chemistry y9 EOY
1/163
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
164 Terms
define a mixture
two or more elements not chemically bonded. because of this they can be separated using separation techniques.
what is filtration?
a process which separates insoluble solutes in a solution.
method for filtration

what is the solute in filtration
the insoluble solid being separated from a liquid, often referred to as the residue
what is the name if the solution that passes through the filter paper in filtration?
the filtrate
what is crystallisation?
the process to produce crystals of the solute from the filtrate after filtration.
how does crystallisation work?
when you leave a saturated( more solute particles than solvent particles) solution to cool
as the saturated solution cools the solute becomes less soluble in the solvent and some comes out of the solution as a solid crystal.
what is the solvent in filtration
the liquid component of a mixture that passes through the filter to become the filtrate, carrying with it any dissolved substances (solutes) while leaving behind insoluble solids.
what is distillation?
to separate two or more liquids. it can also be used to separate a solute from a solution.
how does distillation work?

what is fractional distilllation?
Fractional distillation of crude oil is the process of heating crude oil and separating it into different useful products (fractions) like gasoline, diesel, and bitumen, based on their different boiling points in a tall fractionating column that's hot at the bottom and cool at the top.
where is chromatography used?
it is used in water soluble mixtures such as inks, dyes/paints and colouring agents.
chromatography diagram
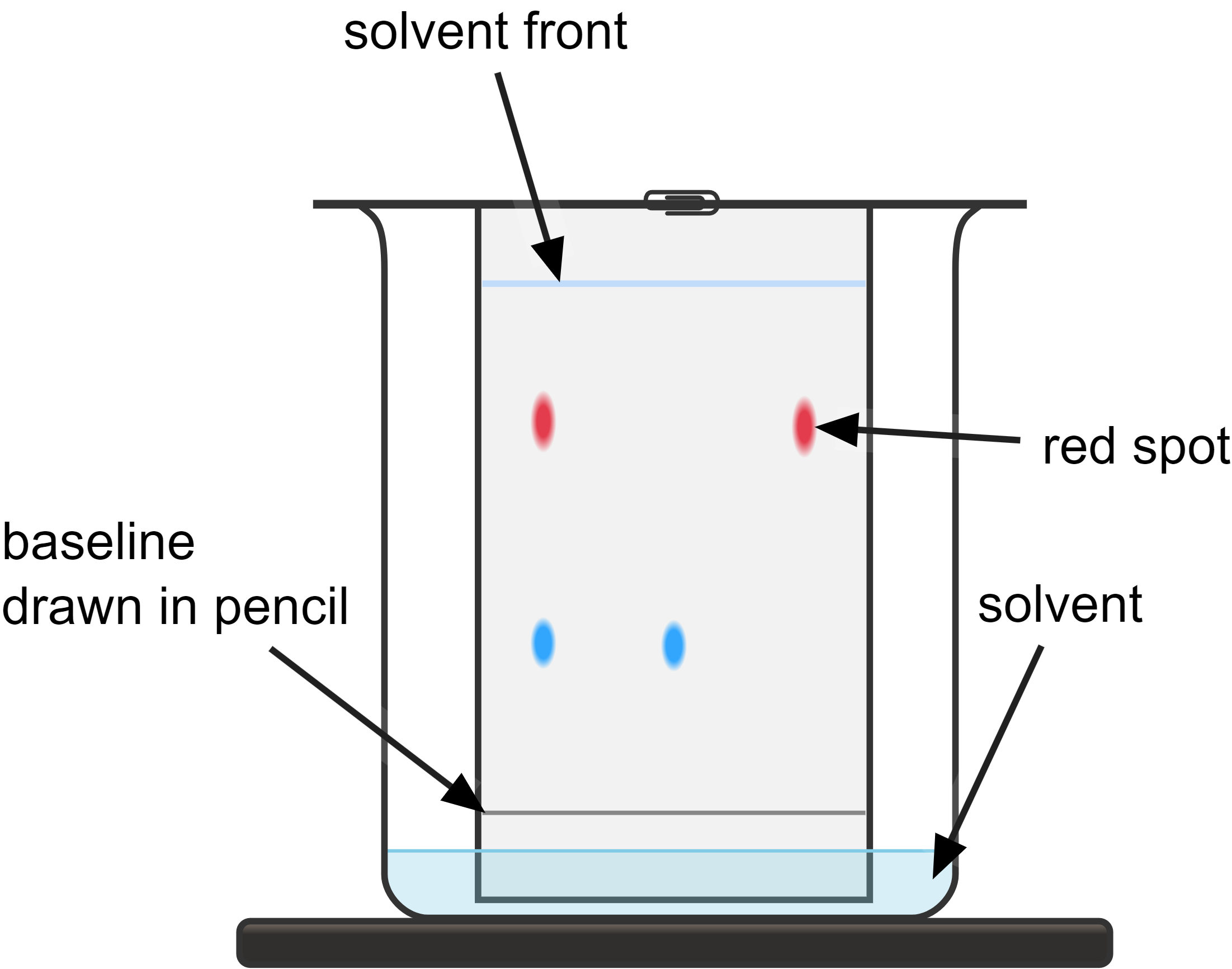
what is the solvent front in chromatography?
the line where the solvent travelled to
what is an RF value in chromatography?
the retention factor is used to identify an unknown dye. it can be compared against known substances as they will have the same RF value.
what is the equation for working out the RF value in chromatography?
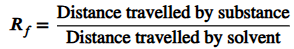
what is thin layer chromatography (TLC)
works in the same way as chromatography but the stationary phase (the chromatography paper) is a thin layer of silica or aluminium powder spread over a plate of glass or plastic.

what are the two stages in paper (regular) chromatography?
stationary phase= the phase that does not move. this is the chromatography paper.
mobile phase= the phase that moves. this is the solvent.
why do different substances have different melting points?
substances that have strong bonds between the atoms require higher temperatures to break the bonds, hence have a higher melting point.
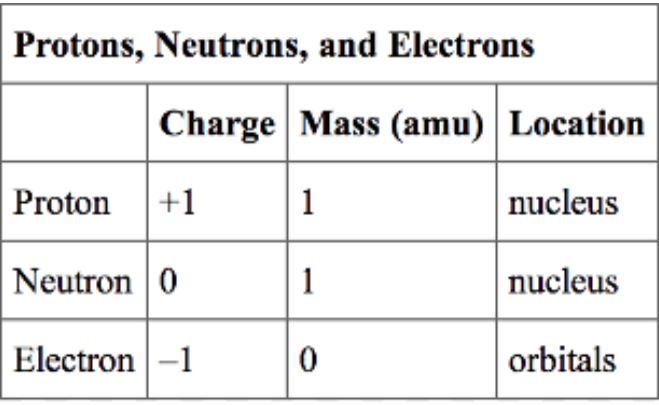
what are the weights and charges of protons, neutrons and electrons?

john daltons atomic model, 1803
suggested substances were made up of atoms that were tiny, hard spheres
atoms were “uncuffable” and indestructable (could not be split or divided)
each element had its own atoms that differed from each other in their mass- different sized spheres made up different elements


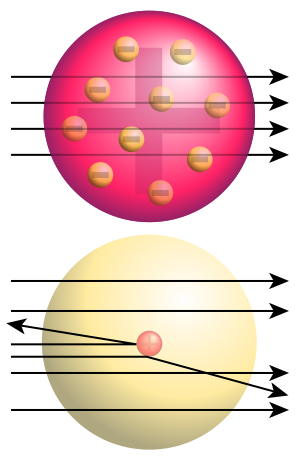
JJ thompson atomic model, 1897🍮
jj thompson discovered the electron (negative particles)
his model was a sphere of positive charge with negative electrons- atoms carry no overall charge
“plum pudding model”


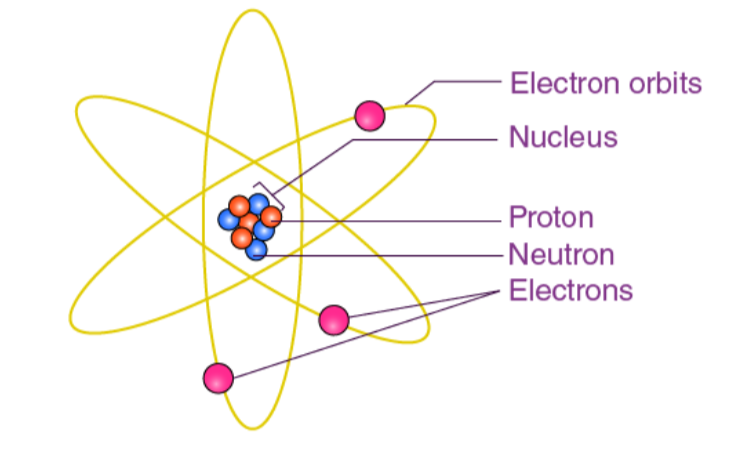
earnest rutherford atomic model, 1908 ☁
“nuclear model”
discovered the nucleus
the idea that each atom has a small, positive nucleus surrounded by a “cloud” of electrons ☁

describe the experiment carried out by geiger and marsden under direction of earnest rutherford and its results
Experiment: Ernest Rutherford and his student conducted the alpha particle scattering experiments where they fired positively charged alpha particles at an extremely thin sheet of gold.
Results: Rather than all of the alpha particles passing straight through the sheet of gold as you would expect from the plum pudding model, some were deflected to the side, and a small number were even deflected backwards.

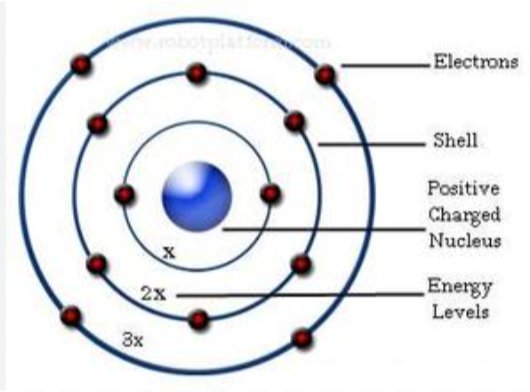
niels bohr atomic model, 1913
built onto the concept that the mass is mostly in the nucleus
theorised that elecrons orbit the nucleus, and are a fixed distance from the nucleus (shells)
contrast to rutherford`s theory which was that electrons were spread in a “general cloud” around the nucleus

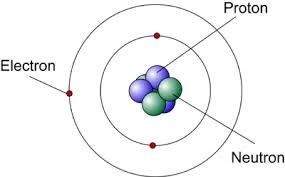
james chadwick atomic model, 1932
discovered neutrons in the nucleus
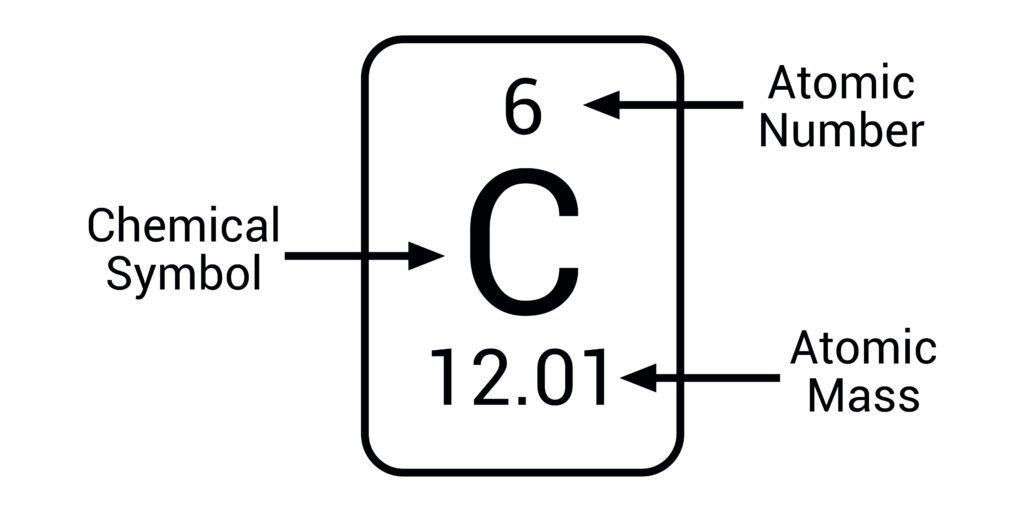
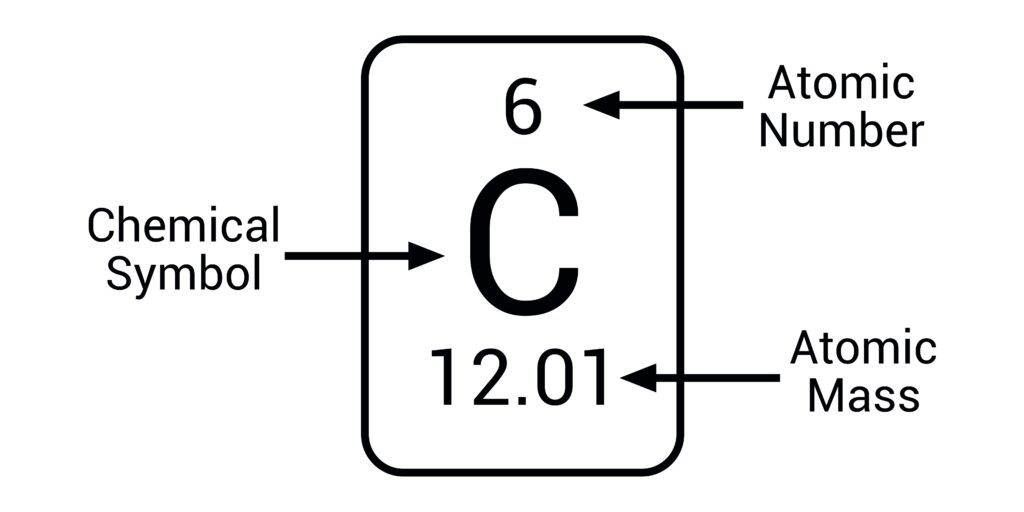
atomic number=
number of protons and the number of electrons (the same)
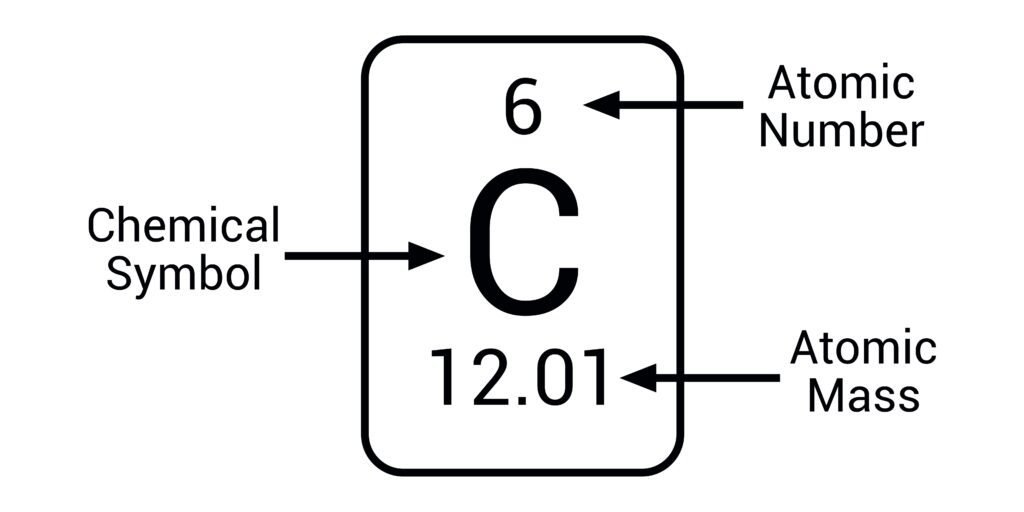
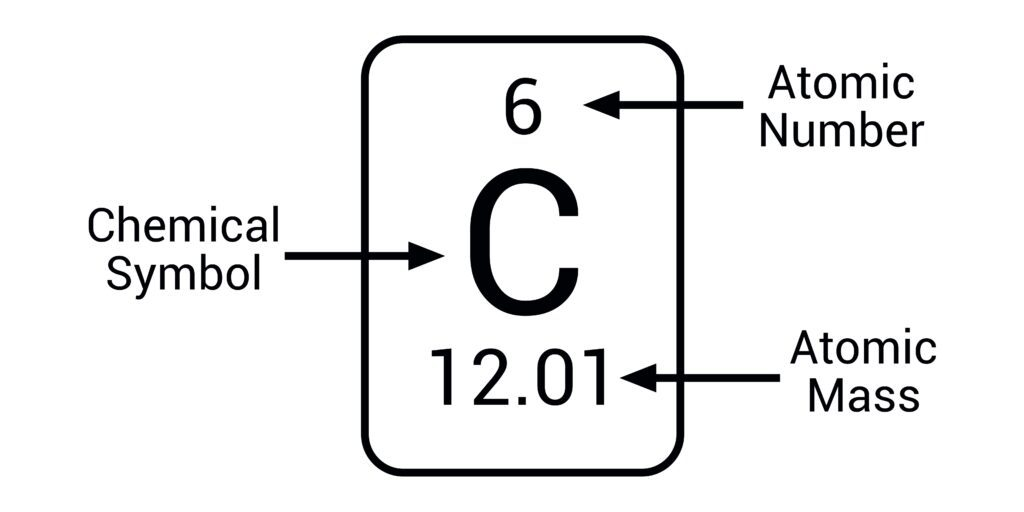
mass number/ relative atomic mass=
total number of protons& neutrons (combined)
what is an ion
charged atom, created when an atom gains or loses electrons
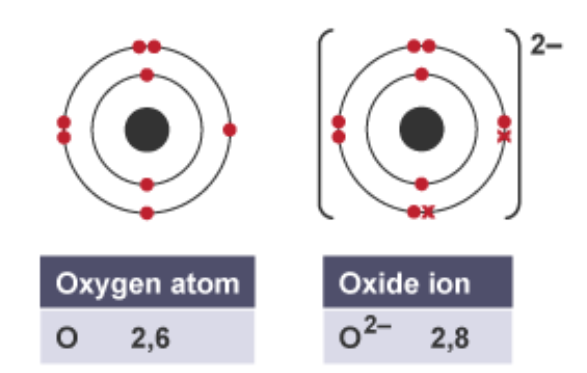
what happens when an atom has lost electrons
it becomes a positively charged ion
what happens when an atom has gained electrons
it becomes a negatively charged ion
example of an atom becoming an ion
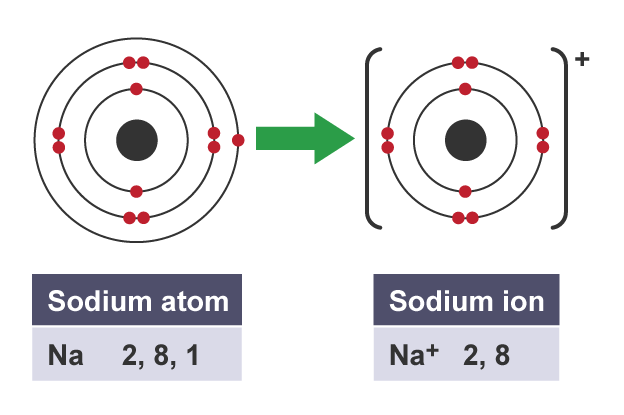
what is special about the electron structures of group 0 elements (noble gases)
they are very stable. their outer shell is full.
what is an isotope?
an atom of the same element with the same number of protons and electrons and a different number of neutrons. isotopes have different mass numbers.
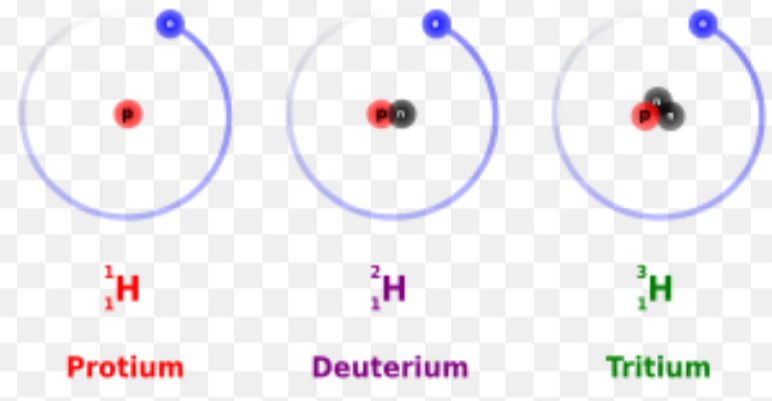
example of isotopes
isotopes of hydrogen
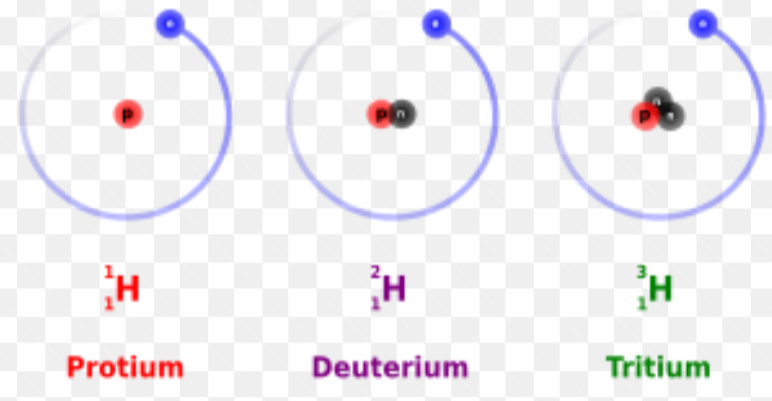
how to work out the number of neutrons in an isotope
mass number- atomic number/ number of protons
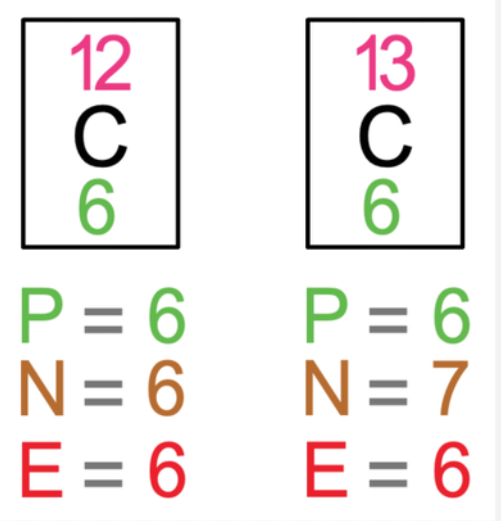
relation between group number and electronic structure
group number= number of electrons in outer shell
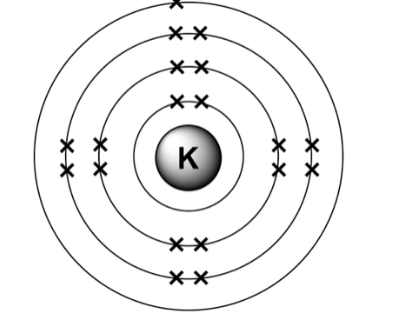
rules for electronic structure
the first shell can hold up to 2 electrons
the second and third shells can hold up to 8 electrons
the fourth shell can hold up to 18 electrons
relation between period and electronic structure
period= number of shells
what are transition metals
typical metals
what do you get when the group 1 elements areadded to water?

metal+ oxygen
metal oxide
metal+ water
metal hydroxide+hydrogen
metal+acid
salt+hydrogen

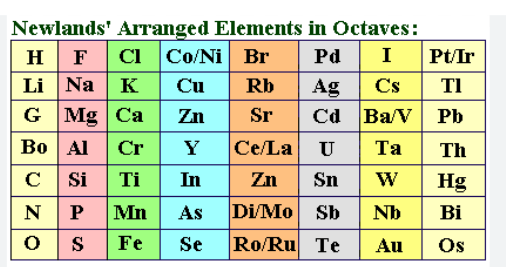
john newland`s contribution to the development of the periodic table in 1864
he noticed that when elements were placed in order of their atomic weights there were sometimes similarities between every 8th element.
produced a table showing his “law of octaves”
he did not leave gaps for undiscovered elements.

john dalton`s contribution to the development of the periodic table in 1808
he arranged the elements in order of their atomic weights


Dmitri mendleev`s contribution to the development of the periodic table in 1869
rearranged elements into order or increasing atomic weight so that a pattern in properties could be seen
left gaps for elements that hadnt been discovered
predicted the properties of these undiscovered elements, and when they were discovered they had similar properties to his predictions
because of this, his table was accepted by other scientists.

Niels bohr`s contribution to the development of the periodic table, 1913
explained that an elements position in the periodic table is determined by its electron configuration
discovered shells, and that elements in the same column have the same number of electrons in their outer shell
this provided a theoretical reason why elements in the same group share similar chemical properties.

james chadwicks contribution to the development of the periodic table, 1932
discovered the neutron.
this proved that isotopes are simply atoms of the same element with different numbers of neutrons.
his work finally allowed scientists to calculate accurate atomic weights, solidifying the arrangement of the periodic table today.
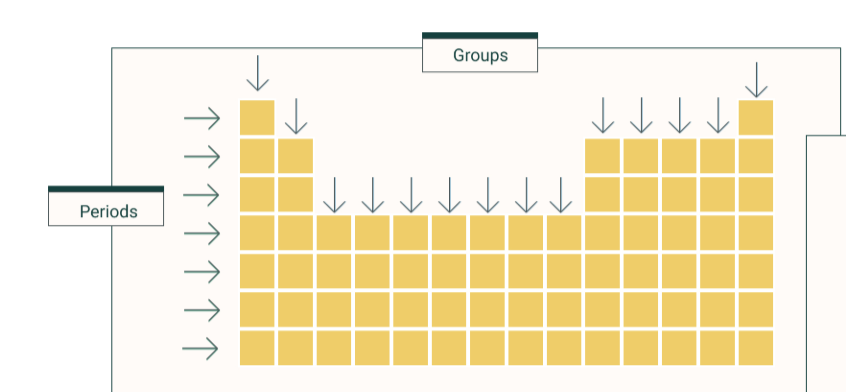
describe the modern periodic table
ordered in increasing atomic number
groups- similar chemical properties, same number of electrons in outer shell
periods- the same number of shells
what is a group
column
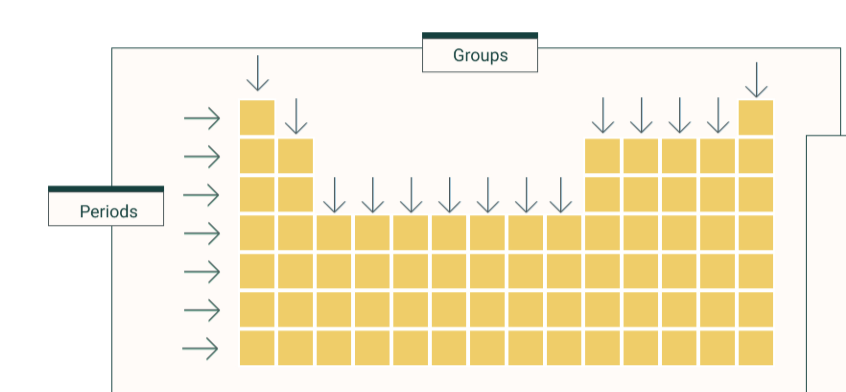
what is a period
row
what are group 0 elements
very unreactive non-metal gases, noble gases
what are group 1 elements
very reactive metals, alkali metals
what are group 2 elements
metals that form ions with a 2+ charge, alkaline earth metals
what are group 7 elements
very reactive non-metals, halogens
what are transition metals
typical metals
explain the trend in group 1
atoms get larger down the group as they have more shells
single electron in the outer shell is attracted less strongly to the positive nucleus
electrostatic attraction with nucleus gets weaker
lose the single electron more easily
also:
alkali metals become more reactive going down the group.
the melting points decrease and
they become softer.
how does the trend in group 1 (alkali metals) apply to postassium
more inner shells shield outer electron in potassium, so it loses outer electrons more easily.

observations during the reaction of lithium in water
smoke+ fizzing
floats on water, then dissolves (low density)
observations during the reaction of sodium in water
fizzing+ smoke
turns into a ball
faster reaction that lithium
observations during the reaction of potassium in water
popping sound
smoke
lilac flame
floating on water
fastest reaction
physical properties of group 7 halogens
the melting point increases going down the group
diatomic molecules (contain 2 of the same atom)
the boiling points increase going down the group
the densities increase going down the group
appearance of the halogen flourine
pale yellow gas

appearance of the halogen chlorine
green gas

appearance of the halogen bromine
orange-brown liquid (easily vaporises into a gas)

appearance of the halogen iodine
shiny grey-black crystalline solid that sublimes into a purple vapour

what is a displacement reaction
when a more reactive element displaces a less reactive element.
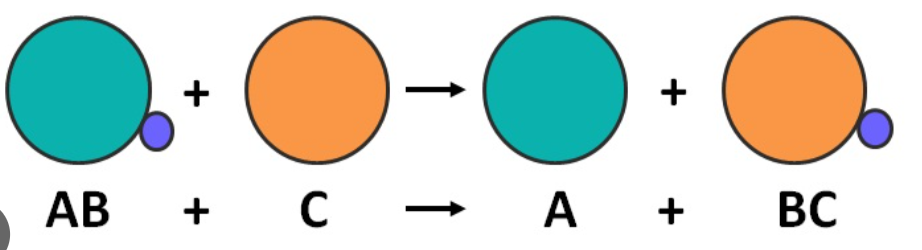
chemical symbol of aluminium ion

chemical symbol of ammonium ion

chemical symbol of barium ion
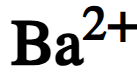
chemical symbol of calcium ion
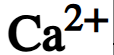
chemical symbol of copper ion
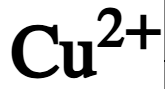
chemical symbol of hydrogen ion
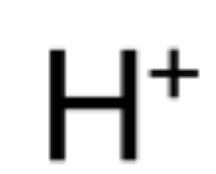
chemical symbol of iron(II) ion

chemical symbol of iron(III) ion
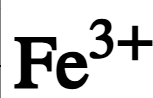
chemical symbol of lead ion
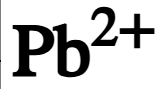
chemical symbol of lithium ion
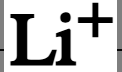
chemical symbol of magnesium ion
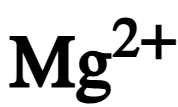
chemical symbol of potassium ion

chemical symbol for silver ion
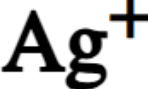
chemical symbol for sodium ion

chemical symbol for zinc ion
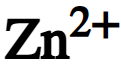
chemical symbol for bromide ion
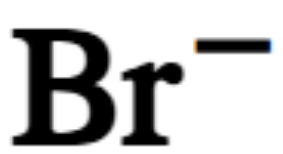
chemical symbol for carbonate ion
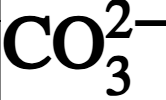
chemical symbol for chloride ion
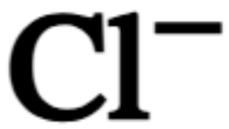
chemical symbol for fluoride ion

chemical symbol for hydrogencarbonate ion

chemical symbol for hydroxide ion

chemical symbol for iodide ion
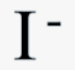
chemical symbol for nitrate ion

chemical symbol for oxide ion
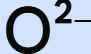
chemical symbol for sulphate ion

chemical symbol for sulphide ion
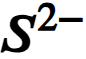
process of a gas becoming a solid
deposition
process of a solid becoming a gas
sublimation
describe a solid
tightly packed together
low energy
particles dont move(only vibrate)
forces of attraction between particles is very strong

describe a liquid
particles touch
more energy that solid
particles move and slide past each other
forces of attraction between particles is weak
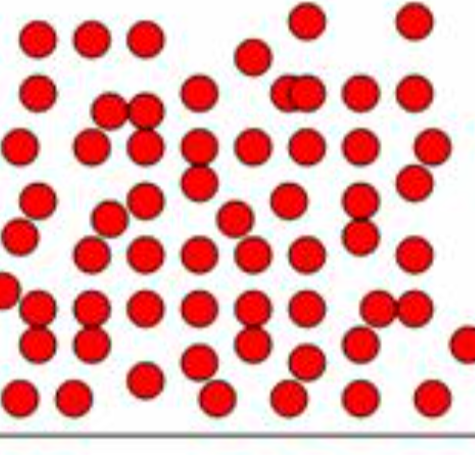
describe a gas
very spread out
high energy
random movement quickly in all directions
no forces of attraction between particles.why
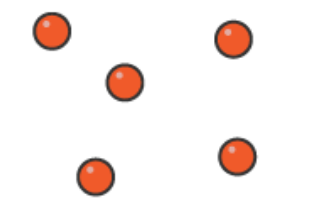
why do different substances have different melting points
dependent on the substance, substances that have strong bonds between atoms require higher temperatures to break those bonds and hence have a high melting point.