Properties of fluids
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Property
Any characteristic of a system
ome familiar properties are pressure P, temperature T, volume V, and mass m. Properties are either intensive or extensive.
Sebarang ciri bagi sesuatu sistem
Beberapa sifat yang biasa ialah tekanan (P), suhu (T), isipadu (V), dan jisim (m)
Sifat-sifat ini sama ada bersifat intensif atau ekstensif
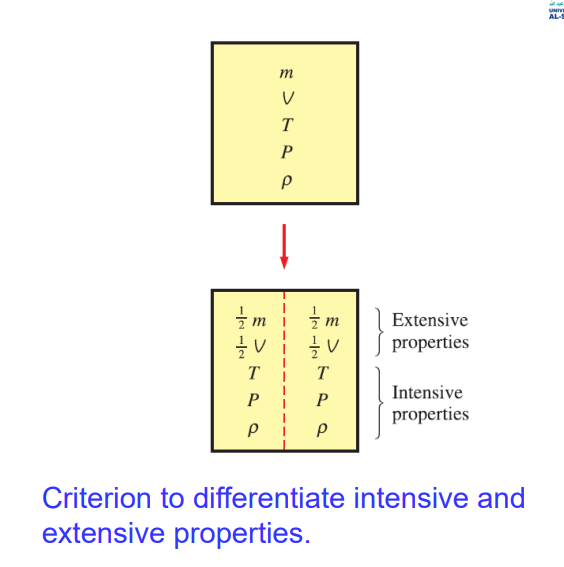
Intensive properties
Those that are independent of the mass of the system, such as temperature, pressure, and density.
Yang tidak bergantung kepada jisim sistem, seperti suhu, tekanan, dan ketumpatan.
Extensive properties
those whose values depend on the size—or extent—of the system. Total mass, total volume V, and total momentum are some examples of extensive properties.
Extensive properties per unit mass are called specific properties. Some examples of specific properties are specific volume (v = V/m).
Sifat yang bergantung pada saiz atau jumlah sistem
Contoh: jumlah jisim, jumlah isipadu (V), dan jumlah momentum
Jika sifat ekstensif dibahagi dengan jisim → dipanggil sifat tentu (specific properties)
Contoh: isipadu tentu, v=V/mv = V/mv=V/m
Continuum
Matter is made up of atoms that are widely spaced in the gas phase
It is convenient to ignore the atomic nature and treat it as continuous, homogeneous matter (a continuum)
The continuum idealization allows properties to be treated as point functions
Properties vary continuously in space with no sudden jumps
This idealization is valid when the system size is large compared to the space between molecules
This applies to almost all practical problems
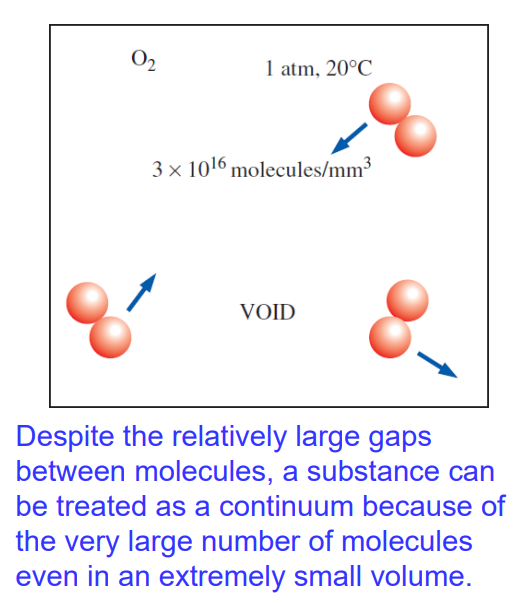
Jirim terdiri daripada atom yang berjauhan antara satu sama lain dalam fasa gas
Namun, lebih mudah untuk mengabaikan sifat atom dan menganggapnya sebagai bahan berterusan dan homogen (kontinuum)
Pendekatan kontinuum membolehkan sifat dianggap sebagai fungsi titik
Sifat berubah secara berterusan dalam ruang tanpa perubahan mendadak
Pendekatan ini sah apabila saiz sistem jauh lebih besar berbanding jarak antara molekul
Ini berlaku dalam hampir semua masalah praktikal
(Caveman explanation):
Gas = tiny atoms, far apart
But brain say: “too messy, ignore atoms”
Treat it like smooth stuff, no holes → continuum
Means we can look at one point and say its property (pressure, temp)
No sudden jump, everything change smoothly
Works because system BIG, atoms SMALL
Density
mass per unit volume
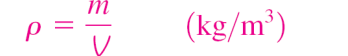
Specific volume
volume per unit mass
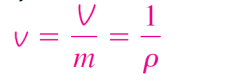
Specific gravity
can be defined as the ratio of the density of a substance to the density of some standard substance at a specified temperature
(usually water at 4°C, for which 𝜌H2O = 1000 kg/m3).
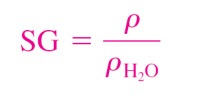
Density of Ideal Gases
The densities of liquids are essentially constant, and thus they can often be approximated as being incompressible substances during most processes without sacrificing much in accuracy
How about density of gases???
Equation of state: Any equation that relates the pressure, temperature, and density (or specific volume) of a substance.
Ideal-gas equation of state: The simplest and best-known equation of state for substances in the gas phase

Ideal gas law
An ideal gas is a hypothetical substance that obeys the relation Pv=RTPv = RTPv=RT
The ideal-gas relation closely approximates real gases at low densities
At low pressure and high temperature, gas density decreases and behaves like an ideal gas
Many common gases (air, nitrogen, oxygen, hydrogen, helium, argon, neon, krypton, carbon dioxide) can be treated as ideal gases with negligible error
Dense gases like water vapor in steam power plants and refrigerant vapor should NOT be treated as ideal gases
This is because they usually exist near saturation conditions
(Caveman explanation):
Ideal gas = “perfect gas” (not real, just idea)
It follows simple rule: push, volume, temp all nicely related
Real gas sometimes messy, but…
Low pressure + high temperature → gas spread out → act like ideal gas
Many gases (air, oxygen, nitrogen) → behave like ideal gas → easy use
BUT… thick/dense gas (steam, refrigerant) → too crowded
When crowded → no longer “perfect” → cannot use ideal gas rule
Viscosity
A property that represents the internal resistance of a fluid to motion or the “fluidity”.
Sifat yang mewakili rintangan dalaman sesuatu bendalir terhadap pergerakan atau “kelikatan”.
Drag force
The force a flowing fluid exerts on a body in the flow direction. The magnitude of this force depends, in part, on viscosity.
Daya yang dikenakan oleh bendalir yang mengalir ke atas suatu objek dalam arah aliran. Magnitud daya ini bergantung, sebahagiannya, pada kelikatan.
A fluid moving relative to a body exerts a drag force on the body, partly because of friction caused by viscosity.
