Development of T Lymphocytes
1/58
Earn XP
Description and Tags
These flashcards cover key vocabulary and concepts related to the development of T lymphocytes, including processes, structures, and terms essential for understanding T-cell biology.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
Thymus
Primary lymphoid organ responsible for T-cell development.
Thymocytes
Immature T cells that undergo key developmental steps in the thymus.
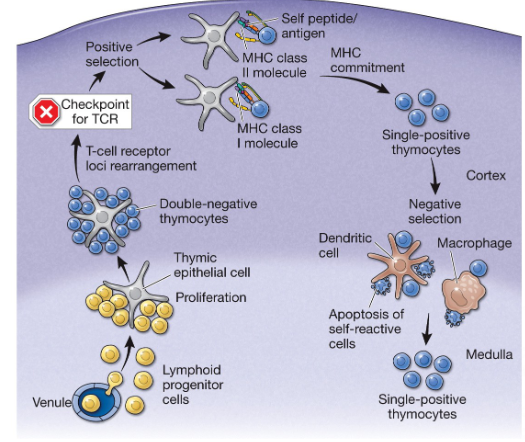
Double-negative thymocyte (DN)
Thymocytes that do not express either CD4 or CD8 and are found in the thymic cortex.
Double-positive thymocyte (DP)
Thymocytes that express both CD4 and CD8 and have a fully rearranged T-cell receptor.
Single-positive thymocyte (SP)
Thymocytes that express only one of the coreceptors (CD4 or CD8) after selection processes.
Positive Selection
Process that promotes selection of thymocytes that can bind to self-MHC molecules (MHC restriction)
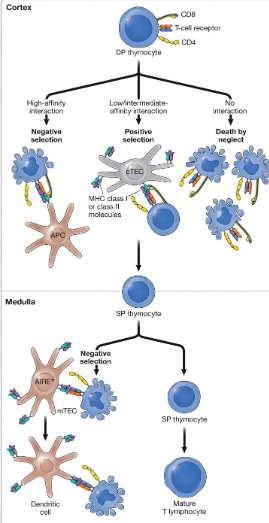
Negative Selection
Process that prevents the release of thymocytes that can recognize MHC-self-peptide complexes with high-affinity, ensuring self-tolerance.
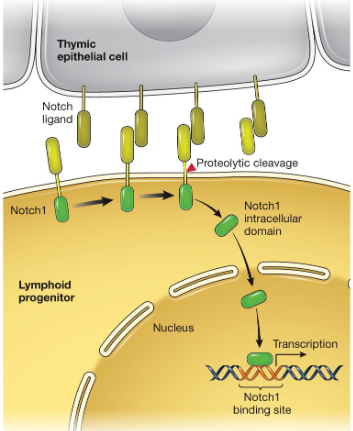
Notch1
A signaling pathway essential for the differentiation of lymphoid progenitors into T cells.
Junctional Diversity
The variation in T-cell receptors achieved by adding or removing nucleotides during recombination.
AIRE
a transcriptional activator (autoimmune regulator) allows cells to express genes not normally expressed by epithelial cells of the thymus
What occurs in the medulla of the thymus?
mature thymocytes undergo negative selection and finish developing into naive T-cells
What do developing thymocytes enter the thymus as?
double-negative thymocytes, which undergo cell surface changes to become double-positive thymocytes.
What is the role of medullary thymic epithelial cells (mTECs)?
mTECs play a crucial role in presenting self-antigens to developing T-cells, facilitating negative selection to prevent autoimmunity. essential in the establishment of self-tolerance in T-cells and express up to 100-fold more cell surface MHC 1 proteins than epithelial cells
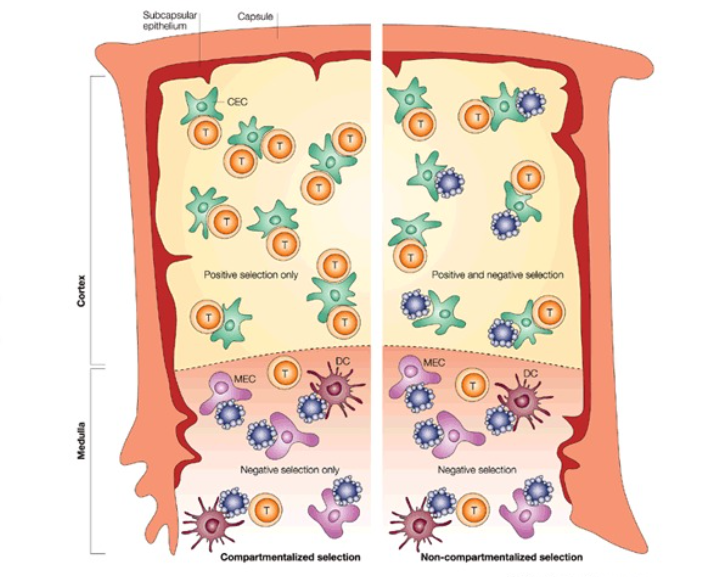
Thymic epithelial cells (TECs)
a diverse population of non-hematopoietic cells in the thymus that supports T-cell development and selection, including both cortical and medullary types.
What is the role of cortical thymic epithelial cells (cTECs)
interacts with double-positive thymocytes to test the affinity of the TCR with MHC-peptide complexes at the cell, supports the positive selection of developing thymocytes by presenting specific peptides to T-cells. can express MHC class I and II molecules
Resident cells of the thymus do what?
drive the development of thymocytes into naive T cells
Hassall’s corpuscles
are structures formed by medullary thymic epithelial cells that play a role in the maturation and selection of T cells, particularly in the induction of regulatory T cells.
Which cells are precursors to developing thymocytes?
Hematopoietic stem cells which are characterized by cell-surface markers present on the stem cell precursors, along with the absence of lineage specific markers.
What are the cell surface markers present on stem cell precursors?
CD34 and CD38
The protein notch
is a cell-surface receptor that interacts with transmembrane ligands on adjacent cells
Double negative thymocytes
are thymocytes that do not express CD4 or CD8 markers, and are found in the thymic cortex during initial development
How do double negative thymocytes develop?
they begin somatic recombination at the T-cell receptor loci and continue until both subunits of the receptor have properly rearranged, now expressing both coreceptors (CD4 and CD8)
Double positive thymocytes
are thymocytes that express both CD4 and CD8 markers and a fully rearranged TCR, indicating they are further along in T cell development and are primarily found in the thymic cortex.
What are the possible fates of double positive thymocytes?
some develop into regulatory T-cells, and come begin the process of positive and negative selection to test the TCR and select for a single coreceptor to become a single-positive thymocyte
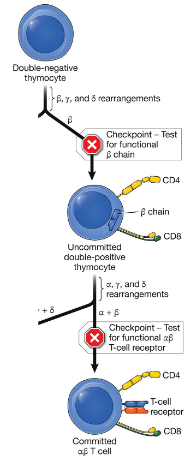
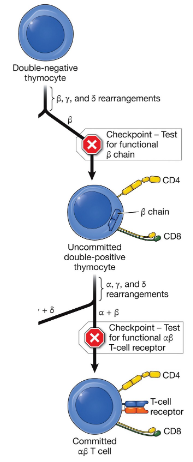
What are the checkpoints a thymocyte must pass to conti8nue development and become a functional T cell?
checkpoint of the β subunit of the T-cell receptor and checkpoint of the α subunit of the T-cell receptor
What causes T-cell diversity?
somatic recombination at the receptor loci that generates a vast array of T-cell receptors through rearrangement of variable (V), diversity (D), and joining (J) gene segments.
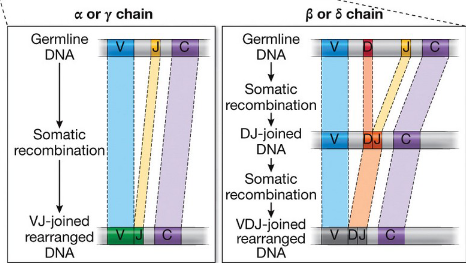
What activates somatic recombination?
the expression of the RAG1 and RAG2 proteins
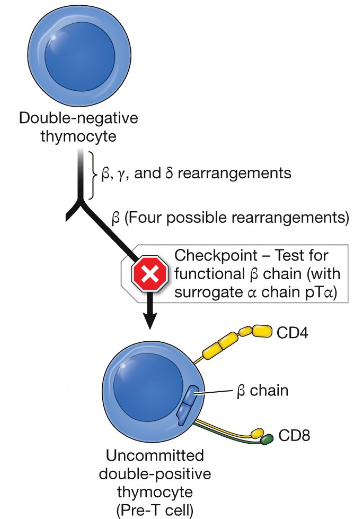
Where do recombination events begin?
at the β loci of the TCR
How many productive β-chain rearrangements are possible?
four
To test productive rearrangement of the β chain
the developing thymocyte must assemble a surrogate T-cell receptor complex
When does recombination of the α-chain locus occur?
once the β-chain checkpoint has been passed
Pre-T α-chain (pTα)
a surrogate alpha chain, assembles with the rearranged β-chain and the CD3 complex to form the pre-TCR
why is the pTα important?
The pTα is crucial because it allows the formation of the pre-T-cell receptor (pre-TCR) and ensures that a functioning T-cell receptor can signal via the same transduction pathways used to activate T cells
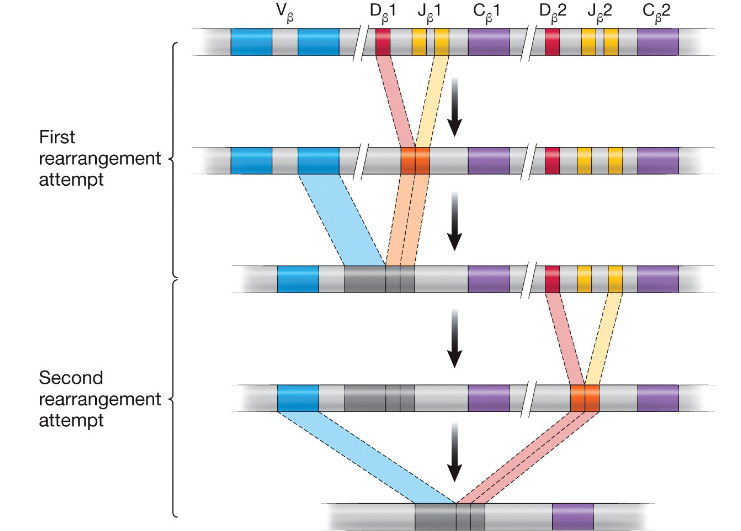
What happens after the cell goes through the β-chain checkpoint?
it proliferates, expresses RAG1 and RAG2 again which begins recombination at the other T-cell receptor loci (α), it then undergoes allelic exclusion
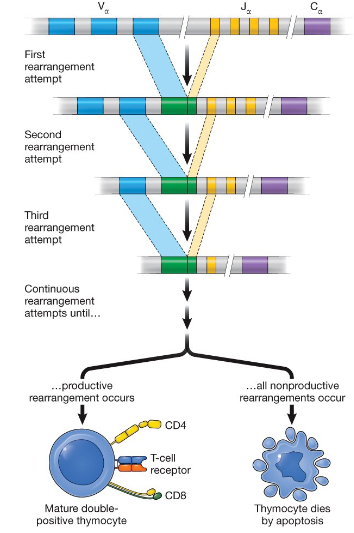
Where is the α chain tested to ensure TCR function?
at the ER membrane,
What happens if the expressed α chain does not function properly
the α-chain locus is further rearranged
What happens if a functional α chain is produced?
the developing thymocyte continues development into a naive T cell
Death by neglect
occurs when double positive thymocytes cannot interact with any MHC-peptide complexes
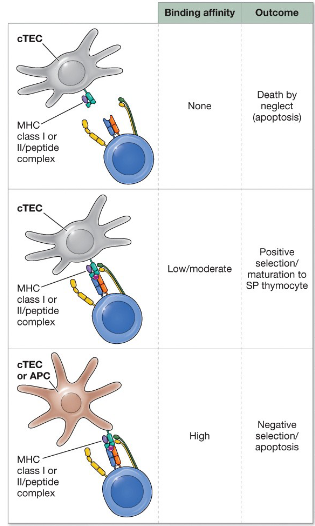
Three possible outcomes of positive selection within the thymic cortex
death by neglect: occurs when double positive thymocytes cannot interact with any MHC-peptide complexes
negative selection: DP thymocytes bind too tightly with an MHC–peptide complex
positive selection: DP thymocytes with TCRs that can interact with an MHC–peptide complex with a low or intermediate affinity survives and proliferate
Lineage commitment
commitment of a thymocyte to express a single coreceptor
During selection in the thymic cortex, double positive thymocytes?
further develop and express only a single coreceptor
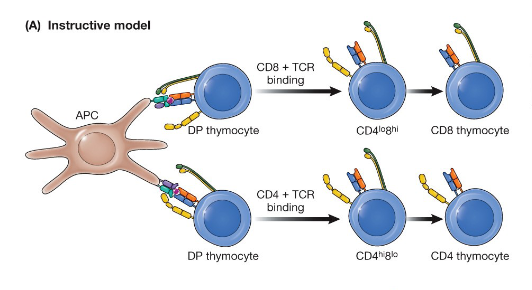
Instructive model
interaction-driven selection, if a TCR engages a MHC1 molecule presenting a peptide, the interaction will also promote CD8 interaction with the complex and shut down CD4 expression, If a TCR engages an MHC class II molecule presenting a peptide, the CD4 coreceptor will engage in the interaction, and the thymocyte will receive a signal to prevent CD8 expression
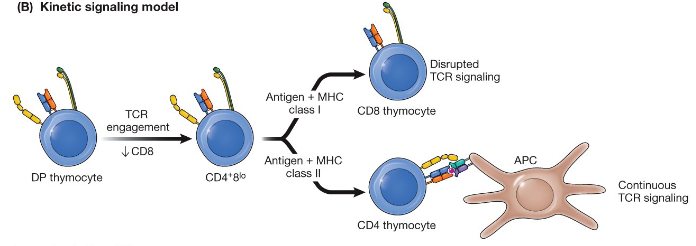
Kinetic signaling model
signal strength-driven selection, positively selected thymocytes will become CD4+ if the T-cell r4eceptor/ coreceptor signal is continuous, positively selected thymocytes will become CD8+ if this signal is interrupted
What are the two proposed models of lineage commitment?
Instructive model and Kinetic signaling model.
Negative selection of the DP thymocytes can occur is the T-cell receptor:
MHC-peptide complex has too high an affinity, leading to apoptosis of those thymocytes that bind too strongly, thereby preventing autoimmunity.
Central tolerance
negative selection processes that occur in primary lymphoid tissues that are responsible for the removal of self-reactive lymphocytes, thereby preventing autoimmune reactions and ensuring tolerance to the body's own antigens.
AIRE ensures that genes of tissue-specific antigens are activated in the thymic epithelial cells to
promotes the expression and processing of these antigens within the thymus
How does AIRE act as a gas pedal for RNA polymerase II?
by causing transcription of tissue-restricted genes and presentation of tissue-specific antigens on MHC within mTECs
What is a complicating factor in the process of central tolerance?
certain endocrine tissues within the body are the source of very specific proteins that may not be expressed in the thymus, making it difficult for developing T cells to recognize and eliminate all self-reactive cells.
Medullary thymic dendritic cells can
engulf mTECs and present tissue-specific antigens via MHC class II, this allows for negative selection of CD4+ thymocytes that bear a TCR that interacts too strongly with an MHC class II-tissue-specific antigen peptide
Negative selection drives
central tolerance and limits the circulation of self-reactive T cells
Transcription factor FOXP3
is expressed when a subset of CD4 T cells express a self-reactive TCR
nTregs
CD4+ and FOXP3+ cells that continue to develop become regulatory T cells (Tregs), nTregs are released into circulation to promote peripheral tolerance
T cells undergo positive selection to ensure
that they have a functional receptor
T cells undergo negative selection to ensure
that they are not self-reactive
Regulatory T cells can provide further means of tolerance by
inactivating self-reactive T cells in their periphery
T cells that have rearranged their T-cell receptor loci undergo
both positive and negative selection in the thymus
Most circulating T cells express an αβ T- cell receptor and when activated
αβ T- cells can differentiate into effector T cells that activate a range of innate or adaptive immune responses, or directly destroy an infected cell
T cell receptor diversity is driven by
The number of V, D, and J subunits present for all chains associated with the receptor, Junctional diversity: adding or removing P and N nucleotides during recombination, and random association of subunits