suicide pact?????
1/574
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
575 Terms
formula for BSA
use total body weight
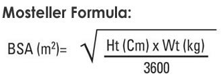
Which of the following structures can serve as an intermediate in the activation of nitrogen mustards?
(tell me what the intermediate is and what to look for)
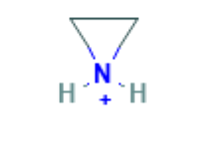
aziridinium (look for triangle)
Mustargen is a potent vesicant. In case of accidental skin contact, which of the following solutions should be applied? AND WHY
2% sodium thiosulfate solution;
bc it makes it water soluble so it can be washed off the skin
sodium thiosulfate MOA
reacts with mustargen to make inactive, highly ionized, water soluble ester that can be washed off
ifex can form what toxic metabolite(s)
acrolein
chloroacetaldehyde
temozolomide is converted to the active metabolite _____
MTIC
AZT MOA
incorporated into RNA to decrease tRNA methyltransderase, leads to inhibited methylation and then apoptosis
cyclophosphamide undergoes ______ via _____ to form what three metabolites?
C-4 hydroxylation via CYP2B6
acrolein, phosphoramide mustard, and carbolamine
Chlorambucil undergoes ____ to form active ________
beta-oxidation rxn; phenylacetic acid mustard
The _______of guanine reacts with the ___ of Lurbinectedin to form an adduct
N2 of the minor groove; iminium intermediate
Which reactive species does Carmustine generate in situ to interact with DNA?
Vinyl cation and isocyanate
6MPMP:
what does it stand for
function
what drugs are CI with it and why
6-thioinosinate or 6-methylthioinosinate
inhibits the first step of purine synthesis and the conversion of inosinic acid to xanthylic acid
xanthine oxidase inhibitors because they inhibit the enzyme that metabolizes it so it stays in the body unmetabolized
toxic effects of acrolein include…
hemorrhagic cystitis and bone marrow suppression
how does Mesna treat acrolein exposure
given before chemotherapy with Cytotoxan or Ifex
neutralizes metabolites by binding to -SH (sulfhydryl) moieties
increases urinary excretion of cysteine
lurbinectedin:
MOA
covalently binds to N2 of guanine minor groove to alkylate it
inhibits RNA polymerase II
how does lurbinectedin exert its alkylating effects
by its iminium intermediate
DOX MOA:
intercalation leads to ____ due to topo II inhibition
produces ______ that lead to cellular DNA damage and cardiotoxicity
can be reduced to a _____ by CYP450, which can lead to…
single or double strand DNA breaks
reactive oxygen species
semiquinone radical; reaction with oxygen —> make a superoxide radical —> form hydrogen peroxide and a hydroxyl radical
taxanes MOA
prevent mitosis by altering normal MT organization, preventing proper dissolution of spindles leading to cell cycle arrest
paclitaxel:
class
solubility
taxane MT targeting agent
highly lipophilic and poorly water soluble
docetaxel:
class
formulation
MT targeting taxane
must be strategized to improve solubility
How is docetaxel structurally different from paclitaxel?
Modifications increase tubulin binding affinity
What functional group allows vinca alkaloids to form water-soluble salts?
tertiary amine
What is the mechanism of vinca alkaloids? and what is the result?
Inhibit microtubule polymerization; Prevent mitotic spindle formation → mitotic arrest
What enzyme do epipodophyllotoxins inhibit? and what is their MOA?
(etoposide for example)
Topoisomerase II
Stabilize DNA–topoisomerase complex → prevent DNA religation —> DNA fragmentation —> cell death
SERMs:
MOA
exhibit ____ dependent activity
Modulate estrogen receptor signaling
tissue
Aromatase MOA
converts androgens to estrogens
aromatase inhibitors MOA
reduces circulating estrogen levels sometimes by mimicking the natural substrate
exemestane:
structure and class
reversible or irreversible enzyme inhibition?
requires consideration of the pts ____
steroidal aromatase inhibitor
irreversible
hormonal status
kinase inhibitors:
what do they mimic?
where do they bind and how?
what determines selectivity adn potency?
ATP binding
Hinge regions via hydrogen bonding
structural features
MOA:
type I tyrosine kinase inhibitors
type II tyrosine kinase inhibitors
bind to the active kinase conformation at the ATP binding site
target the inactive DFG out conformation, exposing an adjacent hydrophobic pocket
dexrazoxane:
what is it?
what it do?
iron chelating agent
reduce anthracycline associated cardiotox; limit free radical formation through metal chelation
Leucovorin:
when is it used?
what does it do?
structure / formulation
Rescue therapy after antimetabolites
Restores normal folate pathways
Racemic mixture with active L-isomer
why is platinum used?
electrophile that forms a VERY stable covalent bond with the DNA
idk
WHY does pomalidomide (thalidomide analog) has enhanced potency and improved pharmacological properties?
additional amino group on the 4th carbon of the phthaloyl ring
therapeutic vax stimulate ______ of tumor antigens
immune recognition
immunotherapy definition
using the patient's immune system to fight cancer
ability to activate the immune system and destroy cancer cells
what is the primary defense against cancer
immune system
define innate immunity
first response
does not increase or change in response to repeated exposure
non specific
define adaptive immunity
slower and antigen specific response
consists of B and T cells
increased immunity on repeated antigen exposure
creates long term memory
activation of naiive T cells will not occur in the absence of ____
costimulation
deletion of T cells that react strongly with self
negative selection
behavior of cells that have survived negative selection
interact weakly with antigens and fail to become activated
decreased response
immunotherapy approaches
1. specific and active
2. specific and passive
3. non specific and active
4. non specific and passive
specific and active immunotherapy approach example
vaccines that include long lasting memory and immunity
specific and passive immunotherapy approach examples
monoclonal antibodies and T cell based therapy
includes herceptin and mebtera
nonspecific and active immunotherapy approache examples
bacterial products and biological response modifiers (cytokines)
nonspecific and passive immunotherapy approach examples
LAK (lymphokine activated killer cells) and TIL (tumor infiltrating lymphocytes)
autologous vaccine definition
vaccine created by removing tumor cells from the patient's own body
allogenic vaccine definition
vaccine created by removing tumor cells from someone other than the patient
dendritic cell vaccines description
vaccine in which dendritic cells are generated outside that body that, through gene therapy, are able to recognize antigens
antigen vaccines description
peptide vaccines that are specific for one epitope
anti-idiotype vaccines description
vaccines that are antibodies
cause a patient to elicit an immune response after injection into the cancer
DNA vaccines description
vaccine that introduces a tumor gene
antigens are made on a continuous basis
CAR T cell therapy description
engineered molecules that can be introduced into T cells to enable them to target specific tumor antigens
T cells are removed, components are added, then programmed T cells are returned to the patient
website for searching ongoing clinical trials
go to home page at NIH then access clinicaltrials.gove
name the alkylating agents subgroups
nitrogen mustards
nitrosoureas
alkyl sulfonates
triazanes
platinum compounds
examples of nitrogen mustards
cytoxan
ifex
melphalan
chlorambucil
cytoxan is a nitrogen mustard AND an ____
immunosuppressant
nitrogen mustard MOA
forms reactive intermediate that attacks N7 of guanine to cause inter and intra strand cross links resulting in DNA strand breakage and inhibition of replication and transcription
cytoxan toxicity
hemorrhagic cystitis
nitrogen mustard toxicity
bone marrow suppression
N/V
alopecia
infertility
name a nitrosourea
carmustine
what makes carmustine / nitrosoureas unique
cross BBB
carmustine toxicity
delayed bone marrow suppression
name an alkyl sulfonate
busulfan
busulfan toxicity
tonic clonic seizures
dose limited bone marrow depression
triazene examples
dacarbazine
darcarbazine MOA
decompose spontaneously and methylate the N7 and O6 positions of G in DNA
how is resistance to alkylating agents developed?
quickly, the cancer cells increase the amount of repair proteins
what website can you find updates of clinical trials on?
clinicaltrials.gov
immunotherapy definition
activation of the immune system to destroy cancer cells using the pts immune system to fight cancer
RF of breast cancer for women (4)
***on test 3, select all that apply***
extended length of hormone exposure
getting period before 13 yrs old
starting menopause after 55 yo
nulliparity (not having kids)
what things can increase the risk of BC? (3)
***on test 3***
ionizing radiation
increased BMI (in postmenopausal women)
lifestyle choices
name 3 things that can decrease the risk of BC
***on test 3***
breast feeding
exercise
increased BMI (premenopausal)
when should the following age ranges have mammogram screening if they are asymptomatic for BC?
ages 40-44
ages 45-54
ages 55+ AND have 10+ yr life expectancy
optional
yearly
every 2 years
a 52 yo woman comes into the clinic and has average risk for all cancers, which of the following screenign can she have done today?
a. self breast exam
b. mammogram
c. chest X ray
d. low dose helical CT
e. colonoscopy
b and e
(self exams are not reccomended, does not meet requirements for either a chest x ray or a low dose helical CT)
is it recommended to get a yearly breast exam?
nah
pts who DO NOT HAVE BC, but are high risk for developing it should be started on what preventative measure?
endocrine therapy
endocrine therapy is given to prevent BC in high risk pts.
what is the drug of choice for premenopausal pts
what are the options for post menopausal pts
tamoxifen
tamoxifen, raloxifene, aromatase inhibitor
how long should a pt be on endocrine therapy if they DO NOT have BC but are at high risk?
5 yrs
What receptor statuses are assessed in nonmetastatic breast cancer diagnosis?
Hormone receptors (estrogen/progesterone), HER2/neu, BRCA1/2 germline
TX for Ductal Carinoma in Situ:
local treatment options
lumpectomy and XRT
TX for Ductal Carinoma in Situ:
if the pt had a lumpectomy or XRT and is ER+, they should then be started on _____ (drug) if they are premenopausal for _____ (duration)
tamoxifen x 5 yrs
TX for Ductal Carinoma in Situ:
if the pt had a lumpectomy or XRT and is ER+, they should then be started on _____ (drug) if they are postmenopausal for _____ (duration)
tamoxifen or aromatase inhibitor
5 years
what are the systemic therapy options for non metastatic BC
endocrine
chemo
biologic
immuno
what immunotherapy agent is indicated for non-metastatic BC
pembrolizumab
what chemotherapy agents are indicated for non metastatic BC
anthracyclines (DOX)
carboplatin
cyclophosphamide
taxanes
pts with high risk BC qualify for…
chemotherapy
what is considered high risk diseases when it comes to BC?
When do HR+/HER2− patients get chemotherapy?
pts with HR+ / HER2 - with high risk of relapse (based on additional testing)
HER2+
HR- and HER2- (triple negative)
a pt is triple negative, what treatment would you reccomend
a. TCH
b. TCHP
c. TH
d. TC
***on test***
d
(idk if its right but i think it is)
pts with the following criteria all qualify for…
pts with HR+ / HER2 - with high risk of relapse (based on additional testing)
HER2+
HR- and HER2- (triple negative)
chemotherapy
what does ddAC stand for
dose dense doxorubicin and cyclophosphamide
what does TC mean
docetaxel and cyclophosphamide
what does T mean
paclitaxel or docetaxel
adjuvant chemotherapy for HER2- BC:
what are the three options?
ddAC x 4 cycles then T q2w x 4 cycles
ddAC x 4 cycles then weekly T x 12 cycles
TC q21 days x 4 cycles
Which adjuvant chemo regimens for HER2- require growth factor support?
all but weekly T (paclitaxel)
anthracycline ADE
red urine and body fluids
mucositis
cardiac dysfunction (usually permanent)
heart failure (irreversible)
anthracyclines are CI in which pts
low LVEF
pts on anthracyclines require an ECHO every ____ while on tx
3 months