Liposomes
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Liposomes
•Liposomes are spherical self-closed structures, composed of curved lipid bilayers, which enclose part of the surrounding solvent into their interior. They trap water and other molecules inside. Due to curved lipid bilayer which has resemblance to our lipid cell membrane bilayer they are biocompatible
•The size of a liposome ranges from 20 nm up to several micrometers and they may be composed of one or several concentric membranes.
•
•Liposomes are composed of amphiphilic lipids → essential for their ability to carry different types of drugs whether it is hydrophilic or lipophilic for drugs.
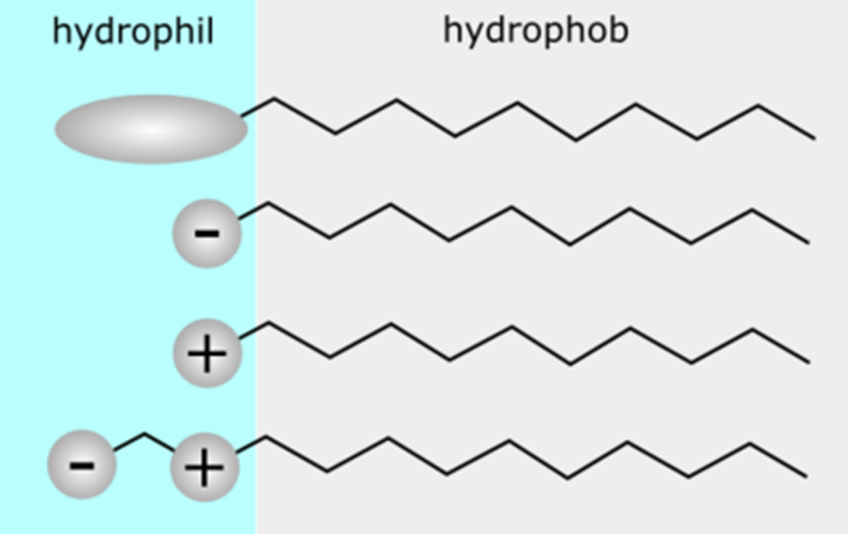
Amphiphiles
•They possess two features:
1- hydrophilicity
2- hydrophobicity
E.g. surfactants like soap. Interact with both water and fats
•
•They tend to aggregate spontaneously. Naturally assemble into structures to hide hydrophobic tails inside and exposing hydrophilic heads to water. This behaviours allows liposomes to form and maintain their structure in an aqueous environment. They take their structure similar to polymeric micelles.
Amphiphilic Lipids
•Composed of a polar head group and a non-polar tail of one or two hydrocarbon chains. Depending on the space requirement of both building blocks, they form micellar (one alkyl chain) or lamellar (two alkyl chains) structures in the aqueous environment of living systems.
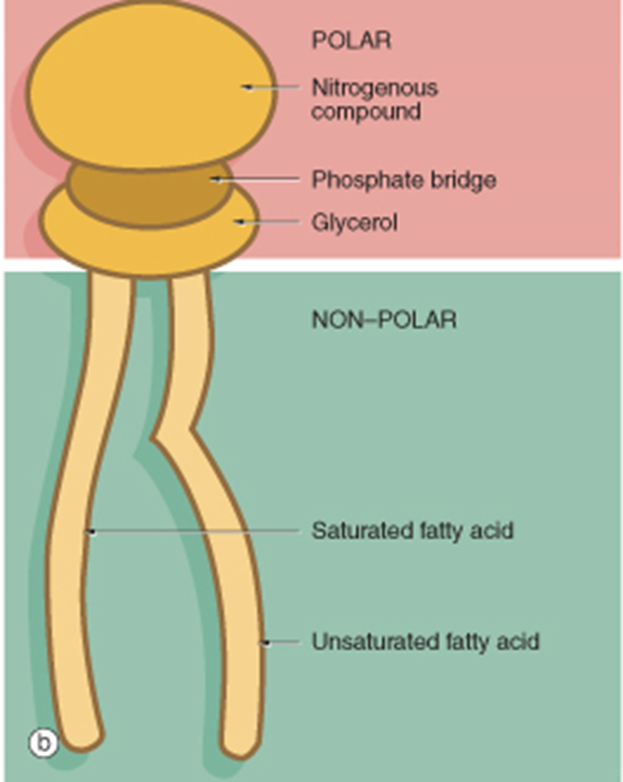
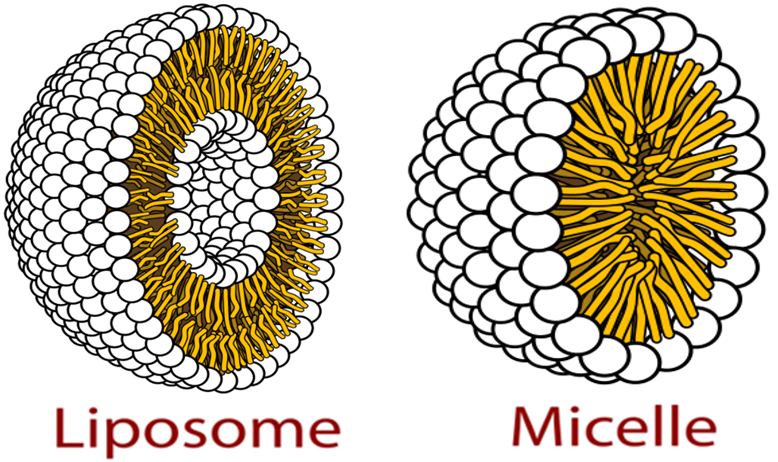
Liposomes are different to micelles as they have a double layer so both the outer layer and the inner core are hydrophilic. Lipophilic part is between these bits
Geometry of amphiphiles and their assembly
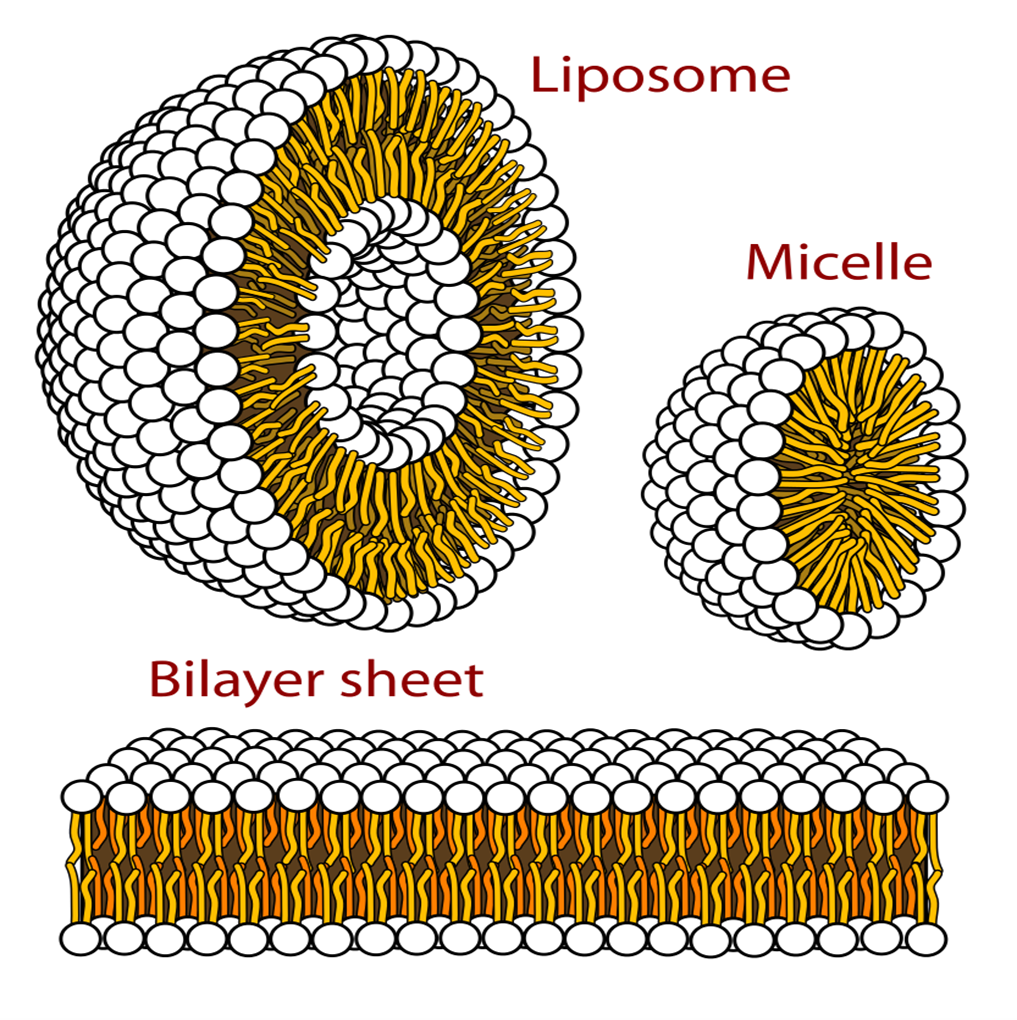
Amphiphilic molecules arrange themselves in different shapes and that depends on the environment around them. They can form a bilayer sheet which is similar to lipid membrane but this eventually will fold in on itself to form liposomes to reduce interaction with water,
Geometry of amphiphiles and their assembly
Amphiphile molecules undergo a geometry-guided rearrangement when dispersed in an aqueous phase to minimise energy and sterilise their structure
The rearrangement occurs in a way that masks the hydrophobic regions of the amphiphile from the aqueous environment.
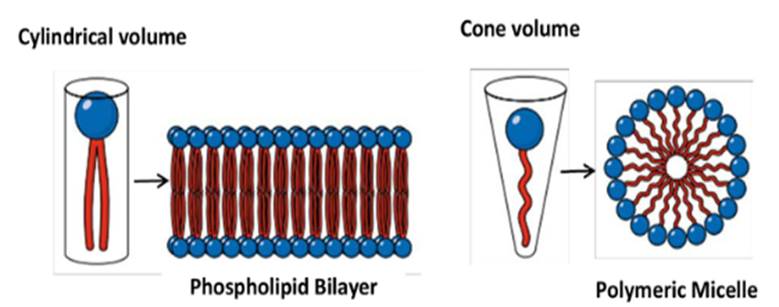
If amphiphile has a cylindrical geometry (i.e. The cross sectional area of polar and non-polar moieties are almost equal), molecules align to form a monolayer that arranges symmetrically with another monolayer to form a bilayer (the lamellae or lamellar phase). Basically saying if the hydrophilic and hydrophobic parts are equal in size then they will tend to form a bilayer that will later on lead to the formation of liposomes. However if they take the cone shape then they will most likely form a polymeric micelle.
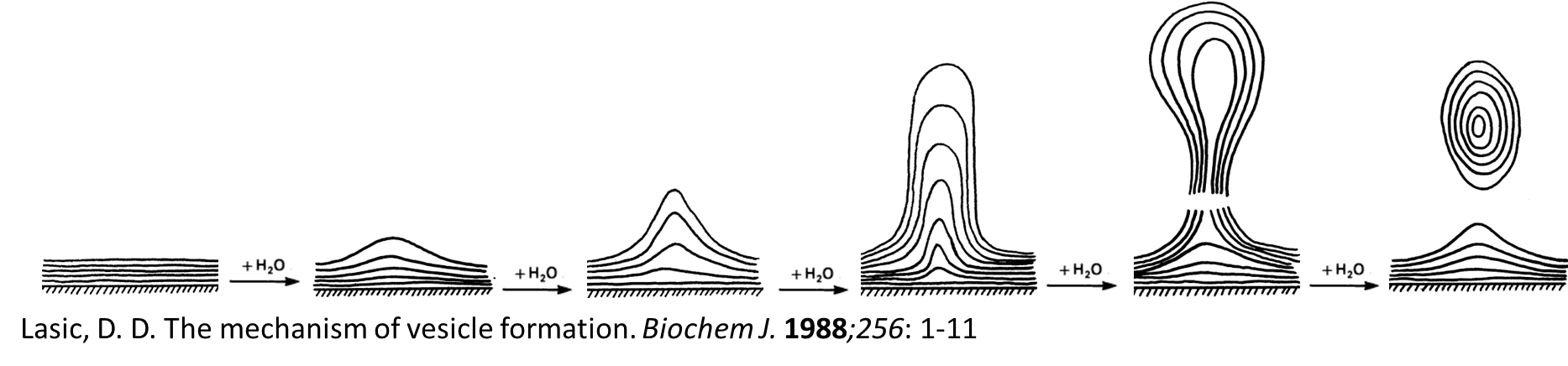
Mechanism of liposome formation
1. The hydration of outer monolyer predominates compared to inner layers. Outer layer absorbs water. Begins to swell and not dissolve as it is a lipid
2. The hydrophilic group of phospholipid expands and forms “blisters”.
3- Aqueous phase permeate through the blisters and form bilayers, resulting in formation of tubular fibrils. Tubular fibrils will try to minimise the energy so it will round up into stable multi lamellar vesicles. This increases the surface area in contact with aqueous phase.
4- The exposure of the hydrophobic moieties in phospholipids to the aqueous environment is thermodynamically unfavourable. This will force the bilayers to round up and form multilamellar vesicles (MLVs).
Mechanism of liposome formation
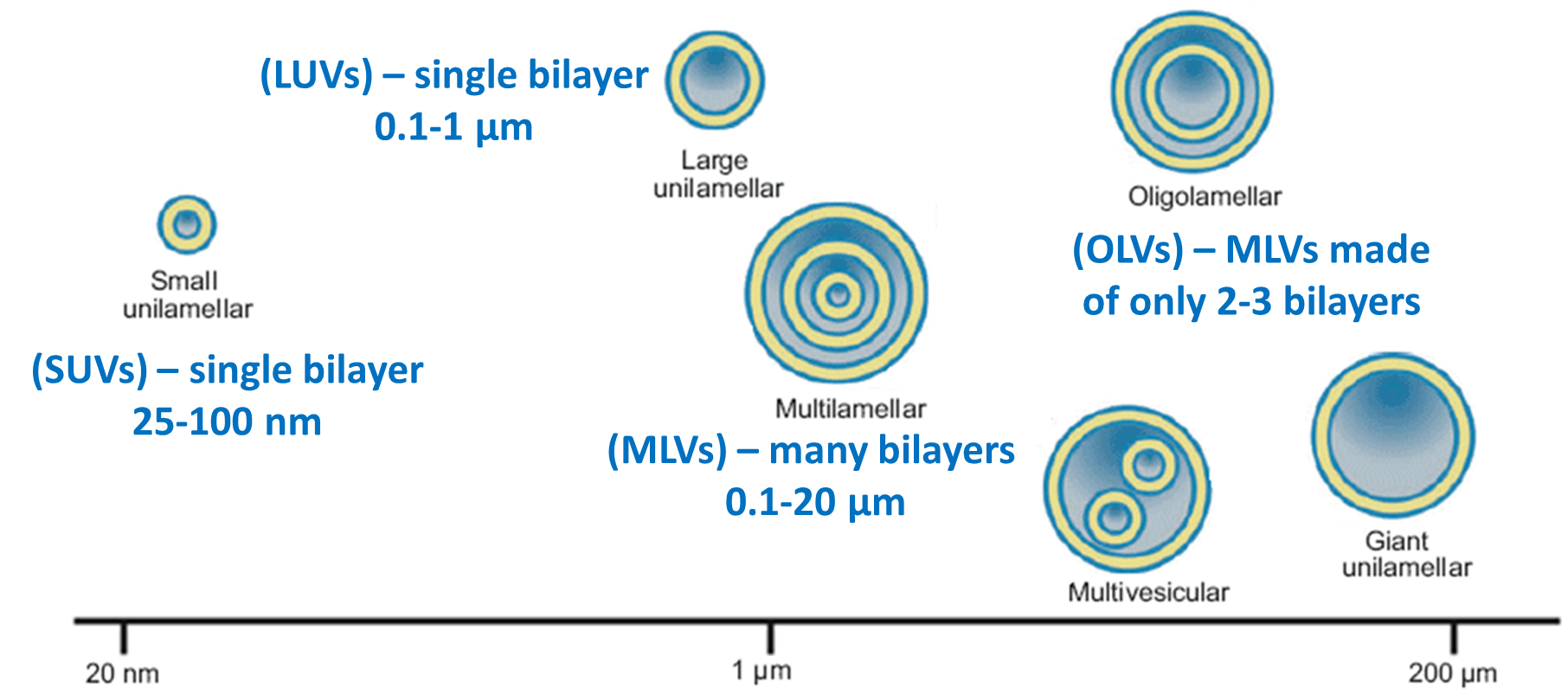
Types of Liposomes
Liposomes are phospholipid vesicles dispersed in aqueous phase and have a size between 25 nm and 25 µm. According to size and morphology of liposomes, they are classified into
DRUG LOADING IN LIPOSOMES
Hydrophilic drugs are entrapped in the central aqueous core and in the aqueous spaces between the successive bilayers of liposomes.
Hydrophobic drugs are entrapped in the hydrophobic region within the bilayers.
How about amphiphilic drugs? They will try to partition themselves between the bilayer and the aqueous core depending on their chemical properties.

DRUG LOADING IN LIPOSOMES
Advantages of drug entrapment (encapsulation) in liposomes:
Provision of sustained release formulation which improves the duration of drug action and reduce the frequency of administration
Targeted drug delivery.
Gene internalization → can carry RNA and DNA.
Protection of entrapped material from metabolic degradation in vivo, especially if we have sensitive molecules.
Solubilisation of hydrophobic materials
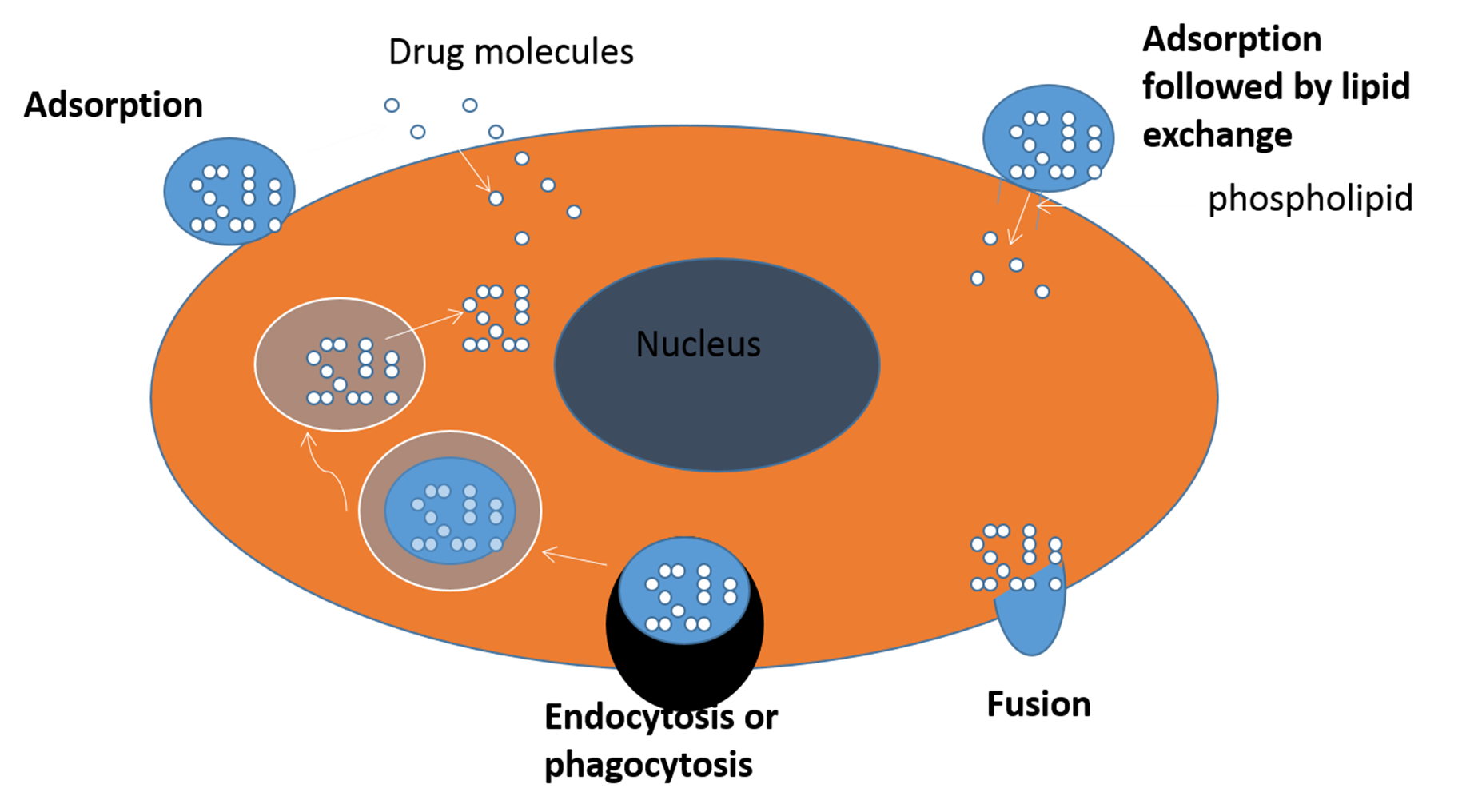
INTERACTION OF LIPOSOMES WITH CELLS
There are four proposed mechanisms for liposome-cell interactions:
ØAdsorption → attaches to cell membrane
ØLipid exchange → lipids in liposome transfer to cell membrane
ØEndocytosis & receptor-mediated endocytosis → entire liposome will be engulfed by the cell and taken inside.
ØFusion → liposome merges with cell membrane and releases its content.
•
The type mechanism is dependent on:
•
ØSize of liposome - larger liposomes may be taken up by phagocytosis while smaller ones will be taken up by endocytosis.
ØSurface charge of liposome - positively charged liposomes will interact better with negatively charged ell membranes.
ØBilayer composition of liposome - stability and fluidity of the liposomes will determine how easily they can merge with the cell membrane.
ØType of ligands attached to liposomes - molecules on the liposomes which can interact with particular cells.
ØType of cell and the cellular microenvironment around the cell - e.g. different cells prefer different uptake mechanisms
INTERACTION OF LIPOSOMES WITH CELLS
qMechanisms of liposome-cell interaction:
ØAdsorption:
Liposome is asdorbed at the cell surface through electrostatic or hyrophobic interactions, followed by extracellular release of the liposome-entrapped drug, which may be taken up by the cell via passive or active transport systems.
ØLipid exchange:
Lipophilic component of the liposome bilayers may be transferred to the cell membranes and released into the cytoplasm. This transfer allows certain hydrophobic drugs to passively diffuse into the cytoplasm of the cell.
These two methods allow for drug delivery without full liposome entry into the cell making them useful for surface based therapies.
ØEndocytosis:
The whole liposome is engulfed by cell into the lysosomes where liposomes are digested and their contents released. Many nanoparticle drug carrier use this to target cancer cells
ØFusion:
Liposome fuses with cellular membrane or endosomal membrane, followed by intracellular release of liposome contents. For fusion to occur, liposomes should be functionalised with fusion proteins or peptides. Lipsomes designed for gene therapy often rely on fusion to efficiently introduce DNA/RNA into cells.
INTERACTION OF LIPOSOMES WITH CELLS
INTERACTION OF LIPOSOMES WITH CELLS
qLiposomes which are large in size (e.g. MLVs) and made of low stability phospholipids are commonly cleared from blood circulation very quickly after parenteral administration. Liposomes are taken up by blood macrophages via receptor-mediated endocytosis or phagocytosis → easily recognised by immune system. Smaller liposomes may also be cleared quickly if they have not been modified appropriately. So in all cases, liposomes must be engineered to avoid immediate clearance so they can reach the intended target tissue and deliver the drug.
•
qLiposomes may enter the endosomes which in turn fuse with the cellular lysosomes, resulting in digestion of liposomes and release of the encapsulated material into the cytoplasm. If drug is intended to act inside the cell then this process is beneficial however if drug is degraded by lysosome then fusion based delivery is preferred as drug is now lost.
•
qThe distribution, fate and interaction of liposomes with cells can be studied by encapsulating a pH-sensitive fluorescent marker that would be activated at low pH (i.e. in the endosomes or lysosomes), or by radiolabelling the liposomes phospholipids
CLEARANCE OF LIPOSOMES
qLiposomes are not excreted through renal clearance, due to their large size that hinders glomerular filtration.
qLiposomes are cleared by the ReticuloEndothelial System (RES) and breakdown of the liposome structure.
qLiposome clearance takes place mainly through liver and spleen, where macrophages are present.
qFactors influencing liposomal drug clearance from blood circulation:
ØLiposome size
ØLiposome surface properties , also lipid charge as highly charged liposomes will be cleared faster.
ØLipid phase composition
Classification of Liposomes
•Liposomes are phospholipid vesicles dispersed in aqueous phase and have a size between 25 nm and 25 µm. According to size and morphology of liposomes, they are classified into:
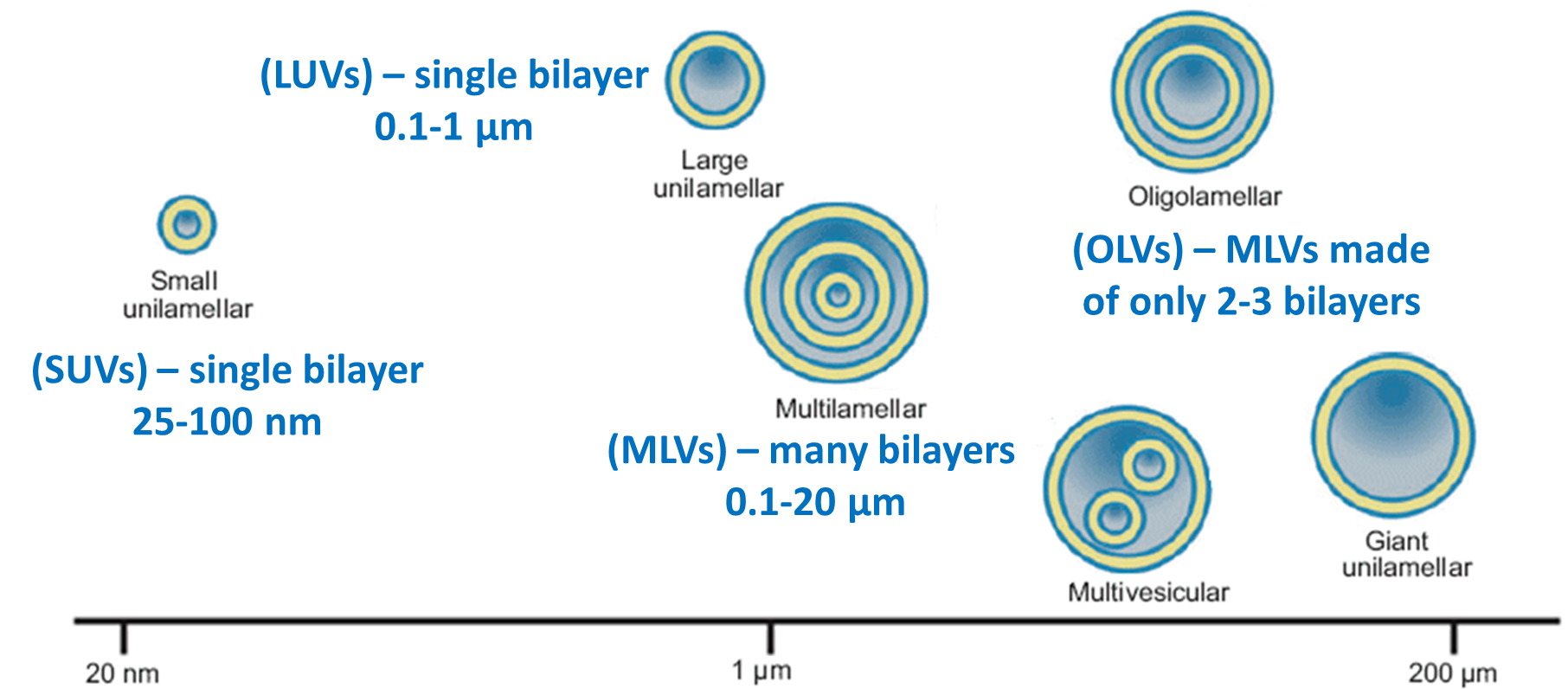
Classification of Liposomes
(according to their biological functionality)
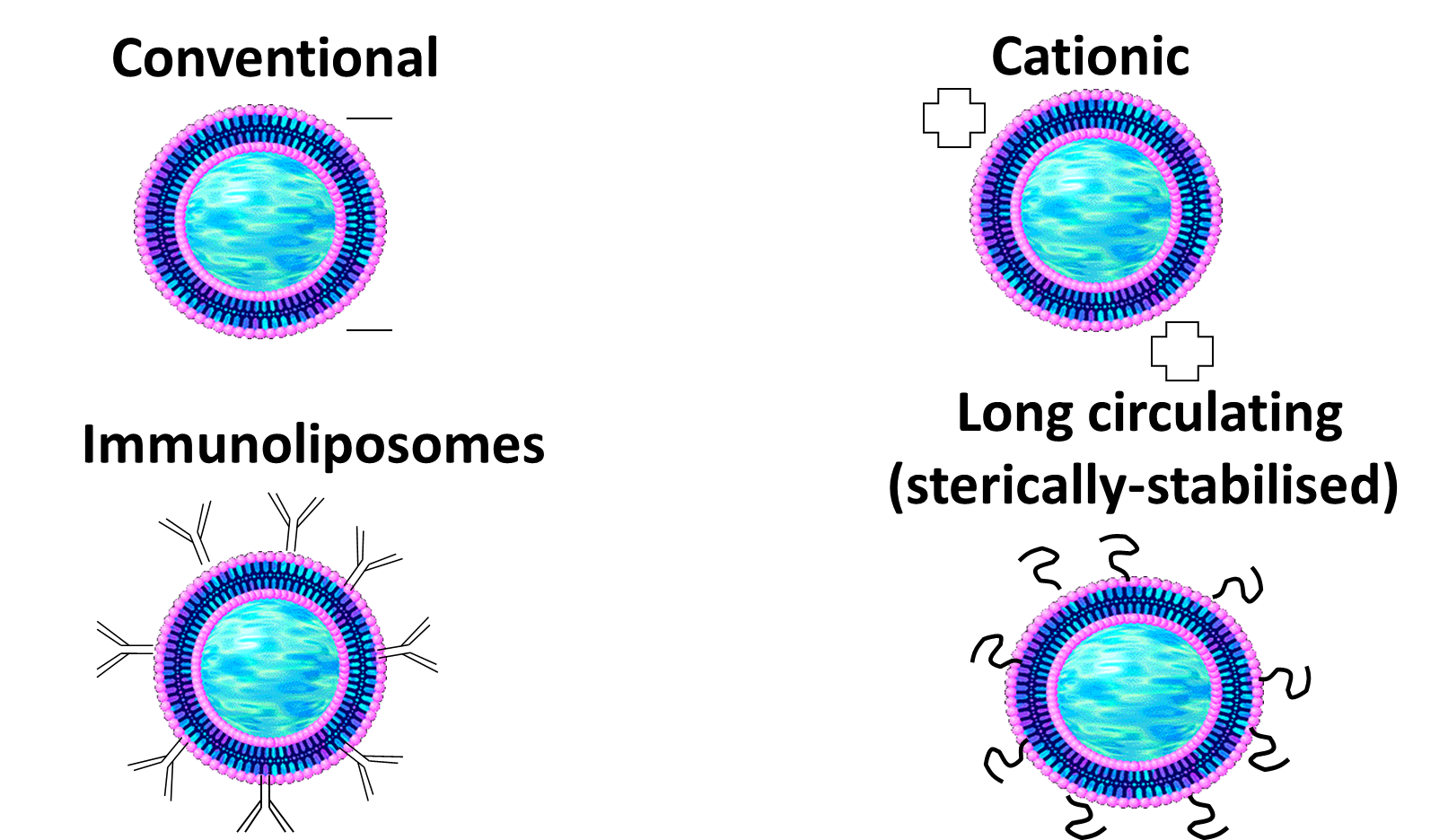
Conventional liposomes are naturally negatively charged but can be modified to be catioinic. These have better interaction/Adsorption on the surface on the cells.
Immunoliposomes are targeted drug delivery using antibodies
Long circulating ones circulate for longer due to presence of PEG molecules.
CONVENTIONAL LIPOSOMES
qPrepared by thin-film hydration method. Thus, they are polydispersed MLVs.
qPhospholipids used are either neutral or negative in charge, which can help increase the interaction with certain cells,
-
qAfter parenteral administration, they are rapidly cleared by reticuloendothelial system (RES) (i.e. short-circulation time in the blood). → quickly recognised by immune system and cleared in the liver and spleen. Limits prolonged drug delivery. This can be good for targeting infections or diseases that happen in liver and spleen as they accumulate there.
q
qThis results in liposome accumulation in Kupffer cells in the liver and in the macrophages of the spleen. This, however, can be exploited to target antibiotics to infected mononuclear phagocyte system in the liver and spleen.
q
qThe use of conventional liposomes is highly confined to research.
LONG-CIRCULATING LIPOSOMES
qProlonging circulation time enhances the possibility of liposome interaction with the target tissue.
•
qSmall liposomes (<100 nm) when made of stable phospholipid (e.g. DSPC*) and cholesterol exhibited much longer circulation time than conventional MLVs.
•
qMikasome® (NeXstar Pharmaceuticals, CA, USA) comprises amikacin antibiotic encapsulated in SUVs made of stable phospholipids and cholesterol.
•
Prolonging circulation time of liposomes has also been achieved by using glycolipids (e.g. monosialoganglioside) and hydrogenated phosphatidylinositol
STERICALLY STABILISED (STEALTH®) LIPOSOMES
qSterically stabilised (Stealth®) liposomes (SSL) are the new generations of long-circulating liposomes - Called stealth as they can evade detection from immune system extending their time in the blood stream.
•
qSSL are formulated from polyethylene glycol (PEG) derivatives of phospholipids (e.g. PEG-disteroyl phosphatidylethanolamine) to produce vesicles with more hydrophilic surfaces.
qThe hydrophilicity of the liposome surfaces would provide steric stabilisation by making bilayers hindered and thus more resistant to opsonisation and hence less detectable by the macrophages and less RES-mediated clearance.
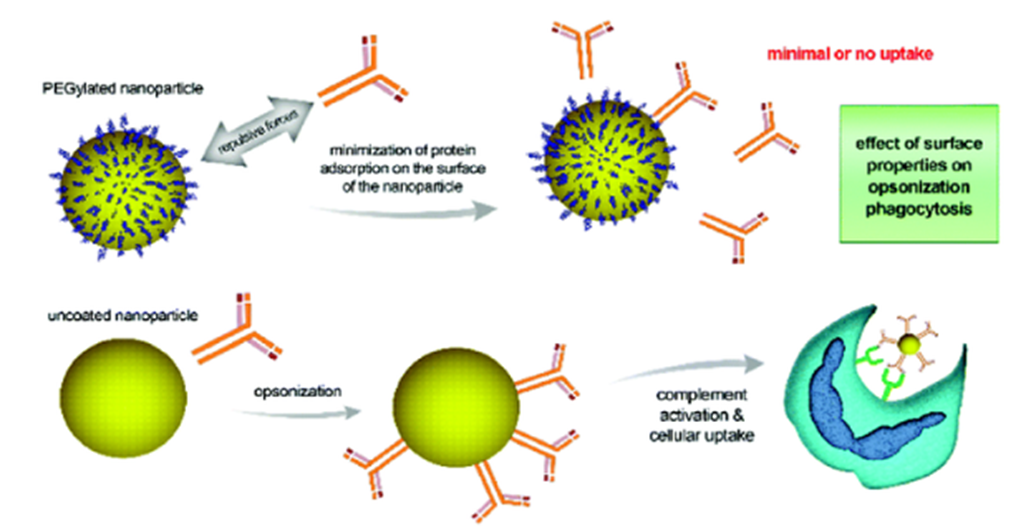
STERICALLY STABILISED (STEALTH®) LIPOSOMES
qStealth® liposomes showed prolonged residence in the blood with less need to reduce vesicle size, increase lipid amount or use lipids with higher Tm.
•
q Stealth® liposomes have applications in treatment of cancer and infectious diseases.
qAn example of drugs that have been encapsulated in Stealth® liposomes is doxorubicin HCl. This has reached the market as Doxil® in USA and Caelyx® in Europe and is used for treatment of Kaposi’s sarcoma.
STERICALLY STABILISED (STEALTH®) LIPOSOMES
Doxorubicin-loaded Liposomes (Doxil®)
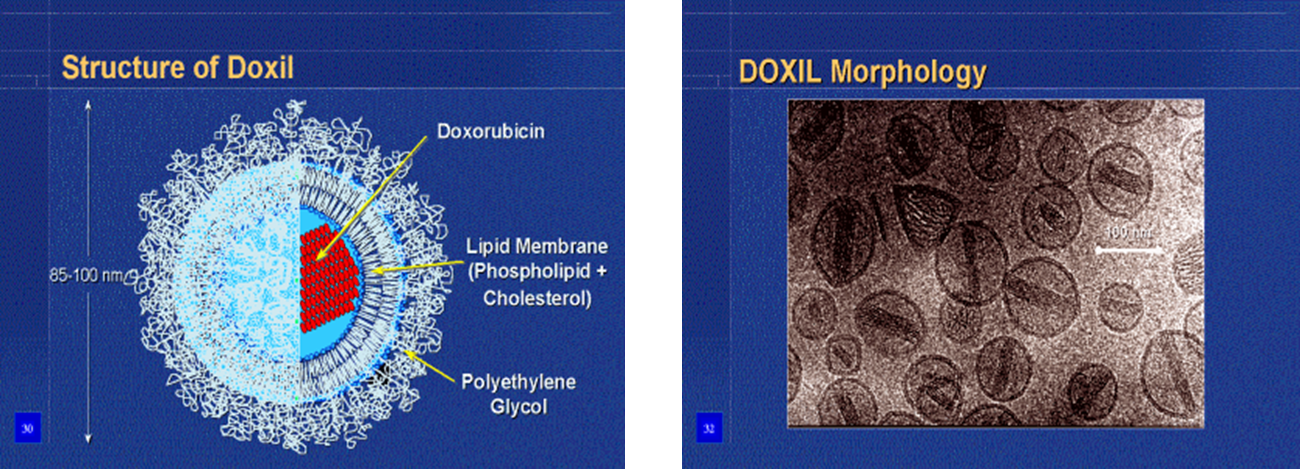
LONG-CIRCULATING LIPOSOMES FOR ANTICANCER DRUG DELIVERY
qLong-circulating liposomes have shown a size-dependent biodistribution when injected parenterally using animal models.
•
qVesicles smaller than 200 nm circulated freely in the blood whilst those larger than 300 - 400 nm were trapped by the spleen not the liver.
•
qLong-circulating liposomes encapsulating anticancer drug (e.g. doxorubicin) may extravasate and accumulate in the tumour interstitium.
LONG-CIRCULATING LIPOSOMES FOR ANTICANCER DRUG DELIVERY
qLiposomes which are coated with polyethylene glycol will have higher hydrophilicity of their surfaces and hence less opsonization and subsequently lower RES-mediated clearance.
qThis means longer circulation time of the liposomes (encapsulating the anticancer drug) and subsequently higher chances of liposome extravasation into tumour cells.
qTumours have Enhanced Permeability and Retention (EPR) effect compared to normal tissues due to the impaired lymphatic drainage in tumors.
LONG-CIRCULATING LIPOSOMES FOR ANTICANCER DRUG DELIVERY
qEnhanced permeability of tumor vessels
ØAngiogenesis
ØIncomplete formation of vessel walls and basement membrane
ØGaps and defects in endothelium
qEnhanced retention because of deficient lymphatic drainage
LONG-CIRCULATING LIPOSOMES FOR ANTICANCER DRUG DELIVERY
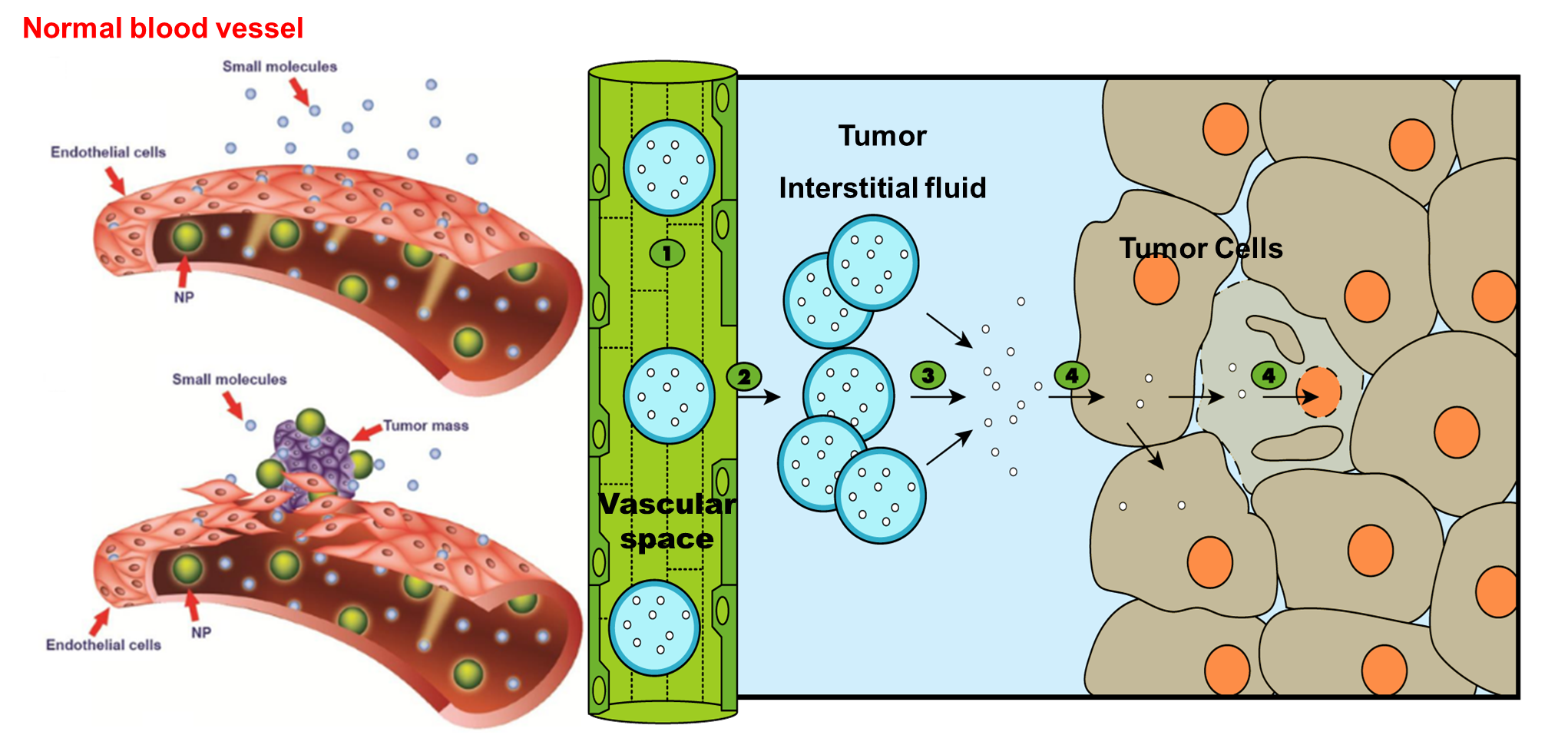
LONG-CIRCULATING LIPOSOMES FOR ANICANCER DRUG DELIVERY
Extravasation of STEALTH® Liposomes
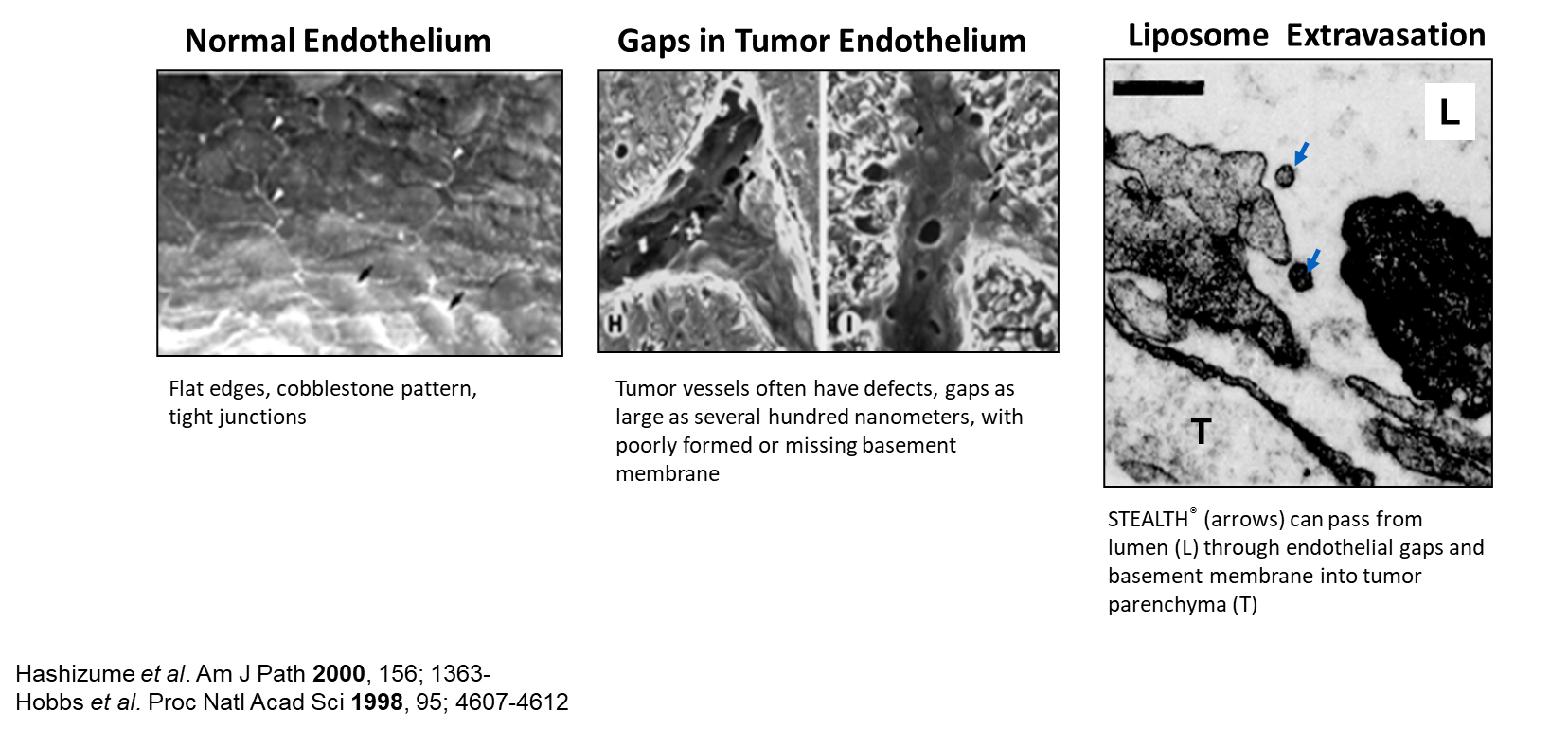
LONG-CIRCULATING LIPOSOMES FOR ANICANCER DRUG DELIVERY
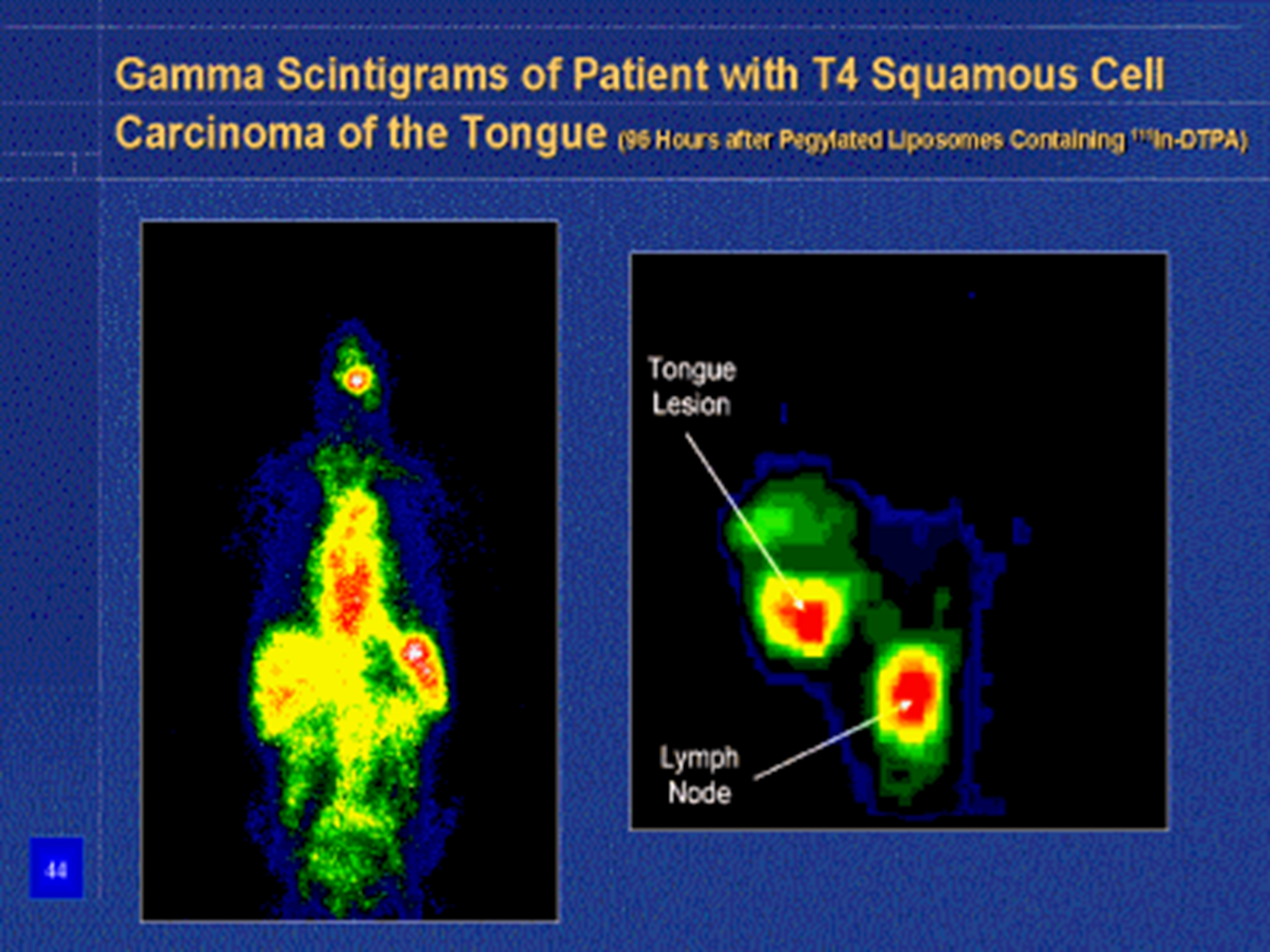
Doxorubicin accumulation in human prostate carcinoma Xenograft using DOXIL®
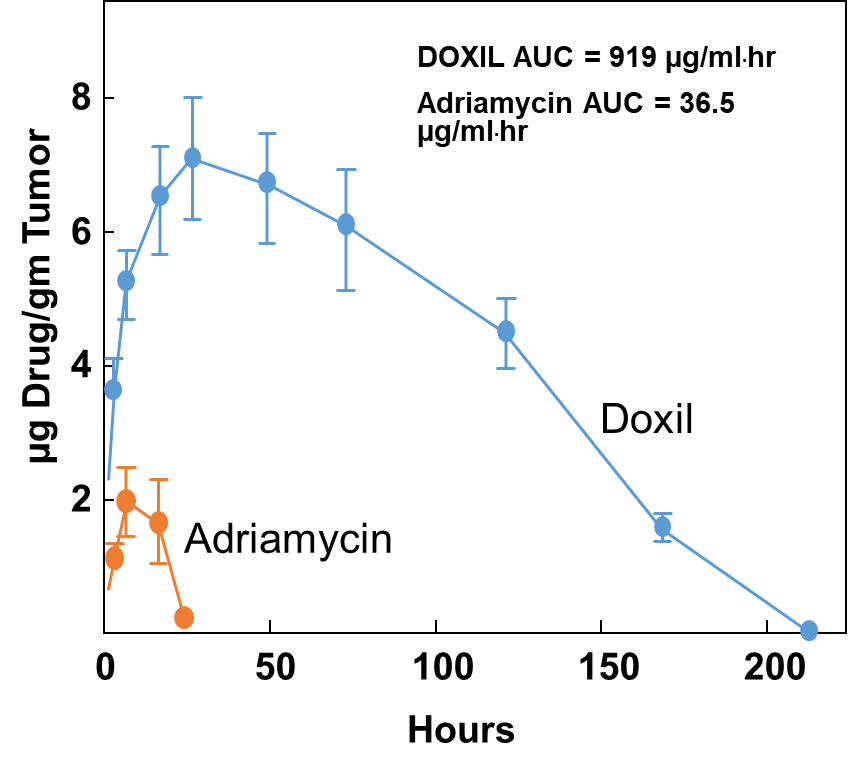
LONG-CIRCULATING LIPOSOMES FOR ANICANCER DRUG DELIVERY
Factors influencing delivery of anticancer liposomes to the tumor
qFormulation factors:
ØLiposome size
ØLiposome surface charge
ØLipid composition of liposomes (e.g. presence of cholesterol, PEGylation, etc.).
qBiological factors:
ØActivity of the RES
ØBlood perfusion into the tumor tissues
ØVascular permeability at the tumor site
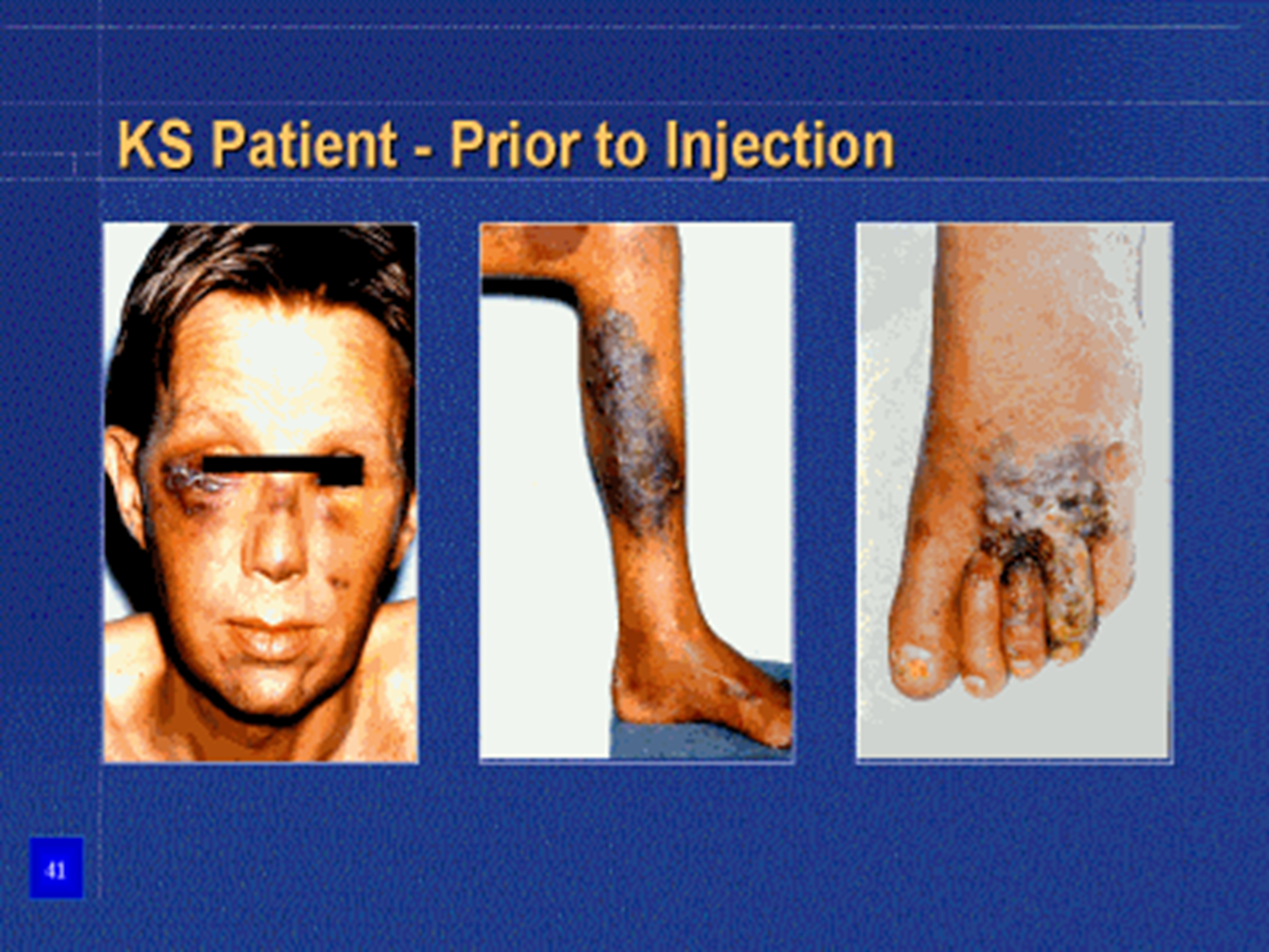
KS Patient - Prior to injection
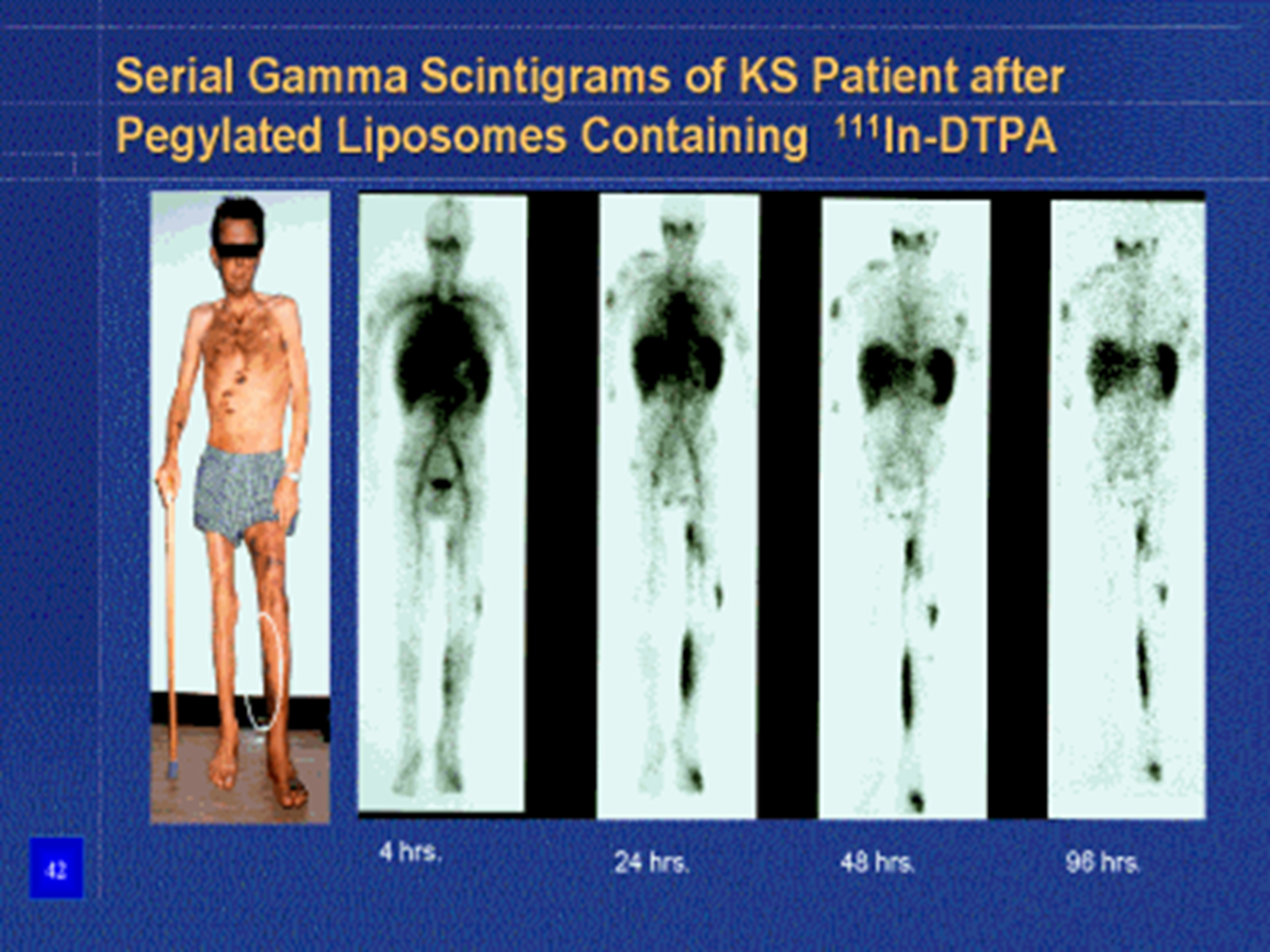
Serial gamma
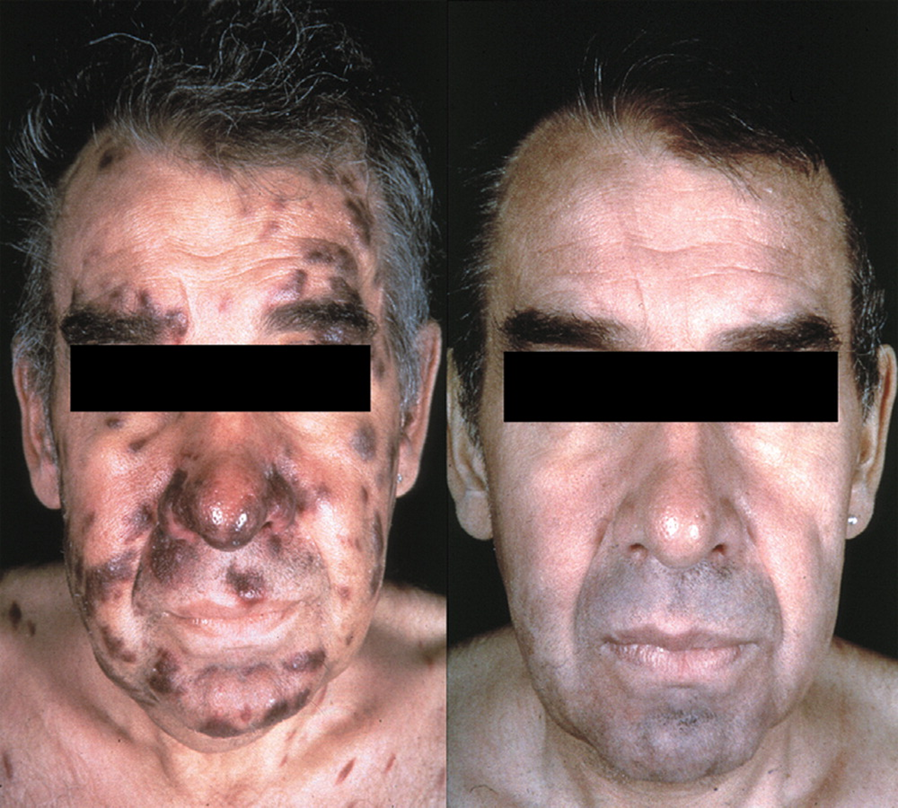
Patient with HIV related KS before and after Doxil treatment
Gamma scintigrams