicate?
The type of reaction catalyzed (e.g., oxidoreductase).
What does the second digit in an enzyme classification number indicate?
The specific type of substrate involved in the reaction.
What does the third digit in an enzyme classification number indicate?
The coenzyme required for the reaction.
What does the fourth digit in an enzyme classification number indicate?
The order in which the enzyme was isolated and characterized.
What is the common name for alcohol:NAD oxidoreductase?
Alcohol dehydrogenase.
What is the role of lipases?
They catalyze the hydrolysis of lipids.
What is the role of proteases?
They catalyze the hydrolysis of proteins.
What do isomerases do?
They catalyze reactions that convert a compound to its isomer.
What do ligases do?
They catalyze reactions that form new bonds between carbon and another atom.
What is the substrate in the reaction catalyzed by sucrase?
Sucrose.
What is the enzyme that catalyzes the hydrolysis of sucrose?
Sucrase.
What is the first step in an enzyme-catalyzed reaction?
The formation of the enzyme-substrate (E-S) complex.
What does the active site of an enzyme do?
It is the region where the substrate binds and is converted to product.
What model describes the enzyme as conformationally rigid?
The lock-and-key model.
What is the current theory that describes enzyme action involving conformational change?
The induced-fit model.
What type of interactions hold the enzyme and substrate together in the E-S complex?
Hydrogen bonding and other electrostatic interactions.
What is substrate specificity in enzymes?
The ability of an enzyme to act on a specific substrate or a group of related molecules.
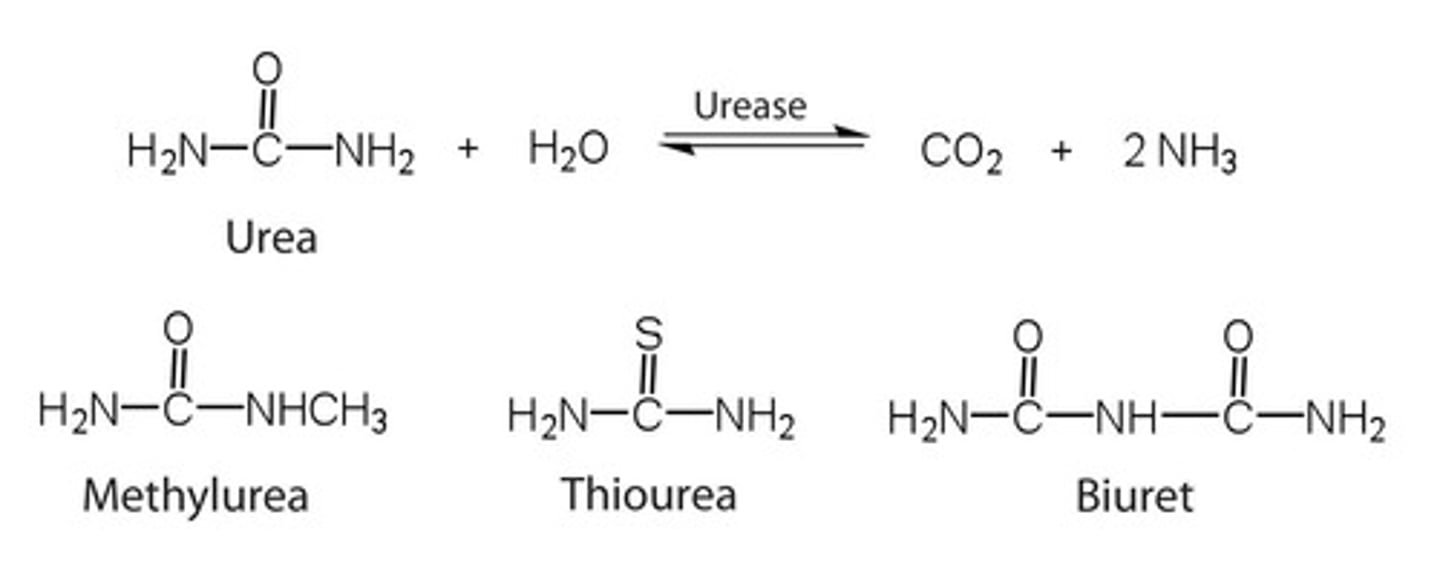
Which enzyme is more specific: urease or carboxypeptidase?
Urease, as it catalyzes the hydrolysis of a single substrate—urea.
What happens to the enzyme after catalysis?
The enzyme resumes its original structure.
What is the role of amino acid side chains in the active site?
They can act as acid or base catalysts and aid in the rearrangement of substrates.
What is the significance of the unique conformation of the active site?
It allows the enzyme to bind specifically to its substrate.
What does the term 'induced-fit' refer to in enzyme action?
The conformational change of the enzyme when the substrate binds.
What is the difference between the lock-and-key model and the induced-fit model?
The lock-and-key model suggests a rigid fit, while the induced-fit model allows for conformational changes upon substrate binding.
What type of enzyme is lactase?
A hydrolase that catalyzes the hydrolysis of lactose.
What is the substrate for cellulase?
Cellulose.
What type of enzyme is peptidase?
A protease that catalyzes the hydrolysis of peptides.
What does lipase catalyze?
The hydrolysis of lipids.
What is the function of amylase?
To catalyze the hydrolysis of starch into sugars.
What does maltase do?
Catalyzes the hydrolysis of maltose into glucose.
What type of enzyme is decarboxylase?
A lyase that removes a carboxyl group from a substrate.
What is the function of dehydrogenase?
To catalyze the removal of hydrogen from a substrate.
What is the role of hydrogen bonding in enzyme-substrate interactions?
It helps stabilize the E-S complex.
How do enzymes increase the rate of reactions?
By bringing reactants close together and aligning them properly.
What is the effect of enzyme specificity on cell chemistry?
It regulates reactions to occur in the proper place and time.
What happens to the enzyme after the product is released?
The enzyme is free to catalyze another reaction.
What is the significance of the spatial orientation of functional groups in the active site?
It ensures proper binding and catalysis of the substrate.
What does the lock-and-key model of enzyme activity suggest?
It suggests that an enzyme is rigid and can only bond to substrates that exactly fit its active site.
How does the induced fit model differ from the lock-and-key model?
The induced fit model portrays the enzyme as flexible and complementary to the substrate only after the substrate is bound.
Which enzyme has greater specificity: urease or carboxypeptidase?
Urease has greater specificity because it can bind only to a single substrate.
What type of interaction occurs between a COOH group and an enzyme's active site?
Hydrogen bonding.
What type of interaction occurs between an NH3+ group and an enzyme's active site?
Ionic bonding.
What type of interaction occurs between an OH group and an enzyme's active site?
Hydrogen bonding.
What type of interaction occurs between a CH(CH3)2 group and an enzyme's active site?
Dispersion forces.
Which amino acid might interact with a COOH group in an enzyme's active site?
Serine, which has a polar side chain capable of hydrogen bonding.
Which amino acid might interact with an NH3+ group in an enzyme's active site?
Aspartic acid, which has a negatively charged side chain.
Which amino acid might interact with an OH group in an enzyme's active site?
Asparagine, which has a polar side chain capable of hydrogen bonding.
Which amino acid might interact with a CH(CH3)2 group in an enzyme's active site?
Isoleucine, which has a nonpolar side chain.
What is the single most important property of enzymes?
The ability to increase the rates of reactions occurring in living organisms, known as catalytic activity.
How does substrate concentration affect enzyme activity?
The rate of an enzymatic reaction increases with substrate concentration until a limiting rate is reached, after which further increases do not significantly change the reaction rate.
What happens when enzyme active sites are saturated?
All enzyme active sites have substrate bound, and excess substrate cannot react until the bound substrate has reacted and been released.
How does enzyme concentration affect reaction rate?
The reaction rate increases as the concentration of the enzyme increases, especially when the enzyme concentration is lower than the substrate concentration.
What is the effect of a 10°C increase in temperature on reaction rate?
It approximately doubles the reaction rate for most chemical reactions, including enzymatic reactions, up to a certain point.
What happens to enzyme activity at high temperatures?
Enzyme activity decreases due to denaturation of the protein structure and disruption of the active site.
At what temperature range does denaturation of enzymes typically occur?
Between 45°C and 55°C.
What is the optimum pH range for most enzymes?
Between pH 6 and 8, although some enzymes like pepsin have optimum pH values outside this range.
What happens to enzymes at extreme pH levels?
Enzymes may be denatured, altering their catalytic activity.
What is the optimum pH for pepsin?
2.0, as it is active in the acidic environment of the stomach.
How do refrigeration and freezing affect enzyme activity?
They slow enzyme activity, which helps preserve food by inhibiting reactions that could lead to spoilage.
What is the relationship between temperature and metabolic processes in hibernating animals?
Lower body temperatures decrease metabolic rates to levels that can be maintained by stored energy.
What is the significance of monitoring substrate disappearance or product formation?
It allows measurement of enzyme activity and the rate of reaction.
What analogy is used to explain enzyme saturation?
The analogy of taxis (enzymes) waiting for passengers (substrates) illustrates how saturation occurs when all active sites are occupied.
What happens to the rate of an enzyme-catalyzed reaction when substrate concentration is low and doubled?
The rate of the reaction increases.
How does increasing enzyme concentration affect the rate of an enzyme-catalyzed reaction?
It increases the rate of the reaction, provided there is sufficient substrate.
What occurs to the rate of reaction as substrate concentration increases in enzyme-catalyzed reactions?
The rate initially increases but eventually levels off.
Why do enzymes become inactive at very high temperatures?
High temperatures denature the protein, decreasing the reaction rate.
What is the optimum pH for an enzyme with a pH of 7.4 if the pH drops to 6.3?
The activity will decrease due to increased acidity affecting the active site.
What happens to an enzyme's activity if the pH increases to 8.5 when its optimum is 7.2?
The activity will likely decrease due to unfavorable pH conditions.
What is an enzyme inhibitor?
A substance that decreases enzyme activity.
What is the difference between reversible and irreversible inhibitors?
Reversible inhibitors can dissociate from the enzyme, while irreversible inhibitors bond covalently and cannot be reversed.
What are competitive inhibitors?
Compounds that resemble a substrate and compete for binding at the active site of an enzyme.
How can competitive inhibition be overcome?
By increasing the concentration of the substrate.
What is an example of a competitive inhibitor?
Malonate, which inhibits succinate dehydrogenase.
What is irreversible inhibition?
Inactivation of an enzyme through covalent bonding with a specific group at the active site.
What is the role of diisopropylfluoro-phosphate (DIFP)?
It irreversibly inhibits acetylcholinesterase by binding to a serine group.
What is a classic example of a reversible inhibitor?
Competitive inhibitors like malonate that can be displaced by substrate.
What is the significance of enzyme-substrate complexes?
They are formed when substrate binds to an enzyme, facilitating the reaction.
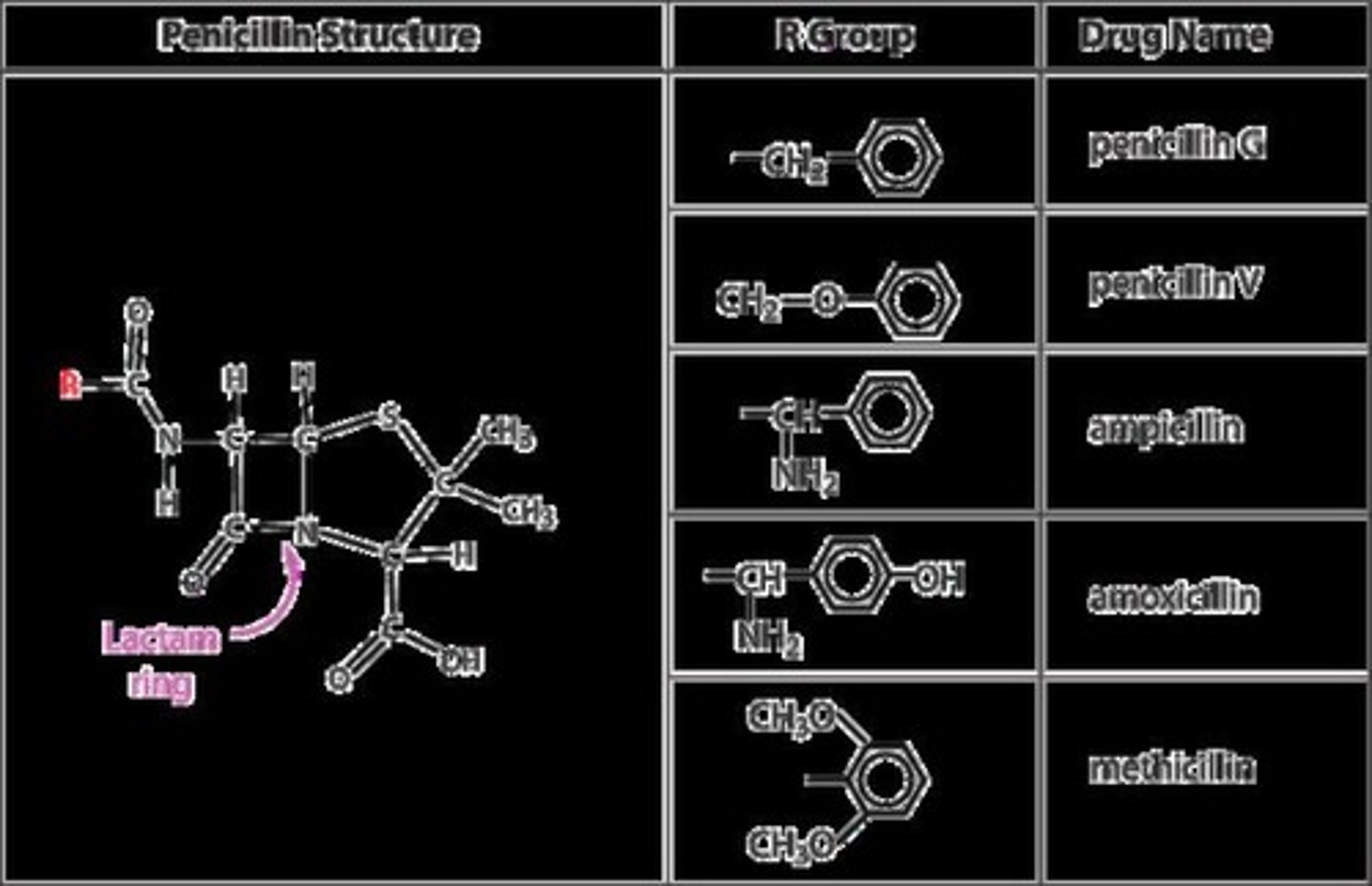
What is the effect of antibiotics like penicillin on bacteria?
They inhibit critical enzymes necessary for bacterial growth.
Who discovered penicillin and when?
Alexander Fleming in 1928.
What was crucial for the large-scale production of penicillin?
Development of a corn-based nutrient medium and a higher-yielding strain of mold.
What are poisons in the context of enzyme inhibition?
Compounds that bind covalently to enzymes, inactivating them.
What is the effect of substrate concentration on enzyme activity when it is in excess?
All enzyme binding sites will be occupied, and further increases in substrate concentration cannot increase the rate.
How do noncompetitive inhibitors work?
They bind to an enzyme at a site other than the active site, reducing enzyme activity regardless of substrate concentration.
What is the relationship between enzyme activity and temperature?
Activity increases with temperature up to a point, after which it decreases due to denaturation.