VMPM 7437 Ruminants, Mastitis, Avian (Unit 4)
1/202
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
203 Terms
Contagious Pustular Dermatitis Etiology
Parapoxvirus → at least 6 different “strains”
Also known as Orf, contagious ecthyma, sore mouth and many other names
Contagious Pustular Dermatitis Epidemiology
Found worldwide in sheep and goat populations - ZOONOTIC
Transmitted via contact with scabs, exposure to contaminated facilities
Very stable virus, once a facility is contaminated often considered contaminated indefinitely
Especially wood and interior areas
Contagious Pustular Dermatitis Clinical Signs
Often 100% morbidity, low mortality
Endemic herds, youngstock only
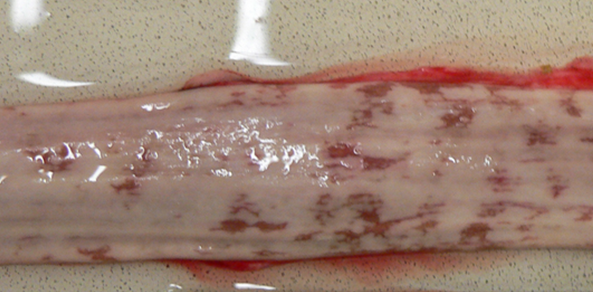
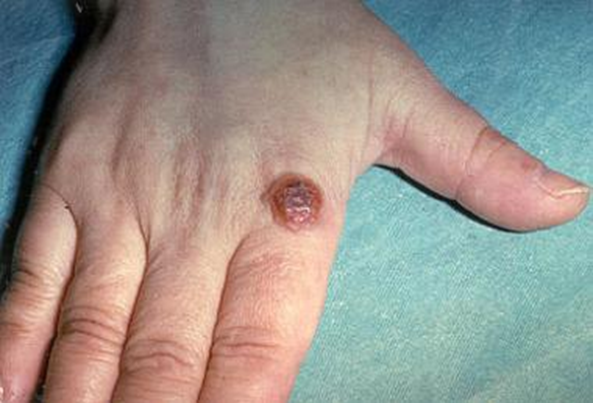
Scab-like lesions at mucocutaneous junctions
Lips
Oral cavity
Teats and udder
From nursing of affected lambs/kids
Very painful → refusal to allow nursing
Predispose to mastitis
Typical 2-3 week course of visible clinical disease
3-14 day incubation period followed by:
Papules → vesicles → pustules → scabs
Scabs heal and drop off in 1-4 wks


Rare potential for severe disease that persists or extends to respiratory or GI tracts
Humans – painful lesions
Contagious Pustular Dermatitis Diagnosis
Typically based on clinical signs
Bluetongue is a potential differential
FMD, sheep pox are FADs that could look similar at certain stages
Electron microscopy, PCR
Available from some VDLs
Contagious Pustular Dermatitis Treatment
Typically not necessary as infection is usually self-limiting
Use of topical ointments or astringents may actually delay healing
Neonates may require supplemental feedings, particularly if teat sores are concurrently present
Secondary bacterial infections can be treated with topical or systemic antibiotics
Gram positive skin infections (Staph sp)
Pneumonia
Contagious Pustular Dermatitis Prevention and Control
Quarantine and examine all animals entering farm
Avoid contact with shared feeders, waterers, trailers, grooming supplies at shows or sales
Isolate affected animals during outbreaks to prevent spread to naïve animals
Vaccination
LIVE VIRUS VACCINE available in US (ovine)
DO NOT USE IF VIRUS IS NOT ALREADY PRESENT ON FARM
ZOONOTIC RISK
Scarification of wool/hair free area followed by application of virus
Ringworm/Club Lamb Fungus Etiology
Trichophyton verrucosum, occasionally T. mentagrophytes ZOONOTIC
Ringworm/Club Lamb Fungus Epidemiology
Infected animals contaminate facilities and equipment, perpetuates in herd
Animals that are housed indoors, in winter most affected
Sheep that go to shows (club lambs) are clipped and bathed regularly
Removes protective lanolin
Creates small wounds that can serve as entry point for organism
Ringworm/Club Lamb Fungus Clinical Signs
Thick, scaly round spots that can become ulcerated or crust over
Spontaneous recovery typically in 8-16 weeks


Ringworm/Club Lamb Fungus Diagnosis
Clinical signs are highly suggestive
Treat as if positive until proven otherwise
Differentials:
Dermatophilosis, external parasites, zinc deficiency Staphylococcal dermatitis
Fungal culture (DTM)
KOH prep
Ringworm/Club Lamb Fungus Treatment
Sunshine + time
All treatments are extralabel and withdrawal times are rarely known so should be used with caution.
Thiabendazole is probably the best product legally as there are veterinary products available; recently sold and not all products may still be available.
Fulvicin (griseofulvan; human antifungal) (10mg/kg) is effective but is extra label. No data on withdrawal times have been published.
Over the counter human products like Tinactin (tonaftate antifungal) have also been used.
Captan (plant fungicide). Has been used topically by mixing with water to make a paste. No withdrawal time has been published and one needs to wear gloves and be careful with the Captan. NOT CURRENTLY RECOMMENDED – EPA product, no extralabel use!!
Ringworm/Club Lamb Fungus Prevention and Control
Isolate affected animals to prevent spread, disease is self-limiting in most cases
Do not allow animals with lesions to attend shows and events!
Sanitize equipment between animals/farms
Do not share grooming equipment at shows and events!
Pediculosis (lice) Etiology
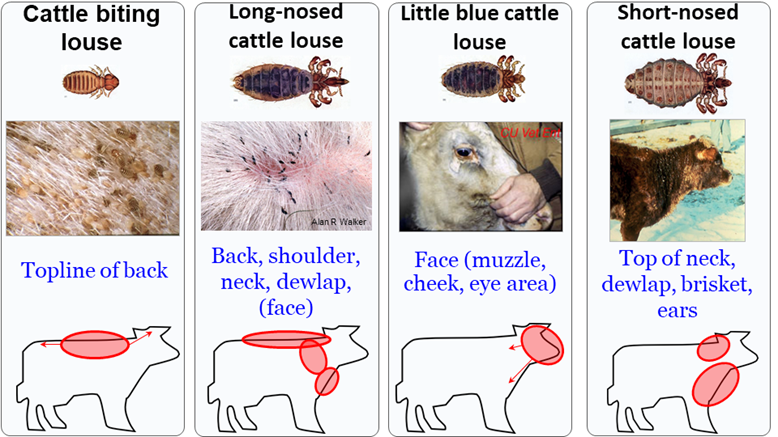
Bloodsucking (Linognathus sp. Solenopotes sp. Haematopinus sp.)
Chewing or biting (Bovicola [formerly Damalinia] sp.
Most are host specific, so unique species of each effect cattle/goats >> sheep
Pediculosis (lice) Epidemiology
Obligate ectoparasites
Most transmission is through direct contact, limited survival in environment and on grooming supplies, etc
Pediculosis (lice) Clinical Signs
Pruritus with a rough, shaggy hair coat, +/- weight loss, decreased production efficiency
Sucking lice may cause anemia, hypoproteinemia, and death – especially when combined with intestinal parasitism
Most commonly observed in late winter

Pediculosis (lice) Diagnosis
Visible via the naked eye
Collection and viewing under a microscope allows determination of sucking vs biting species which can be important for directing therapy
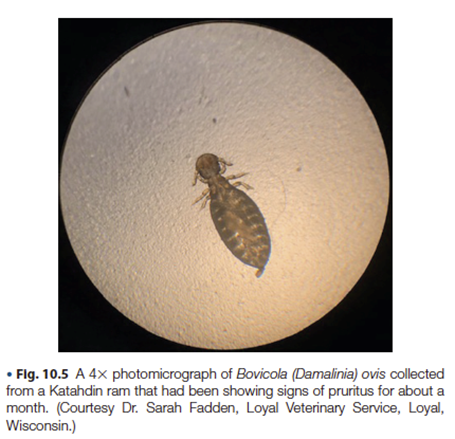
Pediculosis (lice) Treatment and Prevention
Treatment of entire herd with EPA approved insecticide (no extralabel use allowed) for chewing and bloodsucking lice
Most treatments are not ovicidal and thus treatment should be repeated in at least twice in 2-week intervals.
Exception: Clean-up II with IGR
Bloodsucking lice can be treated with injectable deworming products such as the avermectins
Treatment and isolation of new herd additions will prevent herd entry

Mange Etiology
Chorioptic mange: Chorioptes bovis/ovis/caprae – most common mange mite in US; host specific
Psoroptic mange: Psoroptes ovis (cattle); Psoroptes cuniculi (goats, sheep), P. communis var ovis (sheep) - rare, reportable in US; host specific
Demodectic mange: Demodex caprae/ovis – may be common, mostly immunocompromised
Sarcoptic mange: Sarcoptes scabiei var bovis/ovis/caprae – reportable, zoonotic
Mange Epidemiology
Transmission occurs through direct contact or indirectly through contact with equipment or grooming tools
Mange Clinical Signs
Pruritus, crusting, self-trauma and alopecia
Different types have different preferred patterns:
Chorioptic mange tends to affect the hindlimbs, lower, limbs, scrotum and abdomen
Psoroptic mange tends to affect the back and flanks of cattle, trunk in sheep and ears in goats.
Demodectic mange tends to affect the face, shoulders and trunk
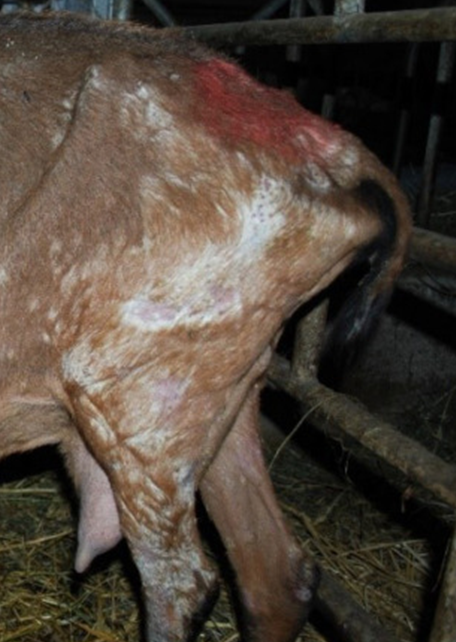
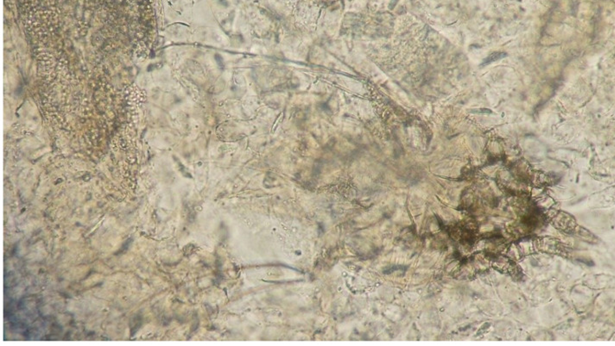
Mange Diagnosis
Skin scrapings viewed under a microscope are necessary for diagnosis
Mange Treatment and Prevention
Treatment of entire herd with injectable or topical deworming products such as the avermectins, repeated in at least twice in 2 week intervals
Lime sulfur and other (coumaphos, toxaphene, phosmet) dips, caution with establishing withdrawal times
Treatment and isolation of new herd additions will prevent herd entry
Foot and Mouth Disease
Cattle | Swine | Sheep/Goats |
Disease Indicators | Amplifying Host | Maintenance Host |
Often first species to show signs | Produce large amounts of aerosolized virus | |
Can carry the virus for up to 6 months Some animals may remain infected for up to 3-1/2 years | Rare, | Can carry/shed the virus for up to |
FMDV carriers are defined as animals in which the virus can be found for more than 28 days after infection. Animals can become carriers whether or not they had clinical signs. How long an animal can remain a carrier of the virus varies with the species. Most cattle carry FMDV for six months or less, but some can remain persistently infected for up to 3.5 years. Cattle are considered indicator hosts because they are often the first species to demonstrate clinical signs. Pigs are not thought to become carriers; however, they are considered amplifying hosts, as they produce large amounts of aerosolized virus. The virus is shed for a short time and swine are not considered long-term carriers; there have been a few reports documenting the presence of viral nucleic acids after 28 days. Sheep and goats are considered maintenance hosts and may shed the virus for up to 12 months in sheep and up to 4 months in goats.
Foot and Mouth Disease DDx in ruminants
DDX for Foot and Mouth disease in ruminants:
Vesicular stomatitis
Malignant catarrhal fever (cattle only)
Infectious bovine rhinotracheitis (cattle only)
BVD mucosal disease (cattle > sheep/goats)
Bluetongue (sheep/goats > cattle)
Epizootic hemorrhagic disease (cattle)
Contagious pustular dermatitis/contagious ecthyma (sheep/goats)
Bovine papular stomatitis
Rinderpest (eradicated)
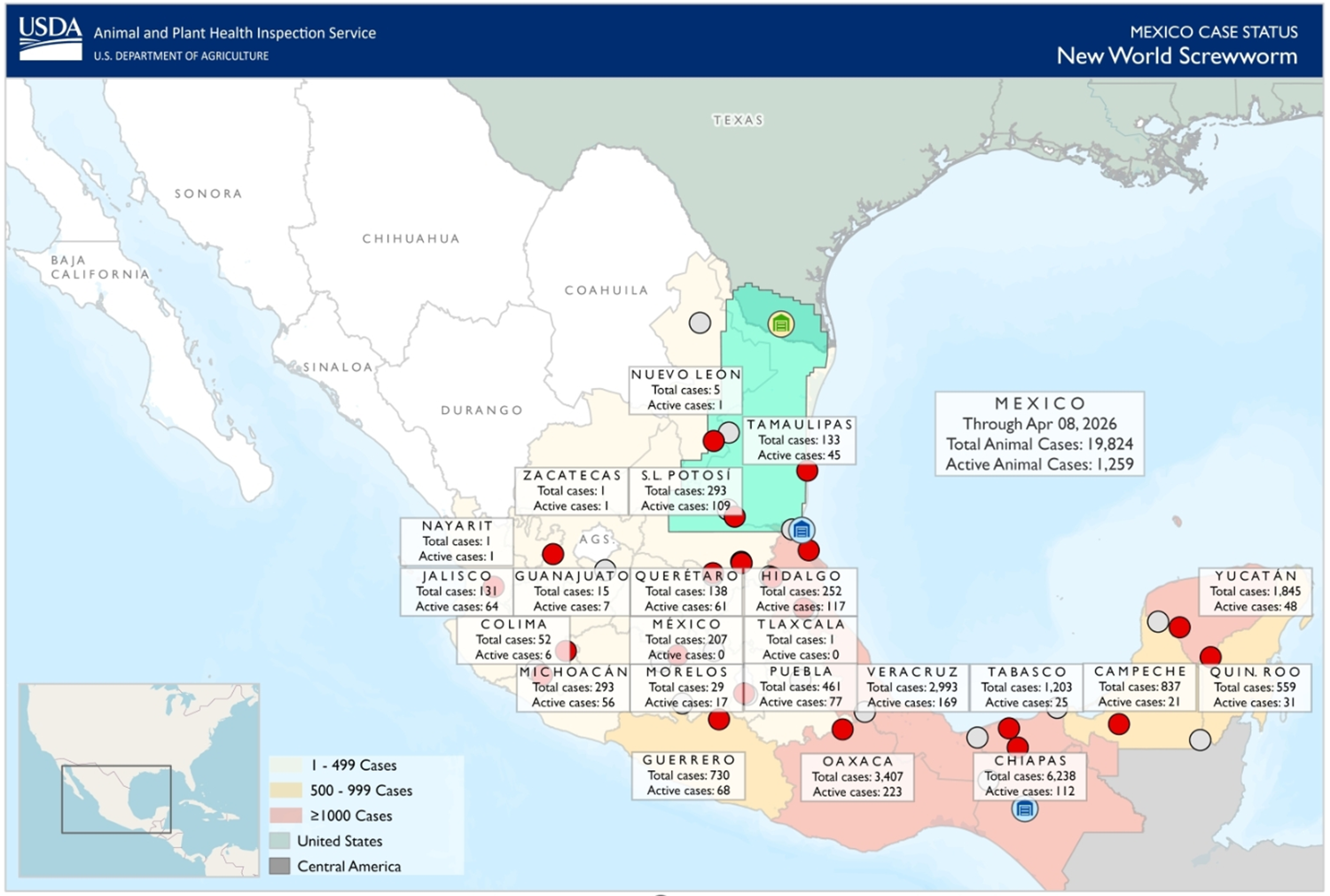
New World Screwworm Etiology
Cochliomyia hominivorax
New World Screwworm Epidemiology
Can affect all warm-blooded animals
Previously present in the US (pre-1970), eradicated through extensive control efforts including a permanent sterile fly barrier established between Panama and Columbia.
Recently paused and moved north into Mexico
An outbreak occurred in the Florida Keys (Key deer) in 2016 but was quickly eradicated.
Recently has been identified within 400 miles of Texas in Mexico prompting closure of US:Mexican border to all livestock as of May 2025.
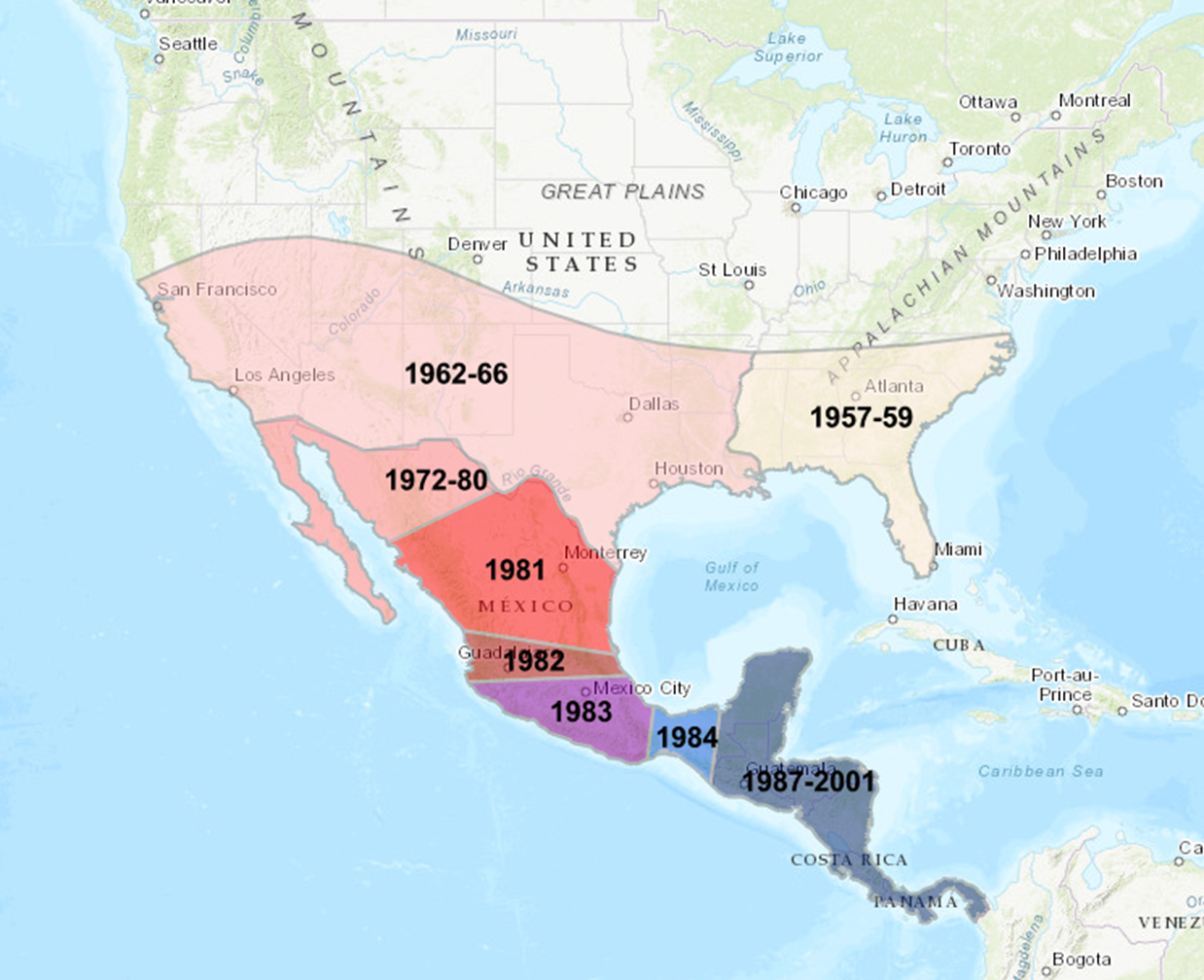
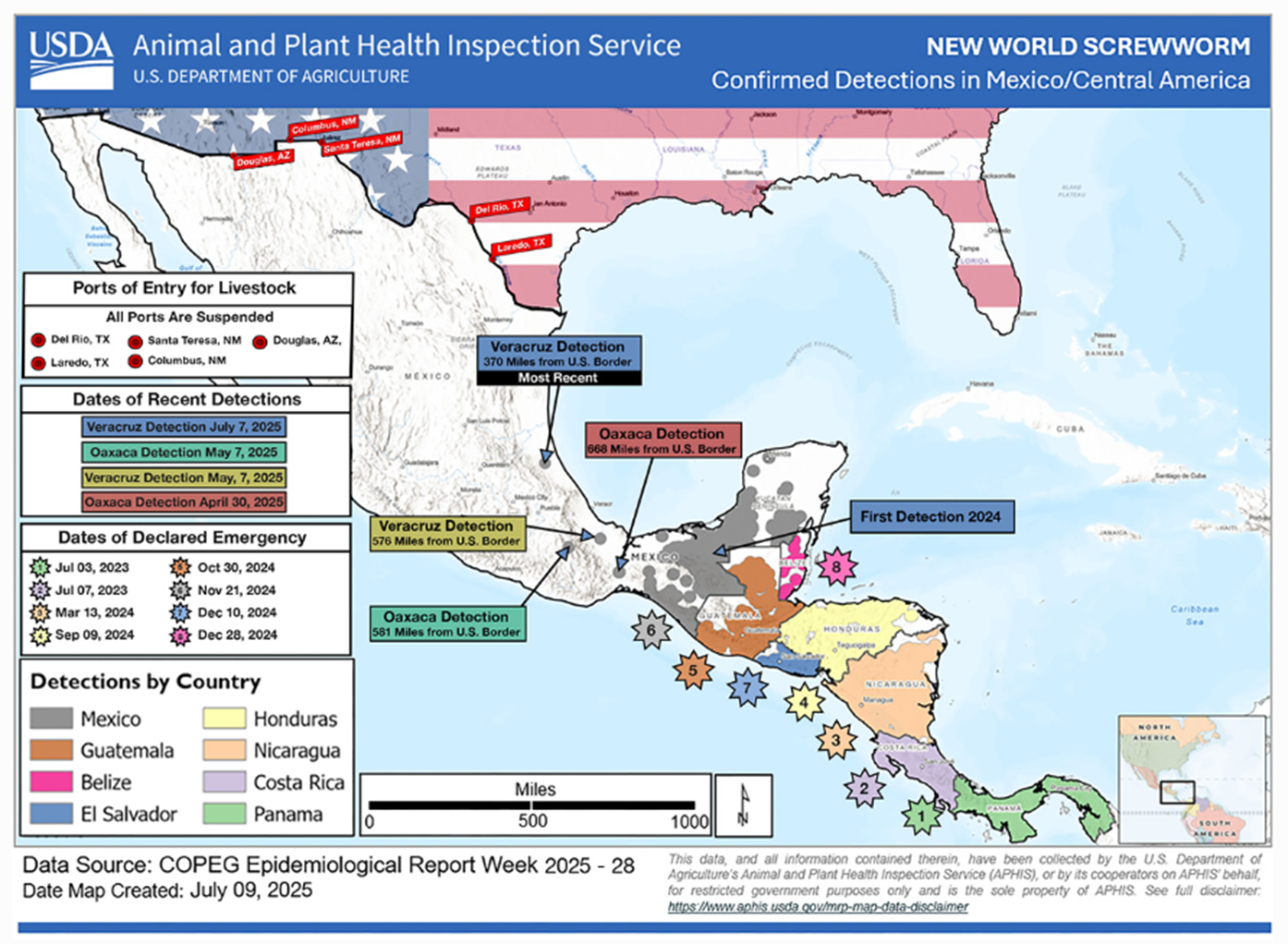
New World Screwworm Clinical Signs
Flies are attracted to wounds.
Animals that have recently given birth or were born, been injured or had a surgical procedure (castration, dehorning, branding) are most at risk.
The fly larvae (maggots) burrow into the living tissue causing very painful lesions (myiasis) that can lead to fatal outcomes.
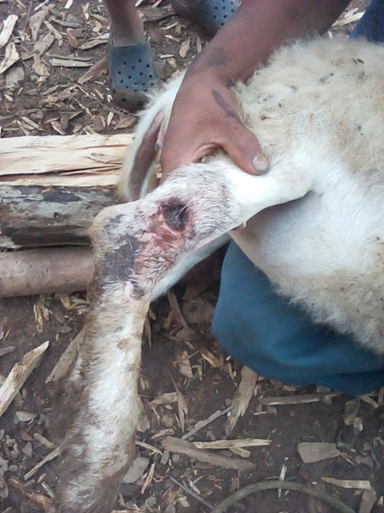

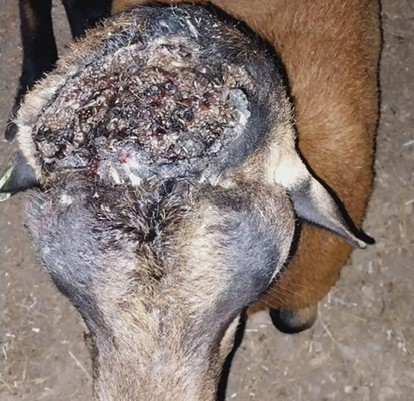
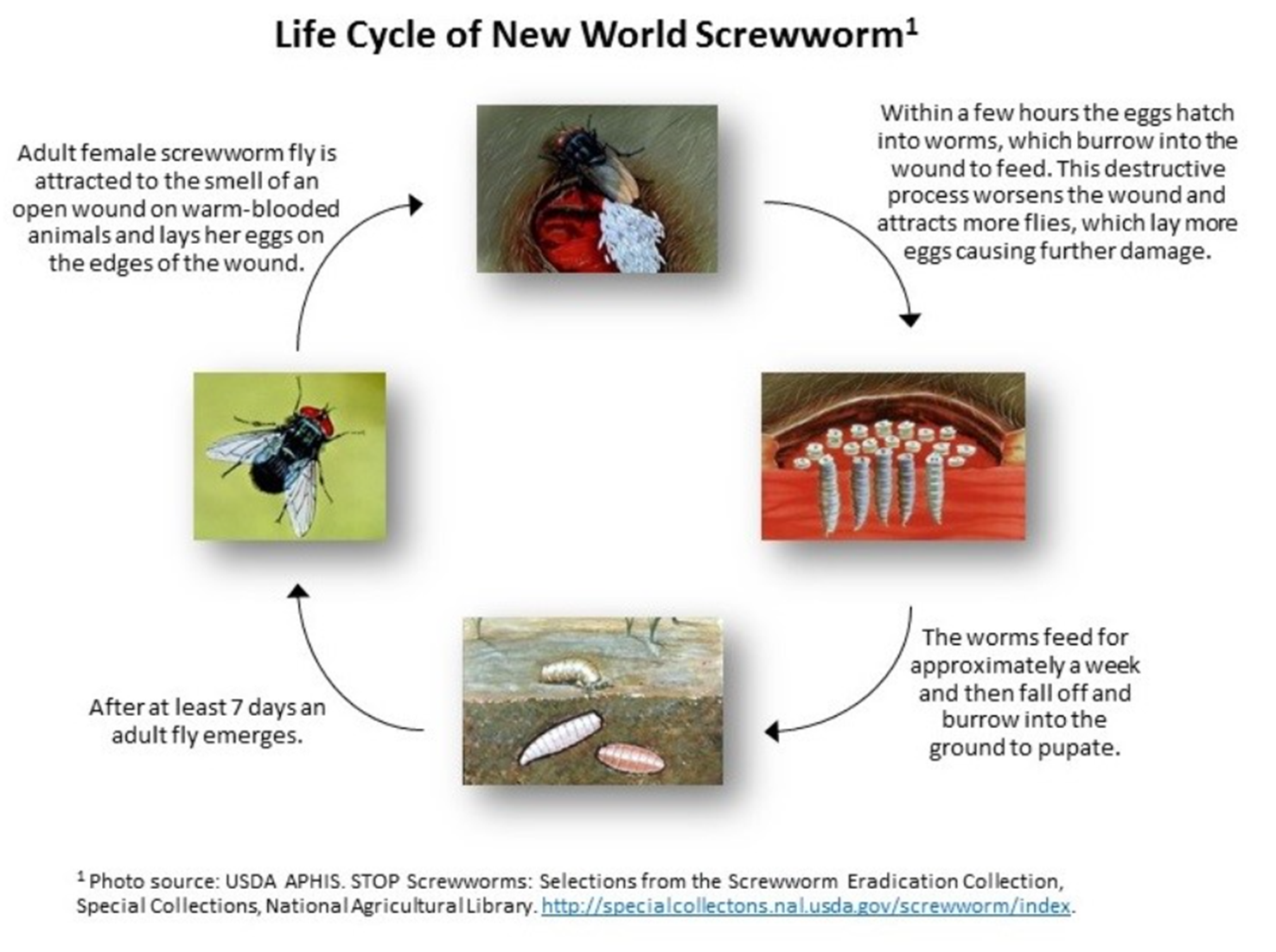
New World Screwworm Diagnosis
Identification of maggots in wounds or other body openings, especially wounds with bloody discharge, foul odor, and those that become deeper and larger overtime.
Adult screwworm flies are about the size of a common housefly (or slightly larger).
Orange eyes, a metallic blue or green body, and three dark stripes along their backs.
Flies, eggs, and maggots are collected and placed in 70% alcohol for identification.


New World Screwworm Treatment
Clean affected area and remove all visible larvae; apply topical antiseptics to treat secondary infections.
Apply topical or systemic antiparasitic treatments to kill remaining larvae.
F10 Antiseptic Wound Spray with Insecticide (benzalkonium chloride, polyhexanide, cypermethrin) – EUA for prevention and treatment
Exzolt Cattle-CA1 topical solution (fluralaner) – conditional approval (also for cattle fever tick) for prevention and treatment
Ivermectin injectable (OTC) – EUA for prevention
New World Screwworm Prevention
Sterile fly eradication program – female flies mate only once, so breeding to sterile males slowly leads to eradication.
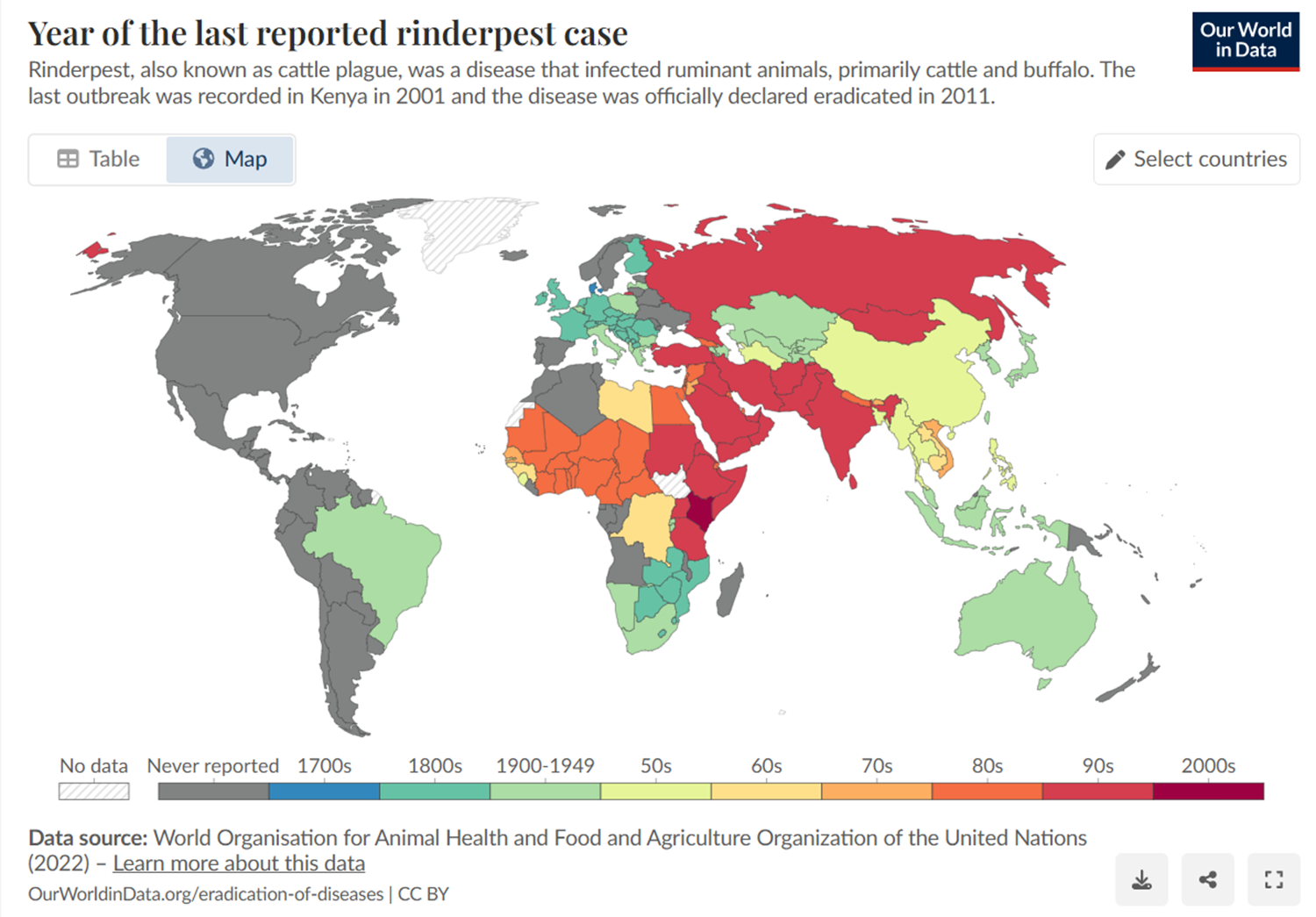
Rinderpest Etiology
Morbilivirus, closely related to measles, canine distemper, PPR
Rinderpest Epidemiology
ERADICATED!!
Considered the deadliest disease of cattle in history
WOAH-listed disease – must be reported
Previously endemic in Africa, Asia, with frequent devastating outbreaks in Europe

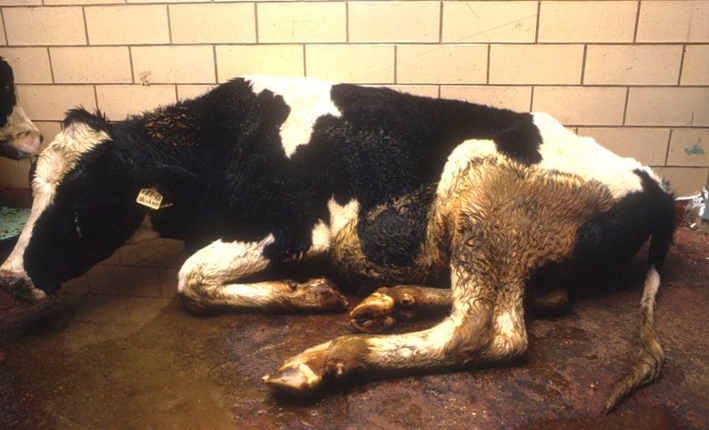
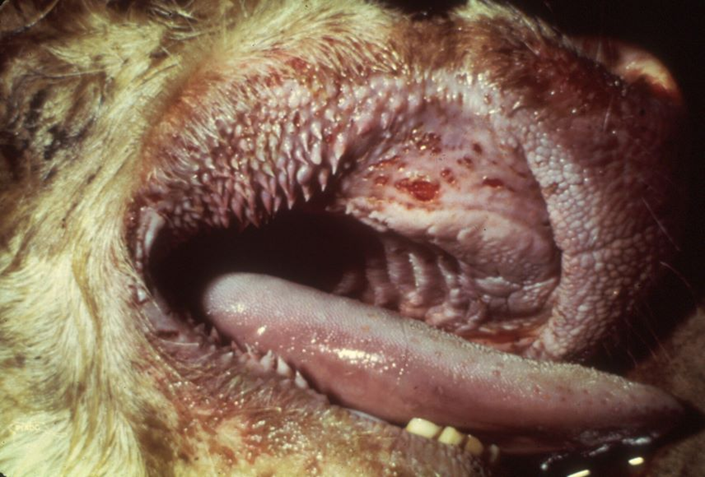
Rinderpest Clinical Signs
Explosive outbreaks with whole herds affected
Four “Ds” = depression, dehydration, discharge, diarrhea
Necrosis and erosions of the GI and upper respiratory tracts
Death in European breeds
Some native breeds more resistant
Rinderpest Diagnosis
Serology, RT-PCR
Rinderpest Prevention
Eradication was achieved because:
Transmission required close contact, did not persist in environment
No carrier state
High quality vaccine (live attenuated) with life-long immunity
Heat stable, did not require cold chain
No longer in use
Effective diagnostic assays
Global effort including strict quarantine and movement controls
Contagious Bovine Pleuropneumonia Etiology
Mycoplasma mycoides subspecies mycoides small colony
Contagious Bovine Pleuropneumonia Epidemiology
Endemic with frequent outbreaks in sub-Saharan Africa; also Asia, India
Eradicated from North America, Europe, Austrailia
The organism is highly contagious and is spread by inhalation of droplets coughed up by infected cattle
About 25% of recovered cattle become chronic carriers of the organism which are difficult to detect in an infected herd
Transplacental transmission can occur
Contagious Bovine Pleuropneumonia Clinical Signs
Rapid disease progression with very painful, difficult breathing (usually rapid and shallow), fever up to 107 F
Some animals will become recumbent and die within 1-3 weeks.
Marbled appearance of the lungs due to thickening of the interlobular septa; fluid (up to 10 L) in the thoracic cavity; thick layer of fibrin.
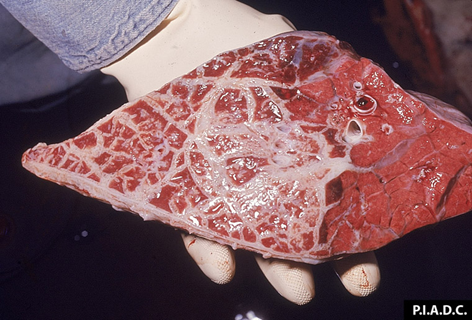
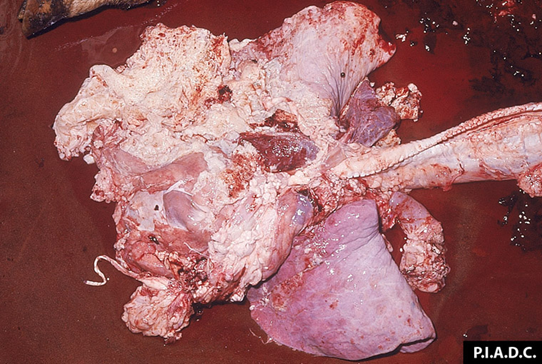
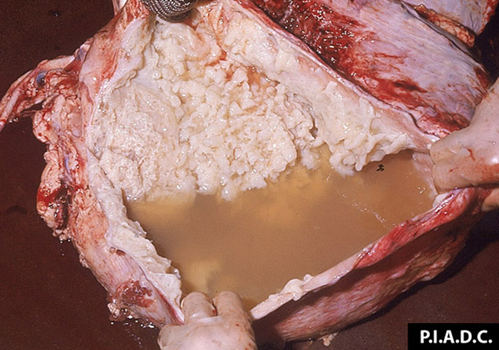
Contagious Bovine Pleuropneumonia Diagnosis
Serology (CFT, cELISA) – often used for international trade
PCR, Culture
Contagious Bovine Pleuropneumonia Treatment
WOAH (OIE) reportable disease. Slaughter of all infected and exposed animals (was used to eliminate from the US).
In endemic areas, macrolides and fluoroquinolones are reported to be effective. Treated animals may remain carriers.
Contagious Bovine Pleuropneumonia Prevention and Control
Vaccination (attenuated live – T1/44 strain) is practiced in endemic areas.
Trade restrictions for endemic regions
Trypanosomiasis Etiology
Cattle, sheep, and goats are infected, in order of importance, by Trypanosoma congolense, T. vivax, and T. brucei brucei.
T. brucei rhodesiense and T brucei gambiense are zoonotic, with people as the predominant host.
Trypanosomiasis Epidemiology
Group of protozoal diseases transmitted by tsetse flies, “sleeping sickness”
Tsetse flies are restricted to Africa, and several different species exist with specific geographic range and preferred mammals on which to feed.
Some mechanical transmission also occurs through other biting flies, especially outside tsetse-endemic areas such as in Central and South America.
Trypanosomiasis Clinical Signs
Localized skin infections (chancres) occurs at the location of the tsetse bite; the incubation period is typically 1-4 weeks.
Primary clinical signs include intermittent fever, weight loss, enlarged lymph nodes, diarrhea and anemia.
Chronic disease often leads to high mortality especially if poor nutrition or stress is present.
Trypanosomiasis Diagnosis
Demonstration of trypanosomes in stained blood smears.
Serology; PCR also possible but not commonly available in endemic areas.
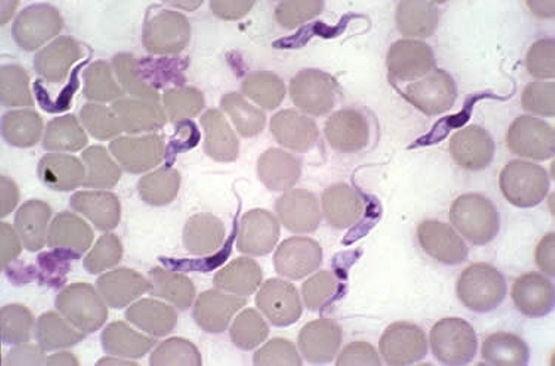
Trypanosomiasis Treatment
In endemic areas, diminazine aceturate or homidium bromide can be curative
Trypanosomiasis Prevention and Control
Breeding of cattle breeds with innate resistance (indigenous West African breeds)
Control of tsetse flies (sprays, dips), sterile fly release
Prophylactic treatment
Peste Des Petits Ruminants (PPR) Etiology
Morbilivirus, closely related to Rinderpest (eradicated)
PPR Epidemiology
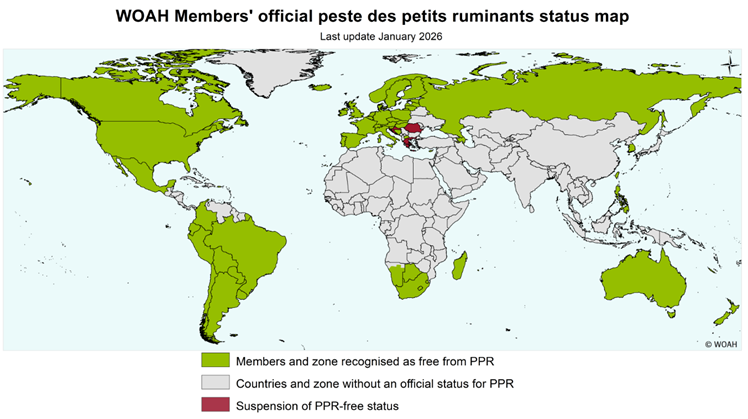
WOAH-listed disease – must be reported
Africa, Middle East, Turkey, Asia, India
Recent outbreaks in eastern Europe (2024-current)
Hungary* (back to free)
Croatia, Romania, Greece (no longer free)
Kosovo, Albania
Vietnam – October 2025
Transmission occurs rapidly via airborne droplets or direct contact, or contact with contaminated environments
Explosive outbreaks, 2-6 days incubation
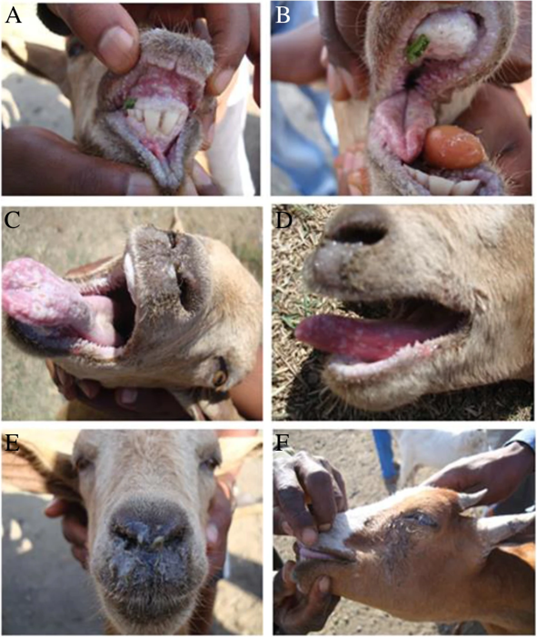
PPR Clinical Signs
Targets lymphoid tissue and epithelial lining of intestines
High fever, depression, mucopurulent ocular and nasal discharge
Extensive erosions of oral/pharyngeal mucosa, enteric lesions (“zebra stripes), profuse diarrhea
Asymptomatic possible but rare
PPR Diagnosis
Clinical signs and post-mortem findings highly suggestive
Antigen capture ELISA, PCR, IHC all possible to ID
Serology also used
Can’t differentiate between Rinderpest
Useful in vaccination/eradication programs
PPR Treatment and Prevention
Non-endemic areas:
Import restrictions from endemic areas
Rapid reporting to animal health officials
Quarantine of affected animals
Test and culling of any affected or exposed animals or total depopulation may be required
Endemic areas
Vaccination is used to control disease
FAO/WOAH Eradication goal by 2030
Only 1 serotype
No carrier state or reservoir outside of domestic small ruminants
High quality vaccine with life-long immunity
Requires cold chain
Goal of 100% vaccination of >3mo old, 70% herd immunity
Effective diagnostic assays
Contagious Caprine Pleuropneumonia (CCPP) Etiology
Mycoplasma capricolum subsp. capripneumoniae
CCPP Epidemiology
WOAH-listed disease – must be reported
Occurs in Africa, Asia, Middle East
Has never been reported in North or South America
Goats >> sheep
Wild ruminants?
Transmission occurs rapidly via airborne droplets or direct contact, or contact with contaminated environments
Explosive outbreaks, 6-10 days incubation
CCPP Clinical Signs
Severe respiratory infection – coughing, dyspnea, frothy nasal discharge
High fever, depression, anorexia
Up to 80% mortality
Survivors have chronic cough, nasal discharge, debilitation
CCPP Diagnosis
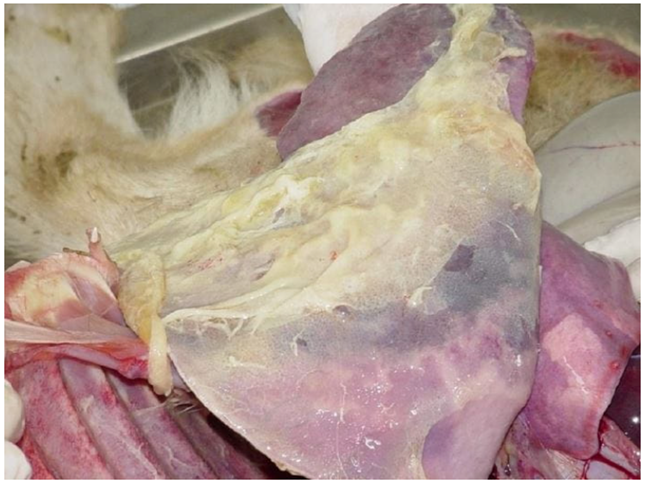
Severe lung lesions present on necropsy
Serofibrinous pleuropneumonia
Consolidated, hepatized lung
Granular texture
Culture can be challenging
PCR, IHC preferred
Serologic testing to identify chronic disease
CCPP Treatment and Prevention
Antibiotic therapy as for typical Mycoplasma infections can be effective but may not eliminate carrier status
Supportive care, most acute cases die
Non-endemic areas:
Import restrictions from endemic areas
Rapid reporting to animal health officials
Quarantine of affected animals
Test and culling of any affected or exposed animals or total depopulation may be required
Endemic areas
Vaccination is used to control disease
Mastitis
A disease with a high component of management
Cost $13 billion worldwide
U.S. Importance
Over $2 billion annually
Thought to be the most costly problem in the dairy industry
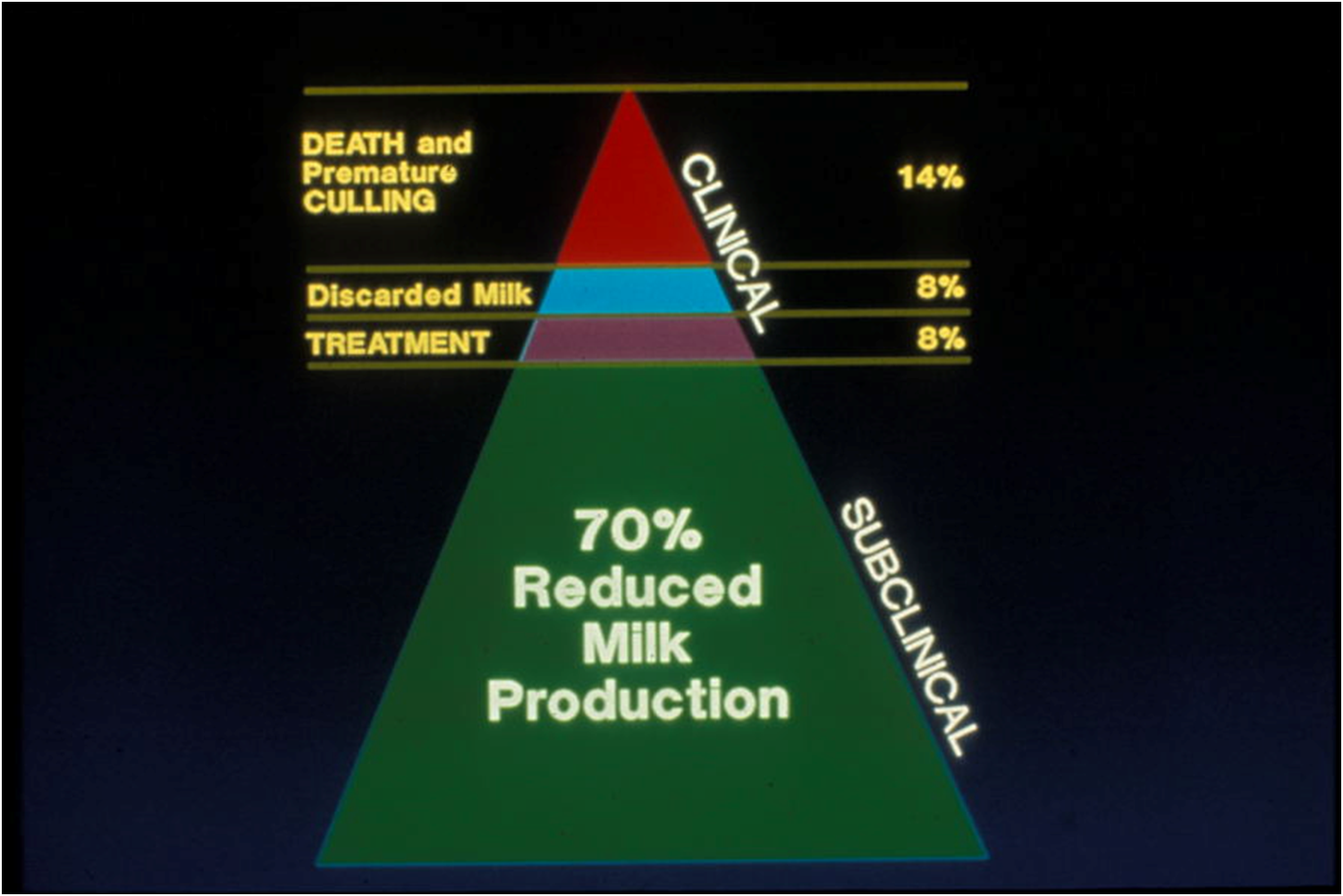
Cost of clinical vs. subclinical mastitis
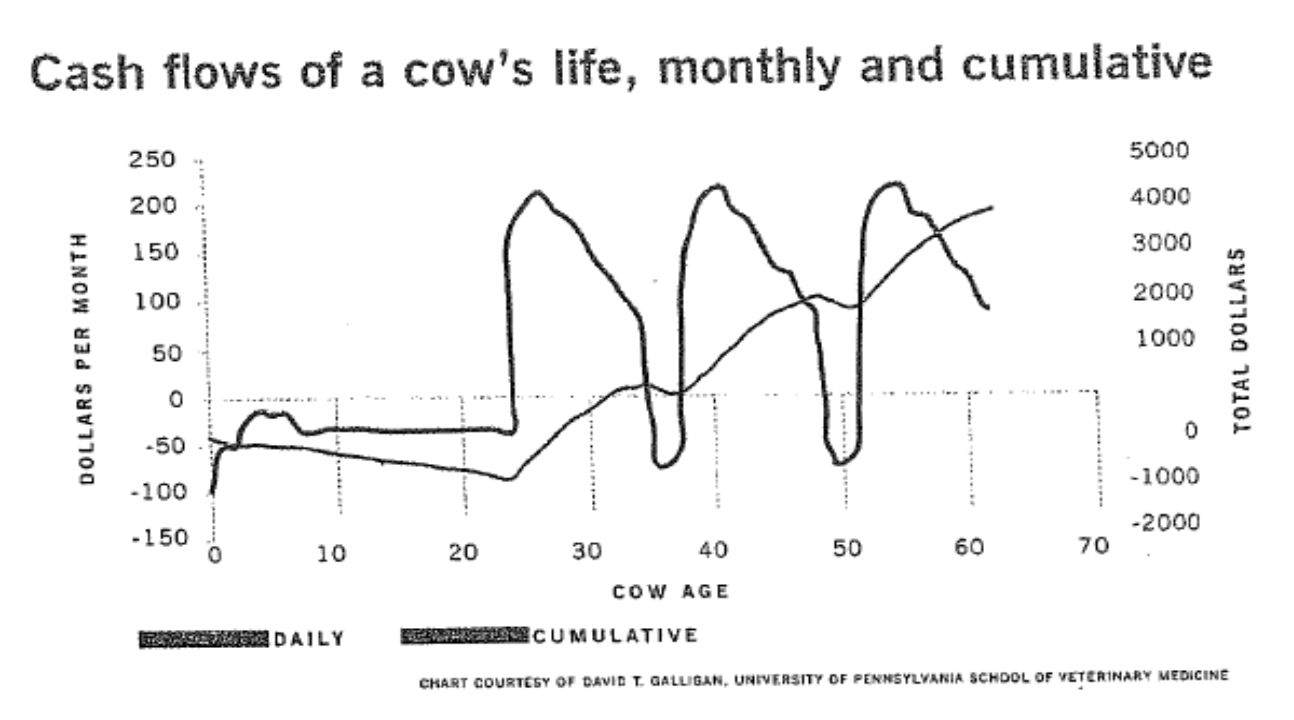
Culling Decisions
Approximately 25% of all culls leave the herd in the first 60 days of lactation
Cows tend to become invisible until they get sick or are culled for low production
True cause for low production may not be discerned and/or recorded
Adequate bunk space, nutrition, sanitation, cow comfort, proper body condition, etc. all play a role in cow longevity
Mastitis Severity Scores: Better System for Records?
Milk is abnormal
Milk and udder are abnormal
Cow is sick with mastitis
Dairy Herd Improvement Association (DHIA) → records “Times Severe” on records
Importance of Mastitis
Loss of production
Loss of financial incentives
Poor quality milk (high leukocyte counts)
Animal Welfare
Painful condition
Toxic mastitis and down cows
Nonspecific Mammary Gland Defense Mechanisms
Anatomical
Smooth muscle → surrounds the teat duct
Keratin → forms a plug
Some high producing cows do not form a good keratin plug
Contains bactericidal fatty acids
Defensins
Also can harbor bacteria
Milk flow
Flushes out pathogens and toxins
Incomplete milk-out can lead to mastitis
Milk leukocytes
Highest in foremilk and stripping milk
Neutrophils → predominant cell in infection
Killing function → Inhibited by fat globules (butterfat)
Milk neutrophil ingested fat droplets
Neutrophil inability to function well in milk
Lack of glucose (lactose is the main milk sugar)
Decreased glycogen
Deficiency of opsonins (complement and antibody)
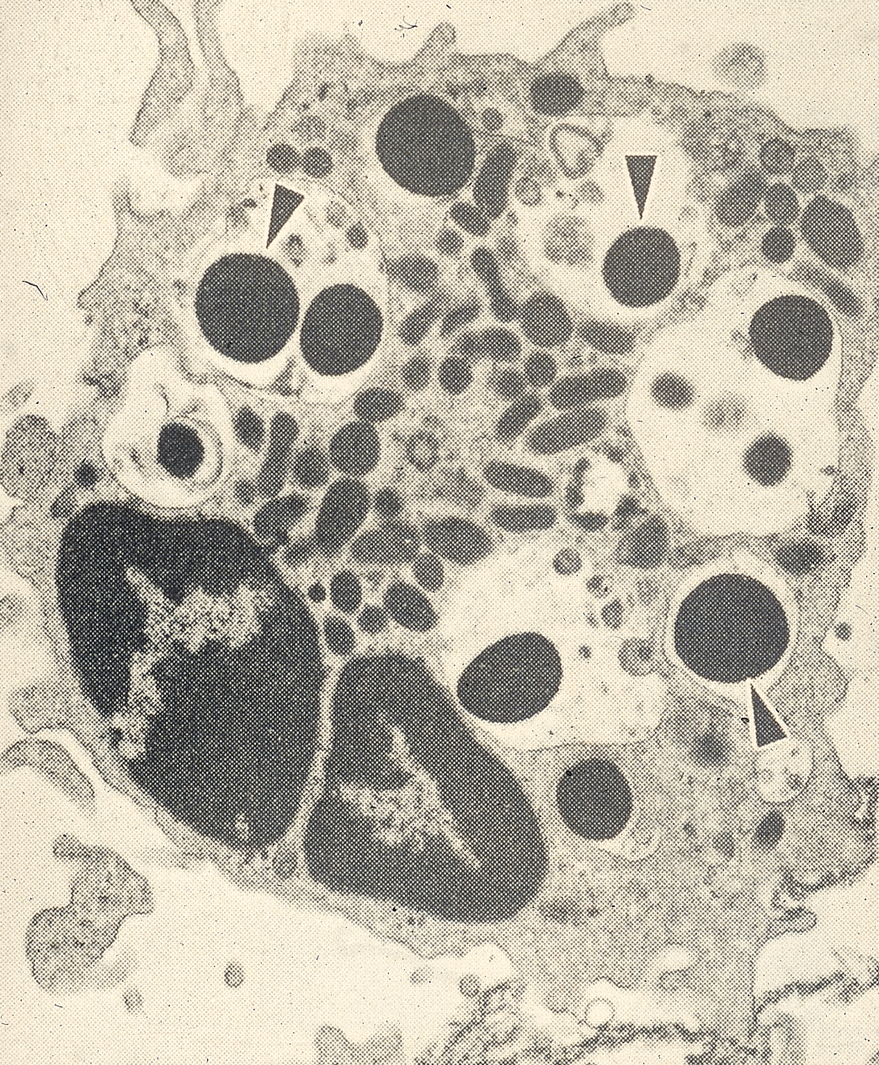
Nonspecific soluble factors
Lysozyme
Breaks down bacterial peptidoglycan
Active only against Gram positives
Low in normal milk
Increases during infection
Lactoferrin
Chelates iron needed by bacteria
Low in normal milk
High in non-lactating (involuted) gland
Defensins
Antibacterial proteins
Produced by a variety of cells (epithelial and neutrophils are especially good producers)
Involution
365 days in a year, 305 days in an average lactation, 60 days of dry period
Reason for dry period: allow regeneration of mammary epithelium
Should aim for 45-60 days
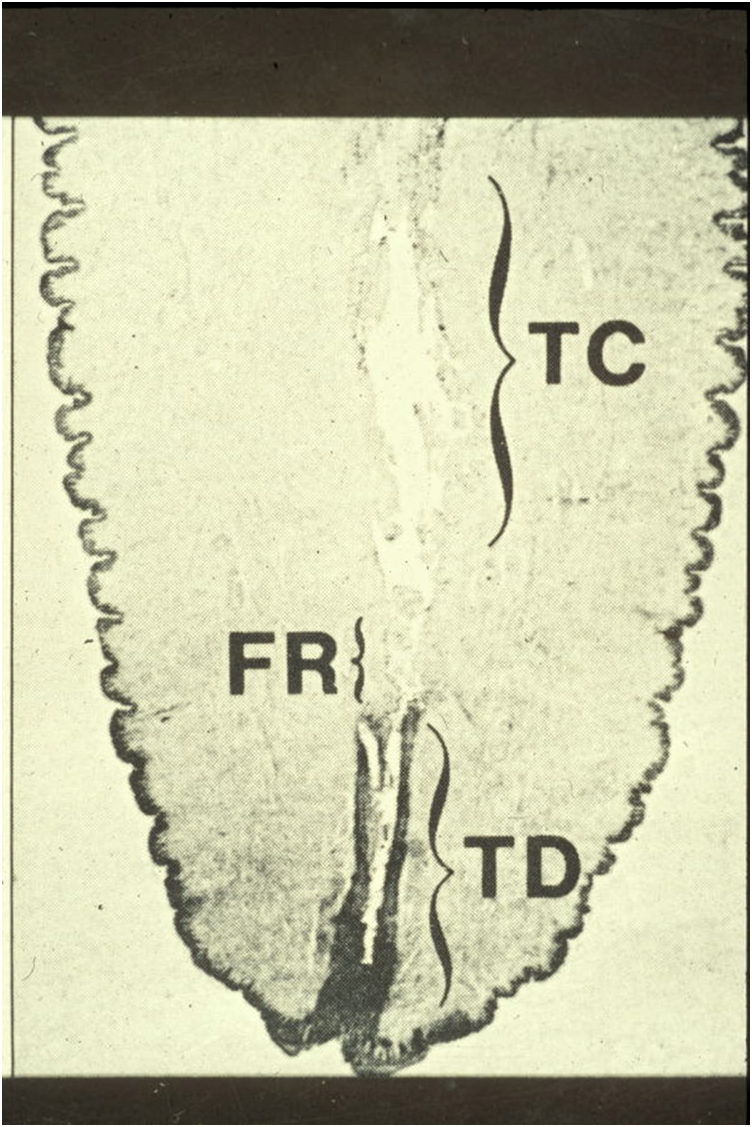
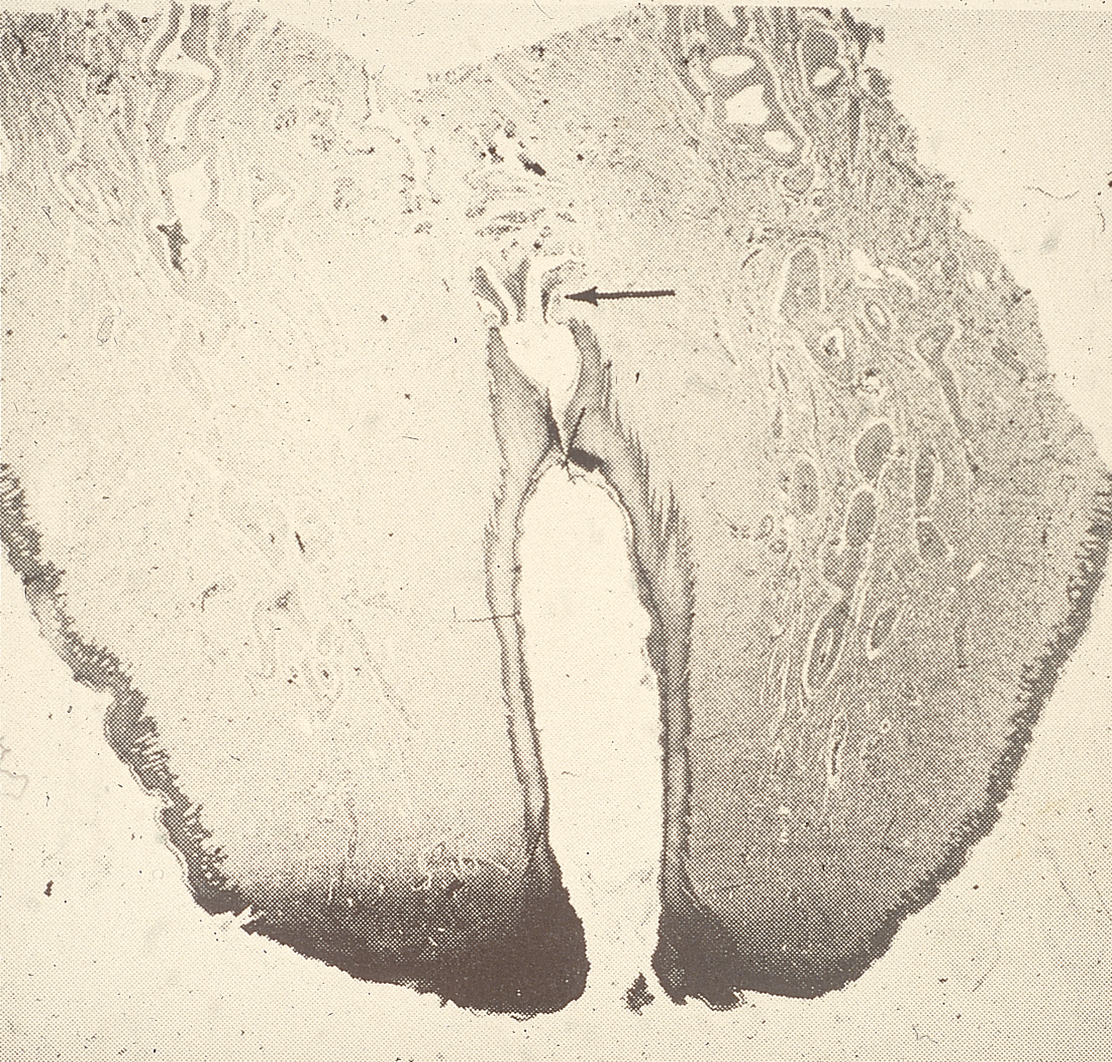
Involution occurs usually in the first 2 weeks of the dry period. Common time for new infections
Diet
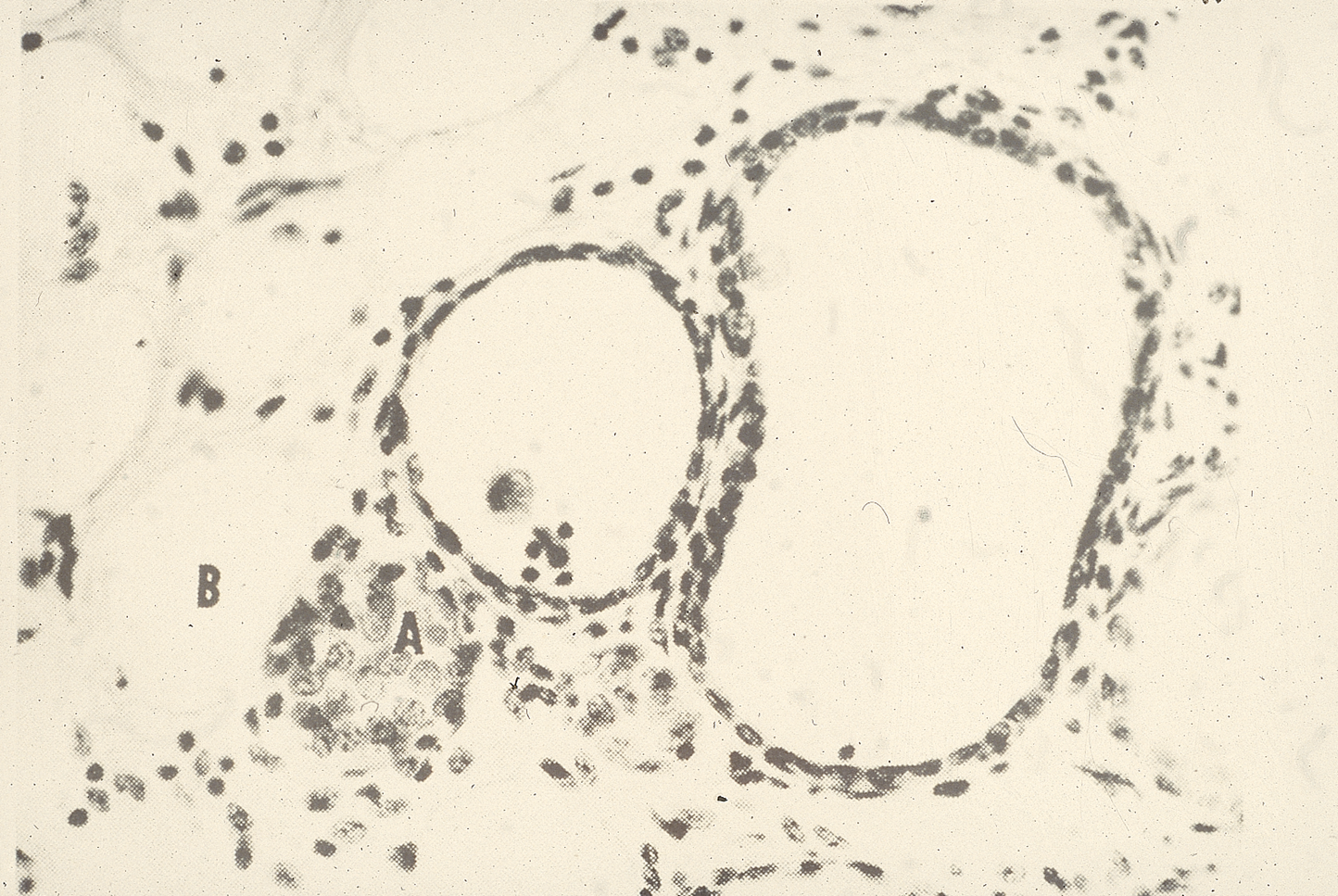
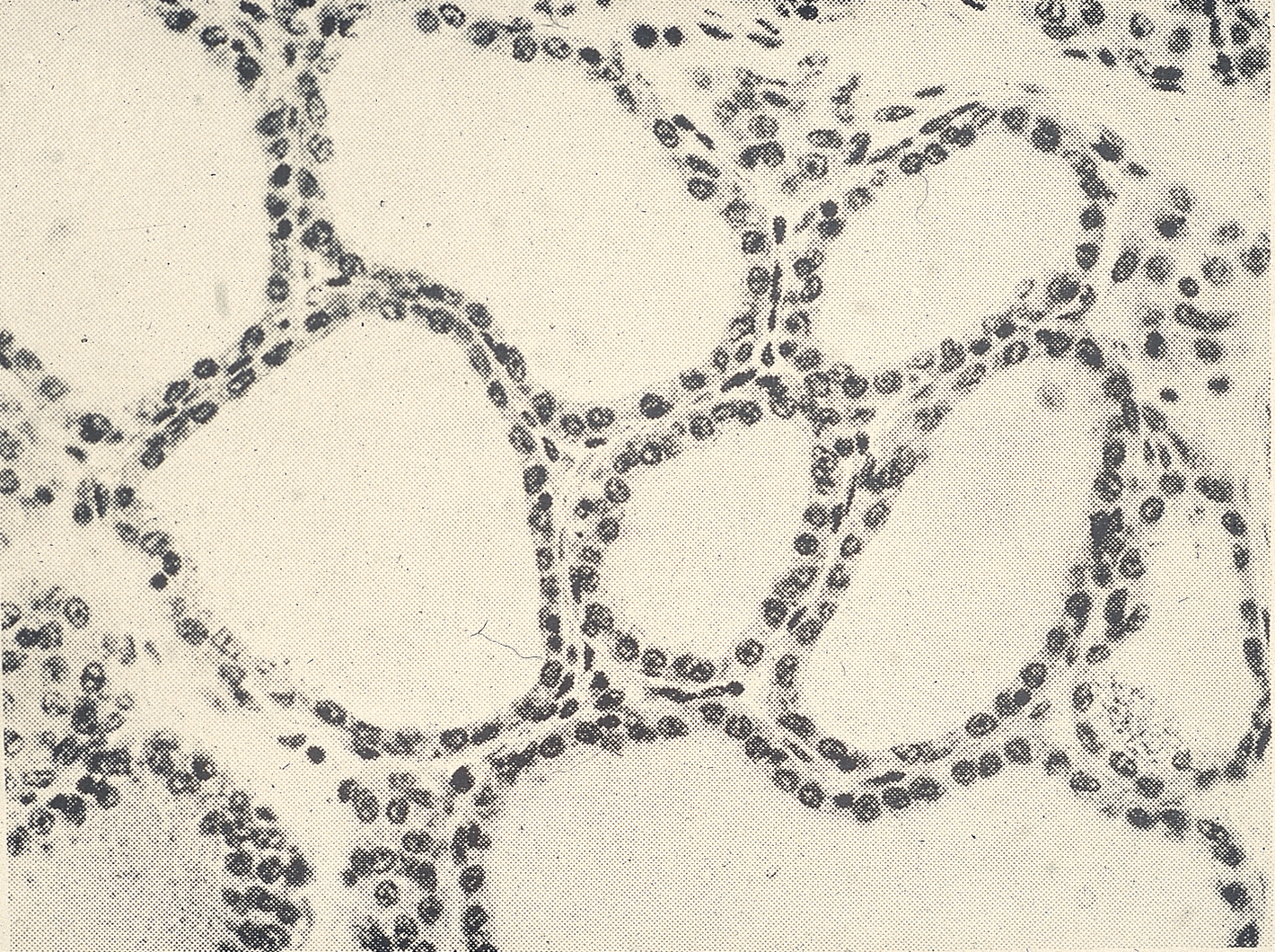
Keratin in streak canal, Smooth muscle
Keratin plug forms in streak canal
Milk Production at Dry-Off
National Mastitis Council recommends that cows be producing 33 lbs or less of milk per day at dry-off
Hard to do in high producing herds
Minnesota average at dry-off is about 60 lbs per day
Cows are more susceptible to environmental mastitis first two weeks
Keratin plug formation can be delayed due to leakage
Cows are uncomfortable due to engorgement of udder
Specific Mammary Immune Mechanisms
Humoral Immunity
IgG1 Predominant in normal milk
Opsonic for macrophages
Not opsonic for neutrophils
Can fulfill some of the functions of IgA
IgG2 Opsonic for neutrophils
IgA may act to:
Prevent adherence to mammary epithelium
Neutralize toxins
Agglutinate bacteria
Prevent multiplication of bacteria
IgM
Good at fixing complement
Antibody concentrations are low in normal milk
Cell-Mediated immunity
CD8+ T-cells attack cells that are infected
Seen with some viral infections and some chronic bacterial infections
Influx of mononuclear cells into the mammary gland: not so good
Obliteration of functional secretory tissue
HPAI causes fibrosis of mammary tissues
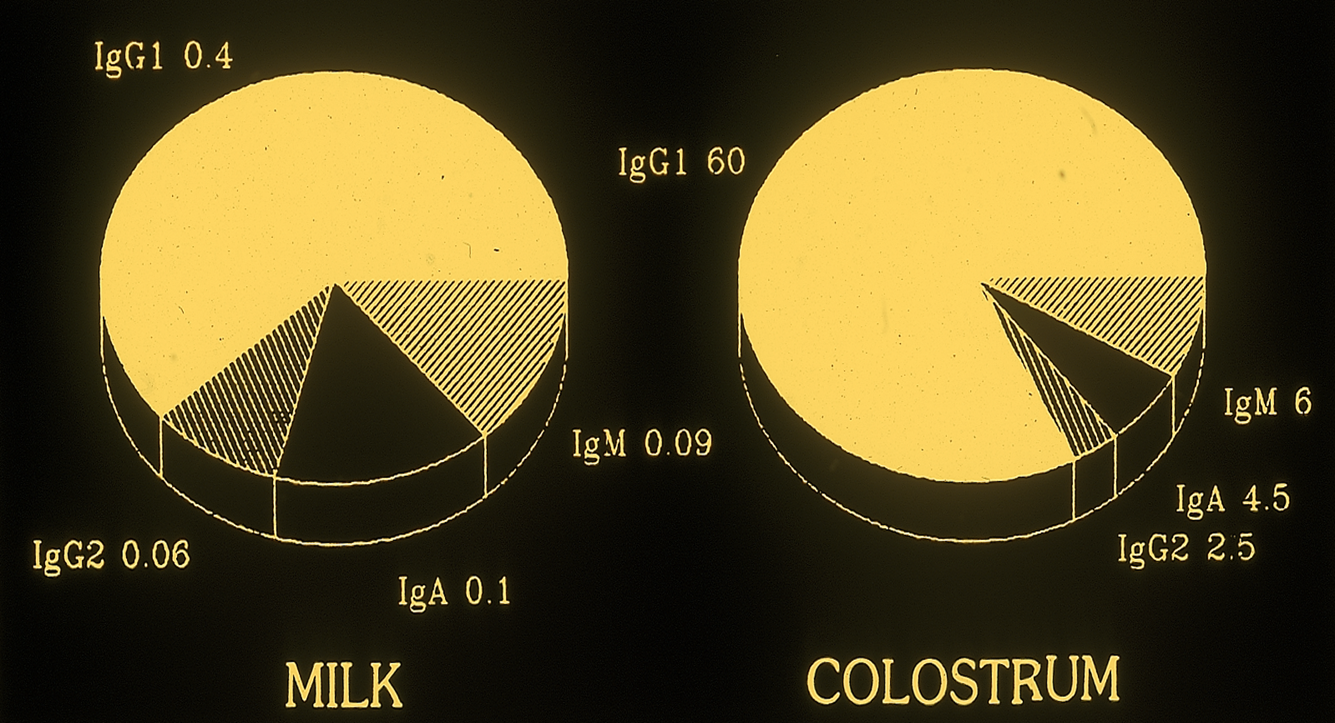
Bovine Immunoglobulin mg/ml
Immunization:
Stimulate antibody production
Stimulate cell-mediated immunity
Bottom Line to Prevent Mastitis
Maintenance of anatomical integrity
Clean environment to lessen exposure
Maintenance of good milk flow to “wash out” bacteria and toxins
Use of the few good vaccines
Good milking techniques/equipment
Many Different Species and Breeds of Poultry and Game Birds
Hobby flocks are becoming more common but HPAI has dampened enthusiasm
Some are adaptable to urban lifestyles
Species and different breeds may have different diseases affecting them
Commercial flocks may be free of certain diseases and hobby flocks???
All-in All-out vs Mixed age
Free-range vs barn-raised vs cage-raised
Straight run (half egg layers, half males) vs. sexed
Broiler vs. Breeder and Laying Chickens
Broilers
Chickens harvested at 5-7 weeks of age
Turkeys 14-18 weeks of age
Breeders and Egg layers
Start laying eggs about 18 weeks of age
Vaccinations should be completed 4-8 weeks prior to start of egg laying if using MLV
Immunization Issues
Available vaccines are often sold in units of 1000 to 10,000 doses
Vaccination in-ovo and as day-old chicks may be the only immunizations received in hobby flocks
Vaccinations through water or mist/aerosol are common
Vaccination and revaccination may not be practical for hobby producers
Short lifespans in broiler chickens
Immunization in-ovo or not at all with some pathogens
Immunize hens for antibody in eggs


Wing web vaccination
Eye drop vaccination- drains through nasolacrimal duct into mouth and stains mouth blue to indicate proper vaccination
Unique Drug Problems
Drug residues in eggs
Similarities to milk residues
All antibiotics approved for use in laying hens must have a zero-day withdrawal
Treatment of Game Birds
Chickens and turkeys are considered major species by the FDA/CVM
All other avian species are considered to be minor species
There are very few drugs approved for minor avian species
Guidance 615.115
Not a law, AMDUCA is law
Major species: cattle, swine, chickens, turkeys, horses, dogs, cats
Provides regulatory discretion for veterinarians to use feed or water for delivery to alleviate pain and suffereing due to diseases
Minor Species extra-label drug feed use
Must be no approved drug that will work
Feed use of the drug must be approved in a major species
Feed must be labeled on the VFD for dose and duration identical to the major species label
Limited to farmed or confined minor species (not wild)
Must have valid VFD: veterinarians are liable for problems that occur
Therapeutic use only (no production drugs)
Use in mammals limited to drugs approved in a mammal species
Use in avians limited to drugs approved in an avian species
Aquaculture drugs have to be approved in an aquatic species
ILHAC Priorities
All Poultry
Avian Influenza infections – Low Path Avian Influenza (LPAI) and High Path Avian Influenza (HPAI) epidemiology, prevention and control, research to identify routes of infection.
Antimicrobial resistance and antibiotic substitutes in poultry production
Vaccine Availability and application: one vaccine versus two killed injections
Animal well-being
Turkeys
Reovirus
Clostridium - Dermatitis
ORT (Ornithobacterium rhinotracheale)
Layers and Broilers
E. coli Peritonitis (#1 disease concern of veterinarians in egg production)
Infectious Coryza (Avibacterium paragallinarum)
Clostridial infection: necrotic enteritis and Focal Duodenal Necrosis (FDN)
Food Safety - Salmonella spp. (Salmonella Enteritidis)
Internal Parasites – including Coccidia and round worms. It is believed these challenges will only become greater with the increased prevalence of Cage-free production. At the present time, there are no treatments available?
Campylobacter hepatitis and Spotty Liver Syndrome - lack of real-time PCR for diagnosis
Erysipelas and Pasteurella multocida
Enterococcus septicemia, inclusion body hepatitis are growing concerns in broilers and broiler breeders.
Cannibalism
Chickens, turkeys and some others are omnivores
Dead birds can serve as sources of disease organisms
Need to remove dead birds promptly
Birds can get buried in litter
Discovered later and consumed
Can serve as source of Erysipelas, botulism, necrotic dermatitis
White Leghorn

Male leghorn chicks are euthanized ASAP after hatching
Brown Egg Layers
Plymouth Rock, Orpington, RI Red

Cornish Cross
Meat breed, very rapid growth

Poultry Litter Conditions
More important for broilers or cage-free egg layers
Complete clean-out may occur once a year or less often in commercial facilities
Partial clean-out between batches
Dust: dry conditions, respiratory disease
Ammonia: wet bedding, respiratory disease
Mold
Pathogenic organisms: bacteria, viruses, fungi
Poor litter condition leads to burnt hock syndrome and foot lesions
Litter Moisture
Litter needs to be friable and dry and clean
Air change rate: probably the most important
Litter material and depth (don’t want them on concrete/dirt)
Drinker design (leaking, wet bedding)
Stocking density (as they get bigger, make sure there is enough space)
Diet (too much fat in diet can lead to greasy bedding)
Flock health
Lymphoid Leukosis/Sarcoma
Avian leukosis virus (retrovirus)
Subgroups A, B, C, D, E, and J
Classically only important in chickens, some disease in game birds
Previously widespread in virtually all chicken flocks except SPF (specific pathogen free) flocks
Infrequent or absent in some large commercial flocks due to eradication efforts since the 1980s. Currently more of a problem in broilers (J subgroup with myeloid tumors)
Tumor mortality is <4% with occasionally higher numbers
Lymphoid Leukosis Epidemiology
Infected hens shed virus into albumen or yolk
Congenitally infected chicks usually remain viremic for life
Most virus spread is horizontal early in life
Once eliminated from breeder hens, it is relatively easy to maintain virus-free flocks
Most commercial chickens have been selected for genetic resistance
Tumors
Most of the economic loss is due to decreased egg production rather than tumors
Usually 16 weeks of age or older
Most tumors result from induction of bursal cells
Apparently a wide variety of tumor types can be associated with these viruses
Lymphoid is most common
Non-lymphoid tumors are dependent on strain of virus, strain of chicken, age of exposure, dose, and route of infection
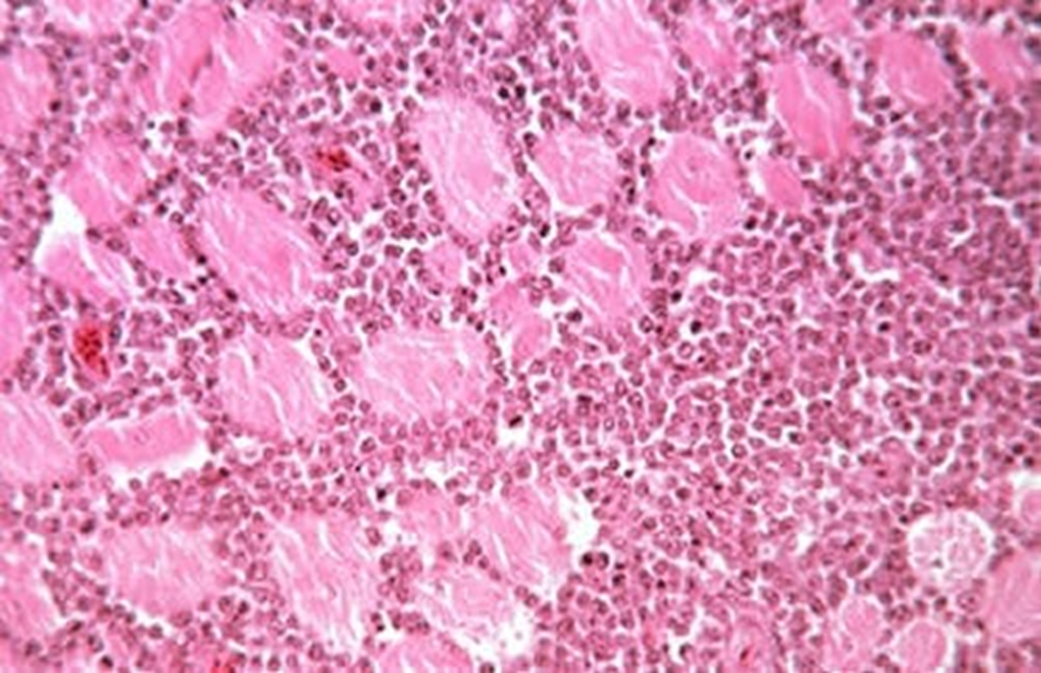
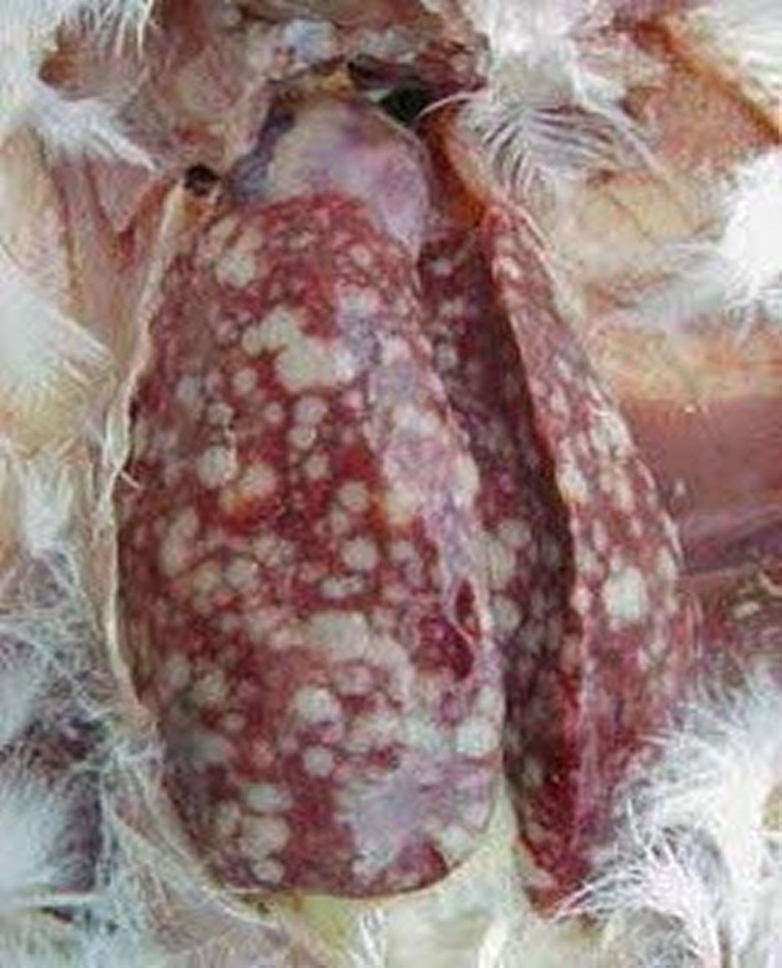
Lymphoid Leukosis Diagnosis
Demonstration of the virus is not helpful unless the virus is supposed to be eradicated from a group of chickens
Gross and microscopic characteristics of the tumors
Liver, spleen, other organs
Bursal tumors are almost pathognomonic
No peripheral nerve lesions
Helps differentiate from Marek’s
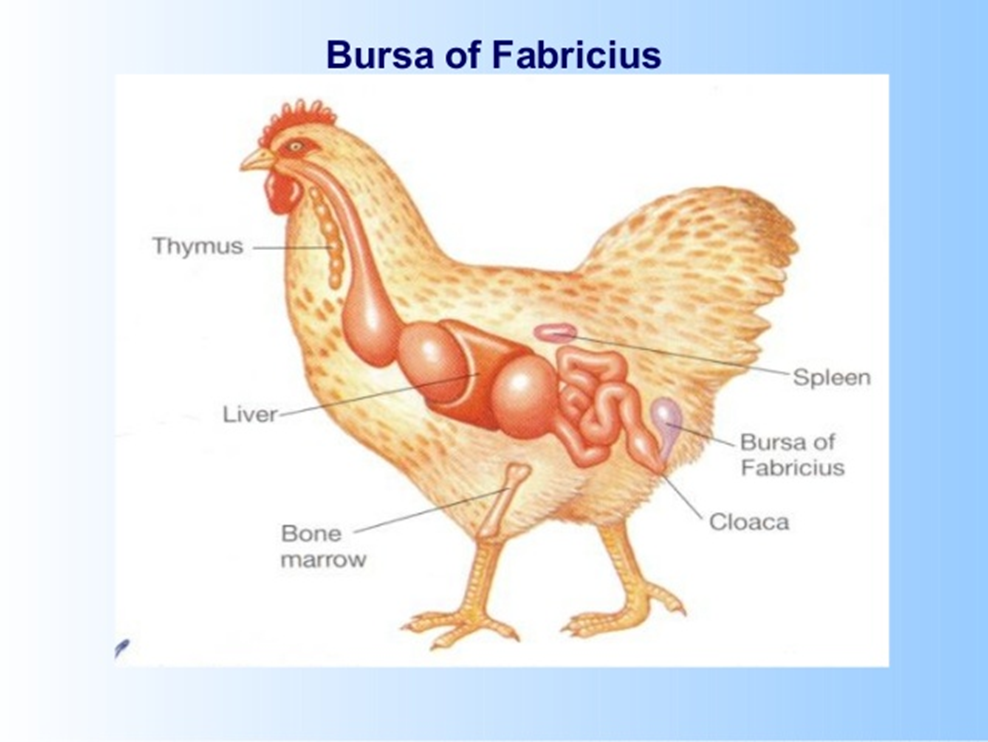
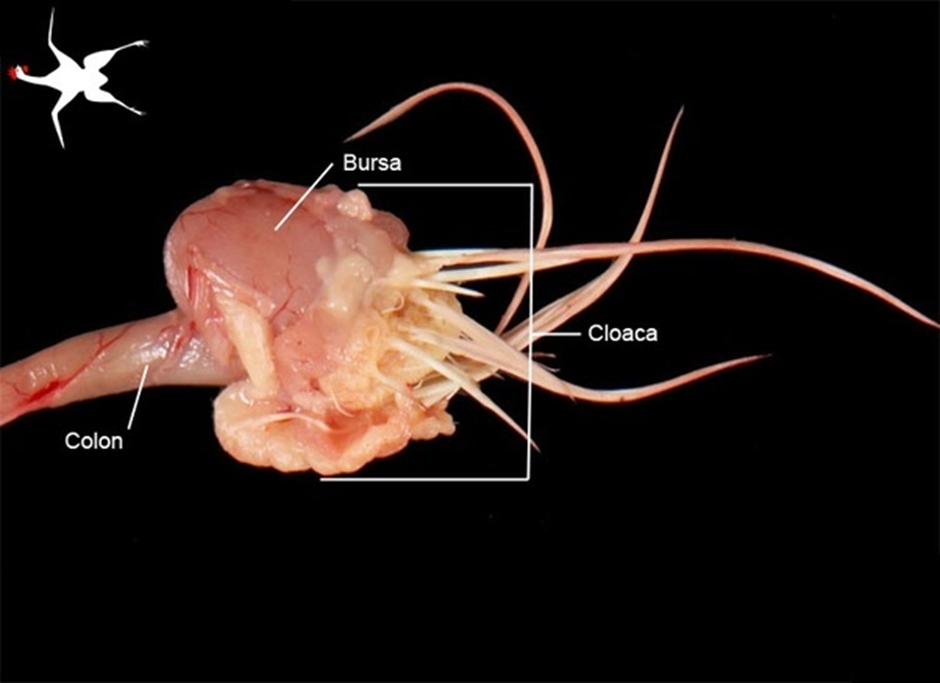
Bursa of Fabricius near cloaca
Control/Eradication of Leukosis
Test eggs from breeder flocks for viral antigen
Discard eggs from positive hens.
Rear breeder hens in small groups to limit the possibility of having an infected hen
Grow genetically resistant strains of hens.
Immunization has not been successful.
Most commercial chickens are infected.
Marek’s Disease
Alphaherpesvirus with lymphotrophic properties of gammaherpesvirus
One of the most ubiquitous infections in chickens
Infects feather follicles: Results in high concentrations in dust and litter of poultry houses
Highly contagious: Once in a barn, all chickens get infected
Not usually a problem in turkeys
Different Marek’s Disease Viruses
Gallid Herpesvirus 2 (MDV1, Virulent for chickens)
Gallid herpesvirus 3: (MDV2, Avirulent for chickens)
Meleagrid Herpesvirus 1 (turkey Herpesvirus) MDV3: Avirulent
Gallid Herpesvirus 2 (MDV1)
Further subdivided
M= Mild
V= Virulent
VV= Very Virulent
VV+ = Very Virulent Plus
VV+ viruses are the most common now
Thought to be a result of routine immunization
Marek’s Disease
Subclinical disease can cause decreased growth rate and egg production
Lymphoid neoplasms
Others:
Transient paralysis (range paralysis), early mortality syndrome, cytolytic infection, atherosclerosis and persistent neurologic disease
Marek’s Diagnosis
Enlarged nerves
Most consistent gross lesion
Paralysis or paresis
Enlarged feather follicles
Skin leukosis
Seen in broilers after defeathering
Cause for condemnation
Lymphoid tumors in viscera
Bursa usually not affected (unlike leukosis)
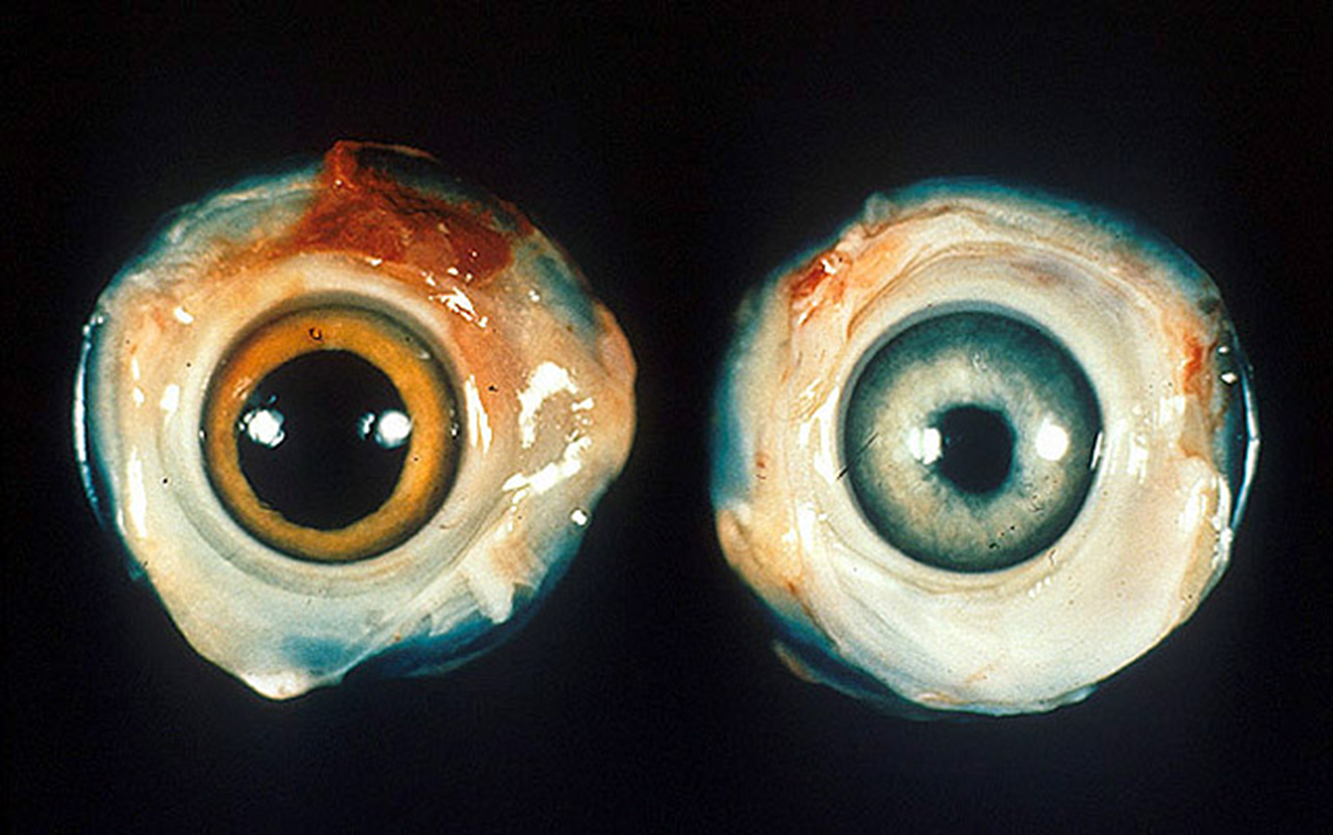
Marek’s on right: rough iris border, left eye normal

Paralysis from Marek’s Disease
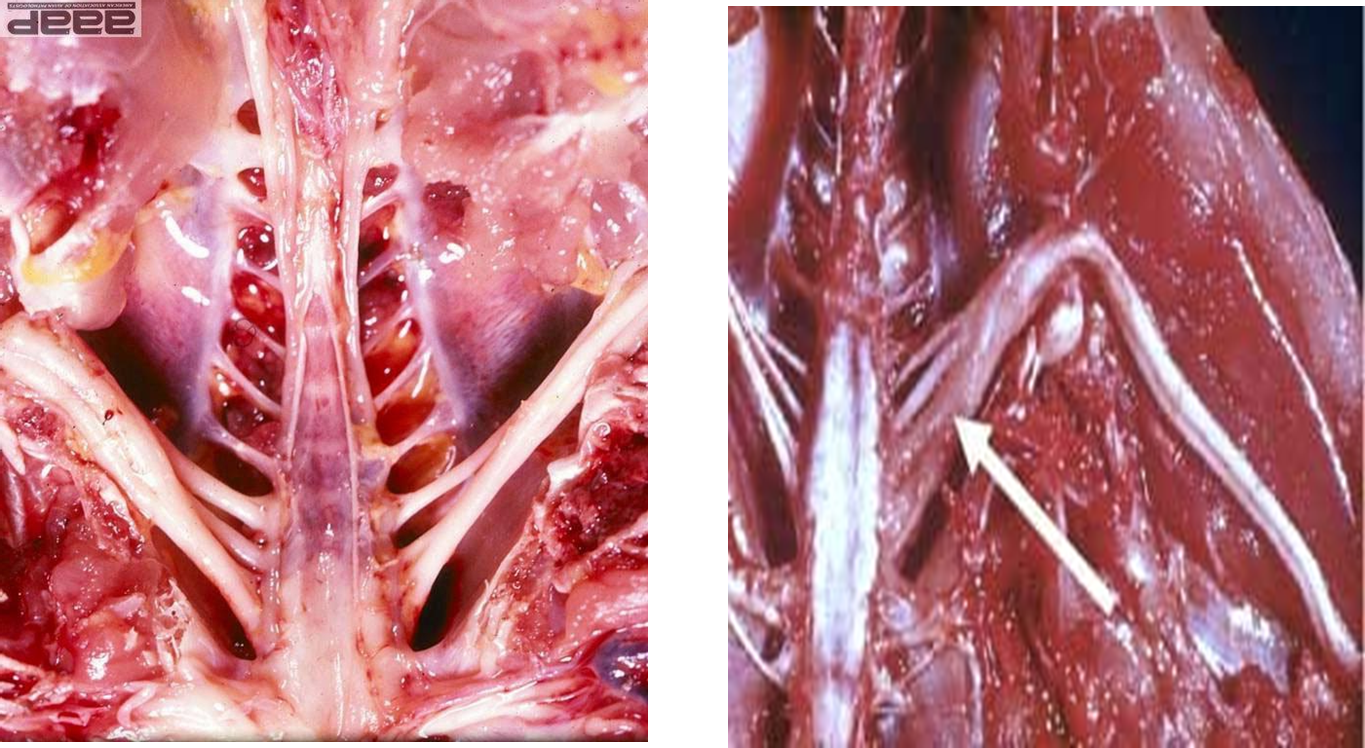
Enlarged sciatic nerves
Tumors
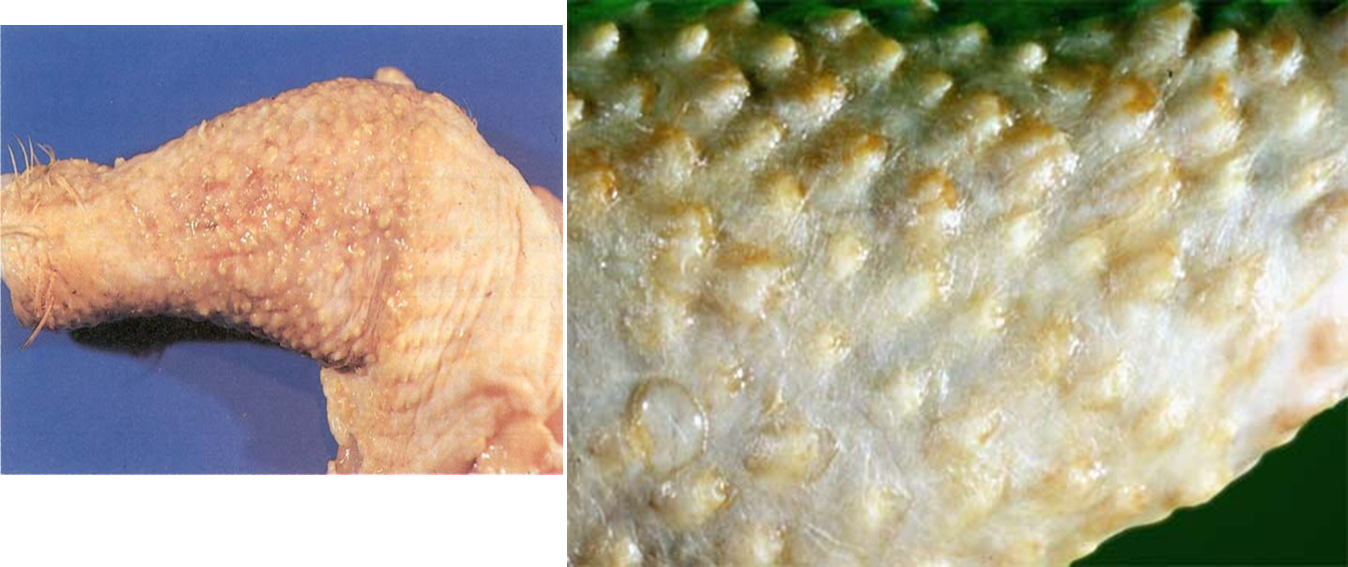
Skin leukosis
Marek’s Control
Strict sanitation: Delays infection until chicks are older.
Once in a poultry barn, it transmits readily via feather dust, etc.
Virus can survive for months in poultry house litter and dust.
Can inactivate virus in 2 weeks with high humidity and 37.5 C temp (100F):
Cleaning and disinfection can delay transmission or lower the infectious dose
Genetic resistance of some chicken strains
Marek’s Vaccination
Immunize initially with turkey virus or a combination of one or more other “avirulent” strains. Also have recombinants.
In-ovo immunization at 17 to 19 days of incubation has been automated and used especially in the broiler industry.
Re-immunize with a more virulent vaccine virus
Needed to protect against the vv+ viruses that are the most common in the industry
Vaccine “leakage”
Avian Tuberculosis
Mycobacterium avium subspecies avium
Serotypes 1, 2, 3 most commonly.
M. genavense is common in pet birds
Most common in parrots but occasionally found in other species
Transmission
Oral and aerosol (from feces) transmission
Natural habitats in soil and bird intestines
Commercial poultry do not usually live long enough to develop disease unless in multi-age layer or breeder flocks.
Backyard flocks, pet birds may develop disease
Avian Tuberculosis Epidemiology
Forms tubercles or ulcerated lesions in the intestine which shed high numbers into the droppings.
Organisms are usually ingested or inhaled from fecal material or other sources.
Dust
Soil
Fomites
Wild birds
Avian Tuberculosis Clinical Signs and Lesions
Domestic chickens and wild birds raised in captivity are most susceptible.
Emaciation, depression, diarrhea, marked atrophy of breast muscles
Tubercles in multiple organs usually beginning with intestine and liver
Usually no lung lesions
Diagnose with culture of tissues or feces
Acid-fast organisms on histopath
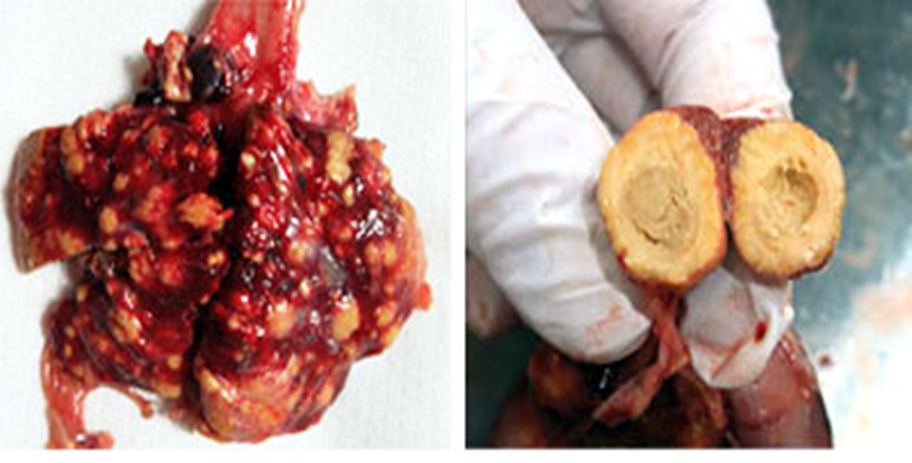
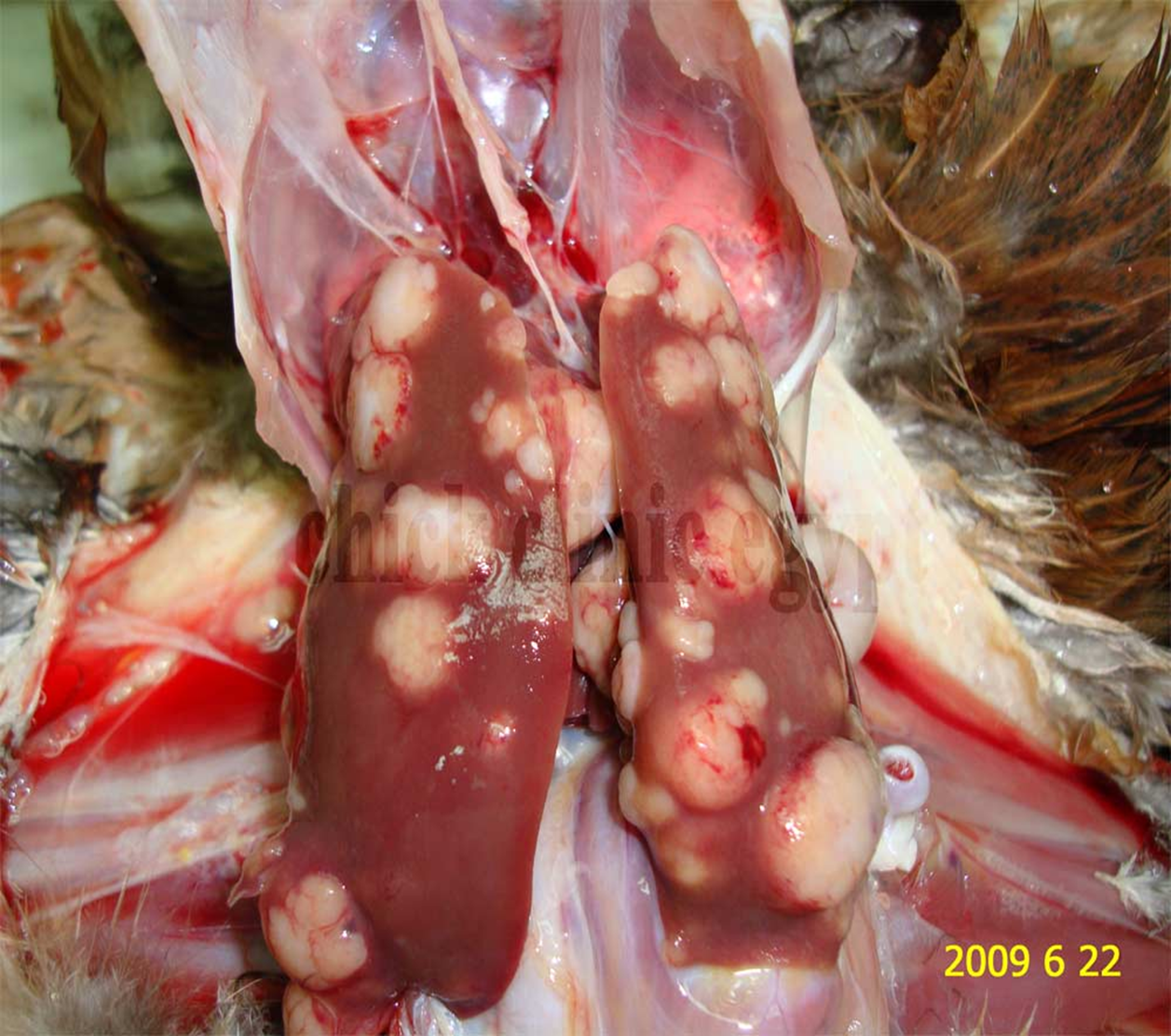
Avian Tubercles
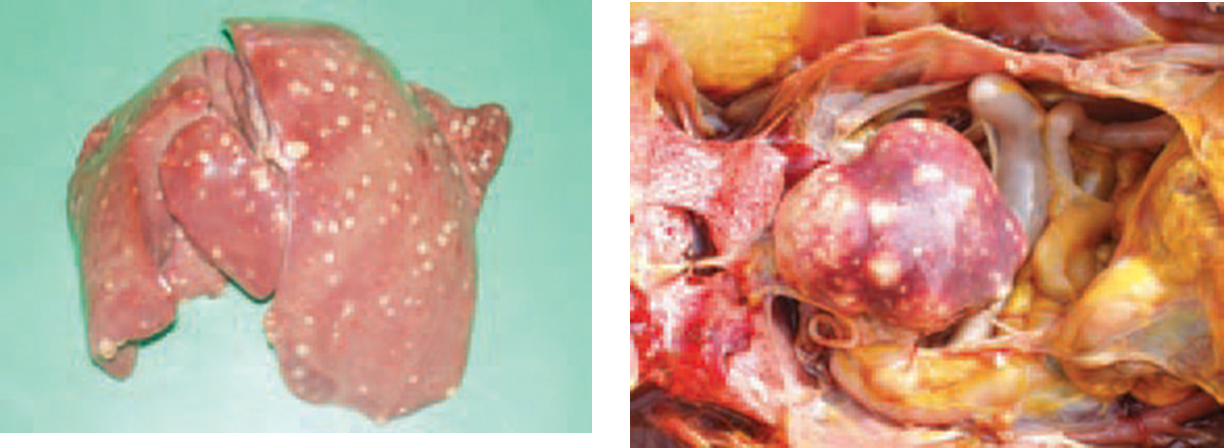
Tubercles in liver and spleen

Tubercles in a chicken liver
Diagnosis and Prevention of Tuberculosis
Tuberculin testing commercial birds in the wattle
Whole blood agglutination for imported birds, waterfowl and others. Can read the reactions in minutes.
Good biosecurity
Infectious Laryngotracheitis
Gallid Herpesvirus 1
Virulent strains and low virulence strains
Life-long infection
Recrudescence when stressed
ILTV
Acute, highly contagious
Affects mostly chickens and pheasants
Usually 14 weeks of age or older (maternal antibody present up until then)
Dyspnea
Coughing with bloody discharge
Up to 50% mortality
Subacute and subclinical forms occur
Reduced egg production
Lesions of ILTV
Acute disease
Necrotizing tracheitis
Blood, mucus, caseous exudates or hollow caseous casts in the trachea
Conjunctivitis
Swelling of infraorbital sinuses
Subacute disease
Punctiform hemorrhages in the trachea and larynx
Low virulence vaccine strains can spread to non-immune birds

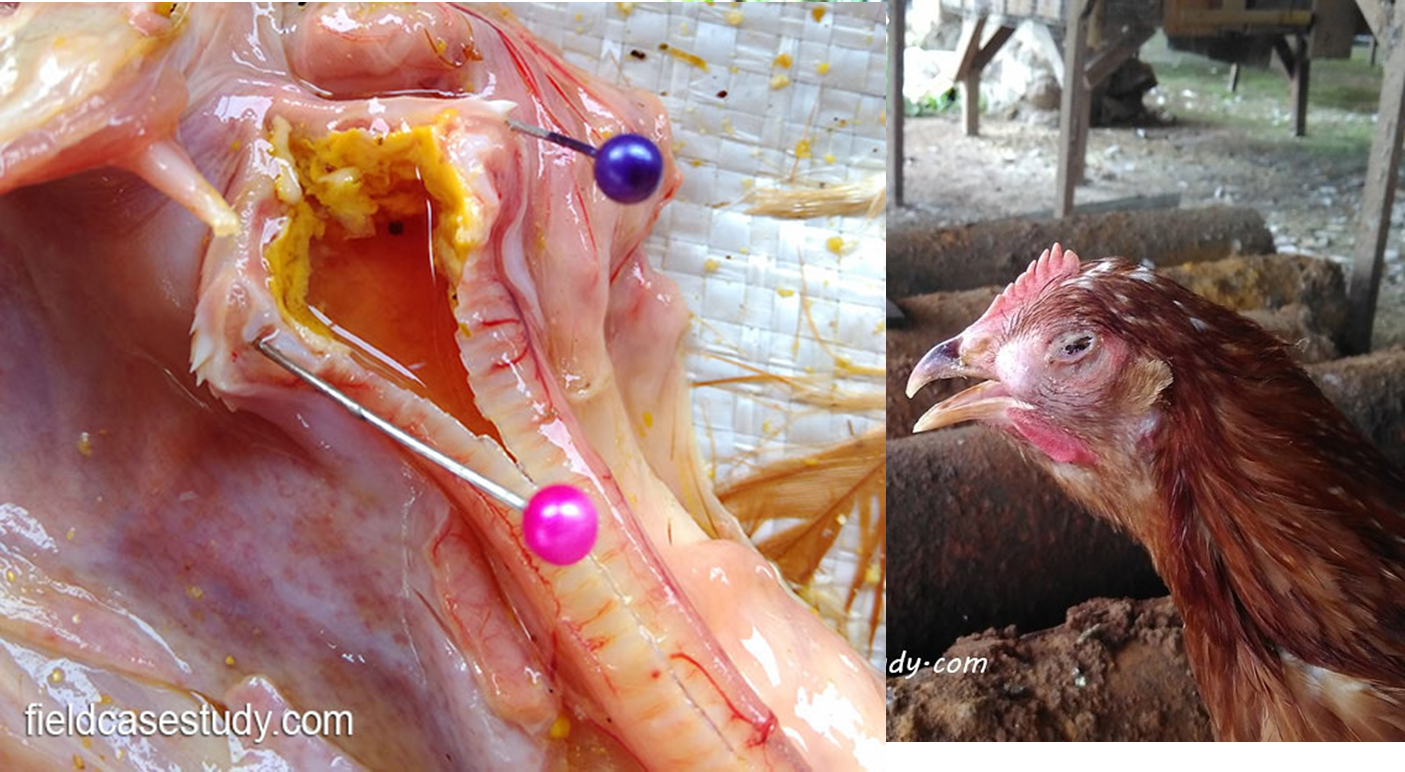
ILTV casts in the trachea and swollen sinus
ILTV Diagnosis
Intranuclear inclusions in tracheal epithelium early in the disease
PCR
Virus isolation
ILTV Control and Prevention
Good biosecurity
Virus remains viable 8-10 days in droppings but longer in winter temperatures
Remains viable up to 80 days in a carcass
ILTV Immunization
Endemic areas
Live attenuated
Eye drop, water or spray
Recombinant vector vaccines
In-ovo, subcutaneous and wing web
Fowl pox and Turkey herpesvirus recombinants
Newcastle Disease
One of the most important diseases worldwide
Avian Paramyxovirus 1
Terms:
Velogenic viruses: Pathogenic to all ages of birds.
Mesogenic: Highly pathogenic for embryonic stages. Less severe in immature and older birds.
Lentogenic: Low virulence viruses, loNDV, can be used as vaccines
Virulent Newcastle Disease (vND)
Velogenic and mesogenic viruses are now referred to as vNDV
Other terms:
Exotic Newcastle Disease (END)
Velogenic ND (VND)
Viscerotrophic velogenic ND (VVND)
2018 vND Outbreak
Hundreds of backyard flocks in California
Once commercial flock