The Rate Constant
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
How do we measure rate of reaction?
We need to measure how quickly reactants are used up or how quickly products are formed
We can then find the change in concentration/time
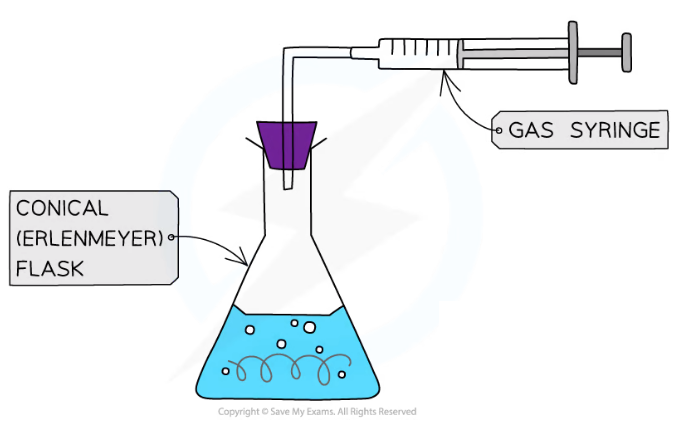
Diagram for gas collection
Conical flask
Gas syringe
Delivery tube
The gas syringe needs to have a scale
How is rate calculated?
Change in Concentration
Time
How could we calculate the change in concentration of a reactant from the change in volume of a product (HCl and H2 for example) at different time intervals?
Find the number of moles of hydrogen gas through the molar gas constant equation (PV=nRT) at the first time interval like 10 seconds
Then from this, find the molar ratio of hydrogen gas to HCl- we know how many moles of HCl reacts fully with hydrogen gas
Take away the moles of HCl that were used up from the number of moles at the start to find the moles of HCl left over
Divide this by the total volume to find the concentration
For each time interval the same thing will always be done (always use the starting amount of moles of HCl to find the number of moles left over)
What does a decreasing gradient concentration against time graph show?
The rate of reaction getting slower
What are some ways we can measure rate of reaction experimentally?
Collection of gas (Only works for reactions where a gas is produced)
Mass loss (Only works for reactions where a gas is produced)
Change in pH
Colorimetry
How can we measure rate of reaction from mass loss?
The mass of a container can be measures at frequent intervals like every 10 seconds
The mass lost is equivalent to the mass of gas produced which can be used to work out rate of reaction
How can change in pH be used to determine rate of reaction?
pH can be converted into concentration of H+ ions which can then be used to calc (short for calculate btw im just using slang guys) rate of reaction
How can colorimetry be used to calculated rate of reaction
The change in the intensity of a coloured pigment could be used to monitor concentration change and then calculate rate
What does a first order reaction mean?
The change in rate of a reaction is directly proportional to the change in concentration of one reactant
What does a first, second and zero order reaction graph look like?
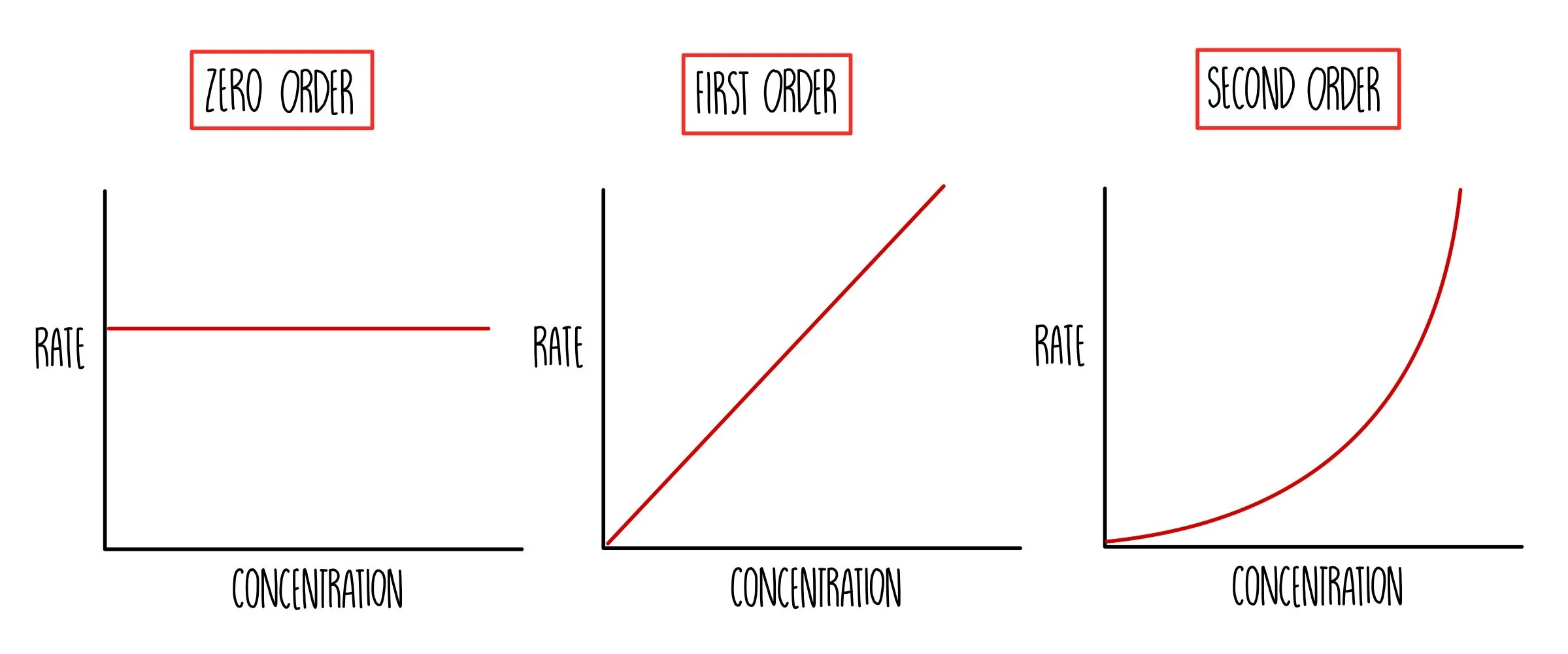
What does a second order reaction mean?
The increase in rate of a reaction is the square of the increase in concentration of a reactant
What does a zero order reaction mean?
The increase in rate of reaction is not affected by the increase in concentration of a reactant
What do we use in rate equations to show a proportionality contant
k
When do we not have to add a reactant to a rate equation?
When the reaction is zero order with respect to that reactant
How do we find units for the rate constant?
You need to cancel out the units in the rate equation:
Units of concentration- moldm-3
Units of time- s
Units of rate- moldm-3s-1
When calculating the units, s-1 will never be cancelled out
How can we find k if we are given the initial rate of reaction and the initial concentration of reactants?
Rearrange the rate equation to find k
Puts the values in making sure to add powers if you have any reactions that are above first order
How can we find the overall order of a reaction?
Add all the orders together
If we have calculated k for a reaction, how can we calculate initial rate for a reaction under the same conditions but with the concentration of reactants doubled?
From the orders of reaction of reactants, find the effect on the rate of reaction if their concentration is doubled
Multiply these rates together to find the overall increase in rate
Do the initial rate of the first reaction multiplied by this number to find the new initial rate
How can we find orders of reaction using kinetics experiments?
If we perform several experiments where we keep one reactant constant and vary the concentration of another
We can then find the order of reaction by comparing the change in concentration of that reactant with the change in rate of reaction
If we have experiments where reactant A changes while reactants B stays constant, whereas B has no experiments where A stays constant, we can find how A’s change should affect the rate independently of B, then find B’s affect on the rate and therefore its order
What is the rate determining step?
Reaction can occur in multiple steps- the products of one step are the starting materials for the next
The slowest step is called the rate determining step
In a chemical reaction, any species after the rate determining step does not determine the rate and therefore will not appear in the rate equation
What is the Arrhenius equation and what are it’s units?
k=Ae-Ea/RT
k= rate constant
A= Arrhenius constant
e= exponential
Ea= Activation energy (J mol-1)
R= Gas constant (8.31 JK-1mol-1)
T= Temperature (K)
How can the relationship between activation energy and the rate constant be shown through the Arrhenius equation?
If we take just the part of the Arrhenius equation with e-Ea/RT, we can keep the temperature constant but increase the activation energy
If we do this, as Ea increases, k decreases
Maxwell Boltzmann distribution curves show us that at higher activation energies, there are fewer particles with E>Ea which can react
How can the relationship between temperature and the rate constant be shown through the Arrhenius equation?
If we take just the part of the Arrhenius equation with e-Ea/RT, we can keep the activation energy constant but increase temperature
As temperature increases, k increases
In a Maxwell Boltzmann distribution, as temperature increases, the number of successful collisions increase as particles gain KE and move faster and a greater proportion of particles with E>Ea
Temperature has an exponential relationship with k
How can we find the mechanism for a reaction through the rate equation?
If there is a reactant (reactant A) that doesn’t appear in the rate equation but another reactant (reactant B) does appear, A must not be part of the rate determining step but B must be
We can conclude from this that reactant B will form an intermediate as part of the rate determining step before reacting with A to form the product of the reaction
You can do kinetics experiments to find the mechanism for a reaction
How can you rearrange the Arrhenius equation to find Ea?
Ea= RT(ln(A)-ln(k))
Explanation
k=Ae-Ea/RT
Take the natural log of both sides
lnk=lnA-ln(e)-Ea/RT
Natural log and e cancel each other out so we are left with=
lnk - lnA - Ea/RT
We can then just rearrange like normal to find Ea
Rearrange the Arrhenius equation to find T
Ea / (lnA - lnk)R
Rearrange Arrhenius equation to find A
A= k/e-Ea/RT
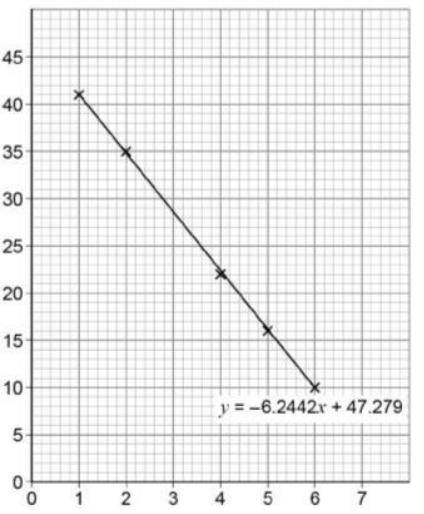
How do we plot an Arrhhenius plot graph?
It is plotted the same as a straight line- y=mx+c
On the y axis we plot the natural log of k
On the x axis we plot 1/T
How can we use Arrhenius plots to find activation energy?
Ea = -gradient X R (Gas constant)
We can use our straight line to find the gradient and multiply by 8.31 to find the activation energy
If a solid is added in large excess to a reactant, why would the rate only be effect by the concentration of the reactant?
The surface area of the solid remains effectively constant so it’s not affecting the rate
What does the graph look like for the change in volume of a reactant that is zero order?
The gradient of the line is constant
The concentration decreasing as the reaction goes on is having no effect on the rate
What can we tell the order of rection is from the units for k being s-1?
First order
This is because the units must have cancelled out in the rate equation so there was only one term with no powers