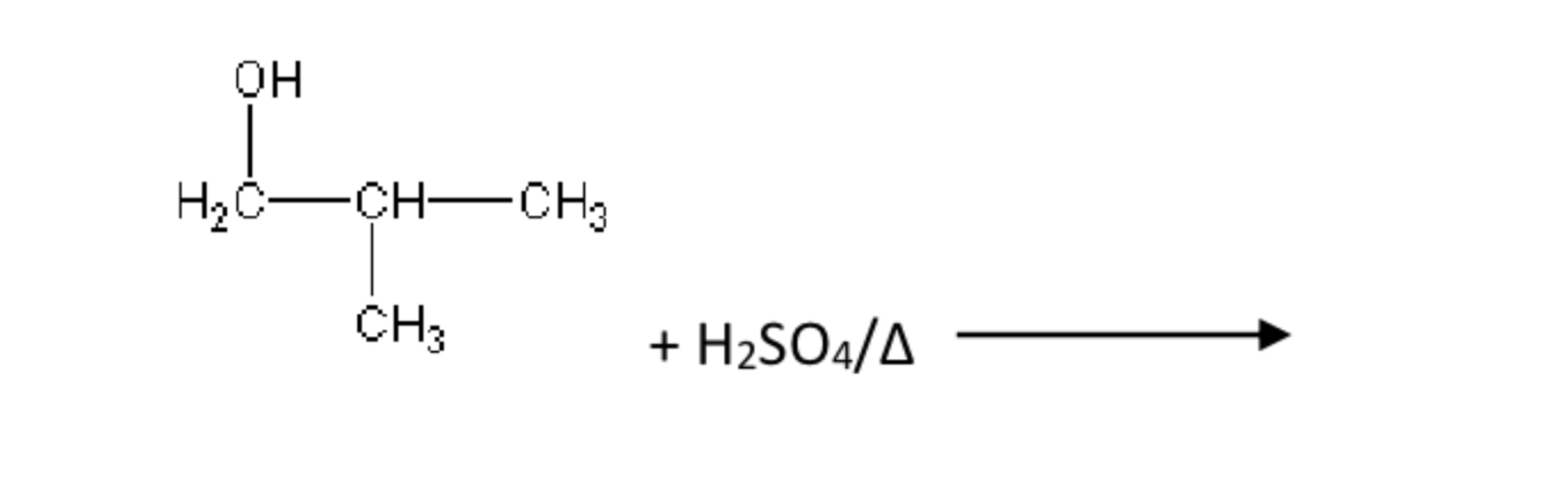biochem exam 1
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
What happens to energy in an endothermic process?
Energy is absorbed.
what happens to energy in an exothermic process?
energy is released
How do you interpret the value of the reaction energy in a chemical reaction?
A positive value indicates an endothermic reaction, where energy is absorbed; a negative value indicates an exothermic reaction, where energy is released.
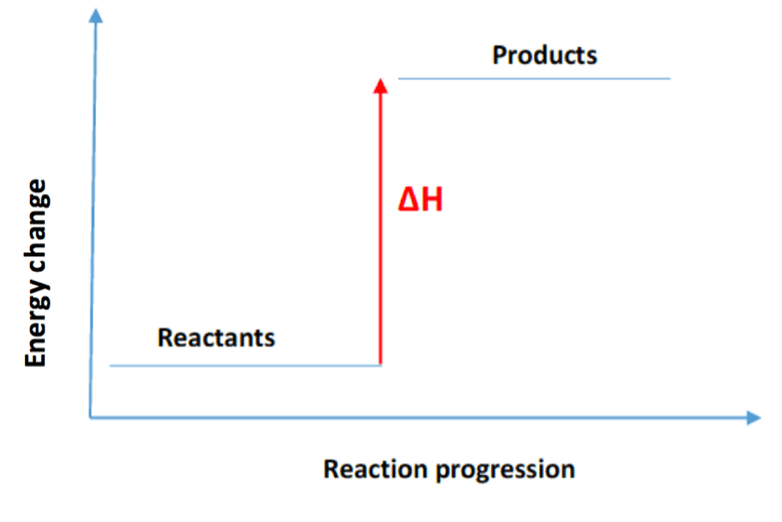
What does an energy diagram for an endothermic reaction look like?
The energy diagram shows reactants at a lower energy level, with a peak representing the transition state at a higher energy level, and products at a higher energy level than the reactants, indicating energy absorption.
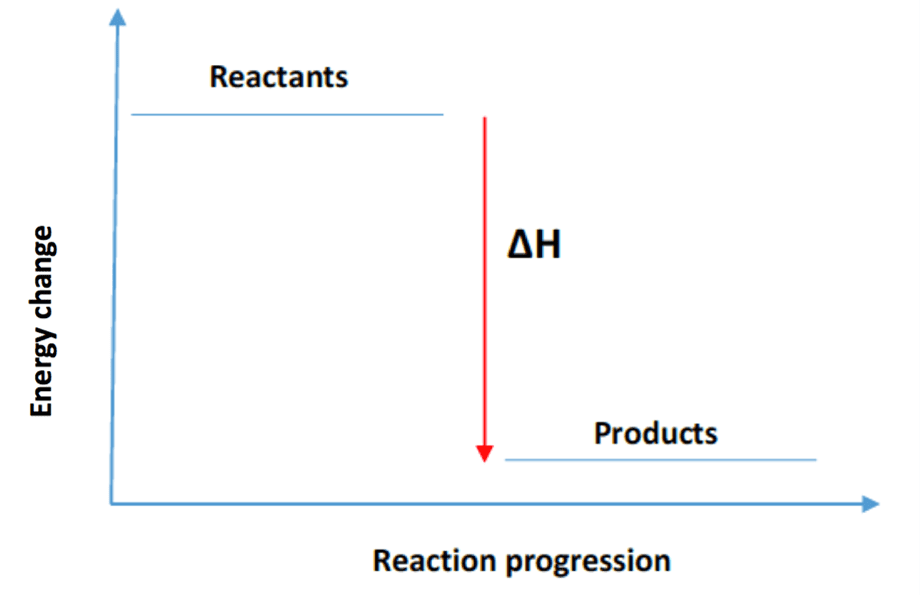
What does an energy diagram for an exothermic reaction look like?
The energy diagram shows reactants at a higher energy level, with a peak representing the transition state, and products at a lower energy level than the reactants, indicating energy release.
difference between ionic and covalent compound
ionic compounds formula metal+nonmetal, metal+ polyatomic ion, NH4 + anion. covalent compounds don’t start with metals or NH4
ammonium ion
NH4+
carbonate ion
CO3 (-2)
Nitrate ion
NO3 (-1)
sulfate ion
SO4 (-2)
phosphate ion
PO4 (-3)
hydroxide ion
OH(-1)
Formula for ionic compounds
cations first then anions
when to specify the number of elements in a compound
only for covalent
polar bonds
when 2 atoms in a bond have different electronegativity electrons are shared UNEVENLY, partial positive and negative charge
nonpolar bonds
when two atoms in a bond have the same electronegativities electrons are shared EQUALLY, no parital negative are positive charge
which elements generally make a polar molecule and why?
F,O,N because they are the most electronegative
in general which elements make a nonpolar molecule?
C,H
Classify polar and non polar molecules
add F+O+N then multiply by 3 this is polar score, Count the number of C atoms for non polar score. whichever is bigger that’ll be the molecule.
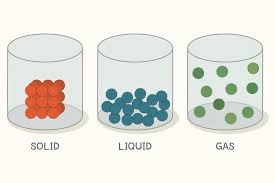
solids
individual particles are in fixed position, vibrate, define volume, shape
liquids
close contact, glide and slide, definite volume, indefinite shape
gas
particles of a substance are operate from each other and move about randomly in space, indefinite shape and volumes
solid to liquid
melting/fusion endothermic
liquid to gas
vaporazition/boiling endothermic
liquid to soil
freezing, exothermic
gas to liquid
condensation, exothermic
Explain and predict trends in melting point and boiling point.
larger molecules stronger forces and higher bp and mp and stronger forces higher bp and mp
water soluble ionic compounds
generally charge of -1 or +1 predict water soluble
covalent compounds water soluble and insoluble
covalent compounds are soluble because water is polar, insoluble water compounds won’t dissolve in water because they are non polar . Use F,ON X3 for polar score and add all C atoms for non polar score
electrolyte vs nonelctrolyte
electrolytes ionize when dissolved in water non electrolytes don’t they conduct electricity non electrolytes don’t, all ionic compounds and all acid and bases are electrolytes. nonelectrolytes are covalent compounds except for acid and bases
molarity
moles of solute per Liter of solution (M/L)
weight percent
the number of grams of dissolved solute per 100mL solution (g/100mL)
determine how many particles
1= non electrolyte, 2 or more = electrolyte ex. C6H12O6 1= non electrolyte, Mg3(PO4)2 5= electrolyte
osmolarity
total moles of all dissolved solutes per L solution. M x # of total dissolved particles ( the # depends on if the solute is electrolyte or non electrolyte)
formula for acids
H + something or something + COOH
formula for bases
metal + OH- or NH4 + OH-, or covalent + N, not a base CH3OH and CH3CH2OH
formula for salt
metal + anything not OH- or NH4 + anything not OH-
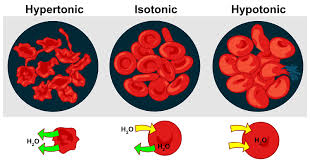
hypotonic solution
< 0.2 Osm (mOsm), water flows in the cell, swell hemolyze
isotonic
0.2-0.4Osm (200-400 mOsm), no osmosis occurs, no effect on red blood cells
hypertonic
> 0.4Osm water flows out of the cell, cell shrinks and may shrivel, crenateNaCl
NaCl
isotoinc weight percent 0.9%
glucose C6H12O6
5% isotonic weight percent
acidic solutions
<7, some salts+ H2O=acidic solution
neutral solution
pH=7, some salts and water equal neutral
basic solutions
pH>7, some salts + water= basic solution
define acid
any substance which donates an H+
define base
any substance which accepts and H+
neutralization reaction
acid + base = salt + water. ex 2 HCL + Ca(OH)2= 2 H2O + CaCl2
which would be lower pH 0.1M NaOH or 0.1M HCL
0.1M HCL
Buffer
maintains about a constant pH even if you add a little acid or base
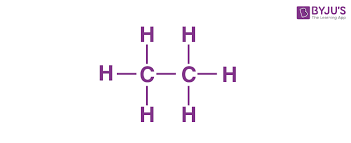
C2H6, complete notation, condensed notation,, skeleton notation, and Line angle
CH3CH3 or CH3-CH3, C-C, line angle must be three or more
line angle
there is a C at every bend and every end
hydrocarbon alkane
bond between C and H only single bond 0 C AND O BONDS
hydrocarbon alkene
double bond between C and H only 0BONDS C and O
alcohol
single bond C to OH R-OH 1 BOND C AND O
aldehyde
R-CHO carbon double bond O with H 2 BONDS C AND O
ketone
carbon double bond to O without H 2 BONDS C AND O
carboxylic acid
R-COOH carbon double bond to O and single bond OH 3 BONDS C AND O
carboxylic ester
Carbon double bond to O and single to O and C 3 BONDS C AND O
how to classify an alcohol
count the number of bonds from c to c not just and random c but the c that has OH attached
primary (1 alcohol)
1 bond c-c
secondary ( 2 alcohol)
2 bonds c to c
tertiary (3 alcohol)
3 bonds from c to c
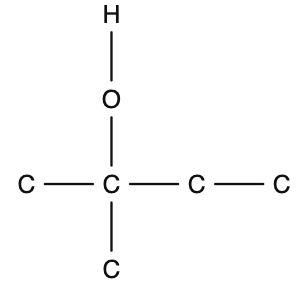
which alcohol is this
tertiary
what’s the formula for acyclic alkanes
CnH2n+2
what’s the formula for pentane
C5H12
what’s the formula for cyclohexane
C6H12
define isomers
2 or molecules that have the exact same formula but something about the structure is different
what a constitutional isomer
same formula but different order of bonding
example : different number of LCC (longest continuous chain or parent chain)
one is cycle one is not
different location of C=C
same formula
bending does not change different order of bonding
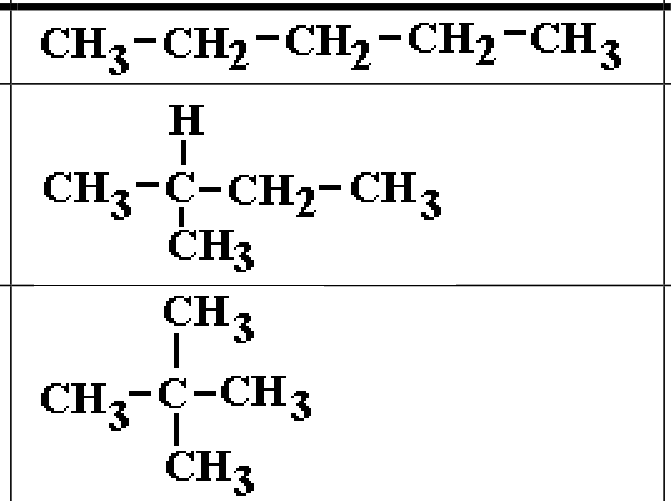
what is this
constitutional isomer
define stereoisomers
same formula
same order of bonding
different 3D shape
examples are cis-trans and enantiomers
define cis trans
molecules that have the same molecular formula and atomic connectivity but differ in the spatial arrangement of their atoms or like two different ways to assemble the same parts. They happen when a molecule has a "stiff" part (like a double bond) that can't rotate.
Cis: Think "Same side." The matching parts are both on the top or both on the bottom. (A trick to remember: Cis starts with C, like the word "Connected" on one side).
Trans: Think "Across." The matching parts are diagonal from each other, like a bridge crossing a river.

which is cis which is trans stereoisomer
left is cis , right is trans
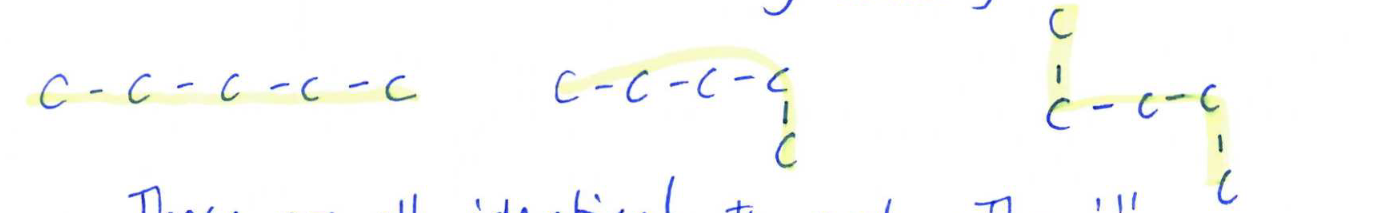
when comparing two compounds
if its not the same formula they are different compounds not isomers, isomers have the same formula
if they have the same formula: same exact bonding or structure they are identical
something about the structure is different its an isomer
chiral molecule
that is not superimposable ( not identical) on its mirror image
achiral molecule
one that is superimposable on its mirror image (they are identical)
nonsuperimposable
(not identical) like are hands when mirrored its different from our mirror image
superimpsoable
two socks from a pair are mirror images and identical
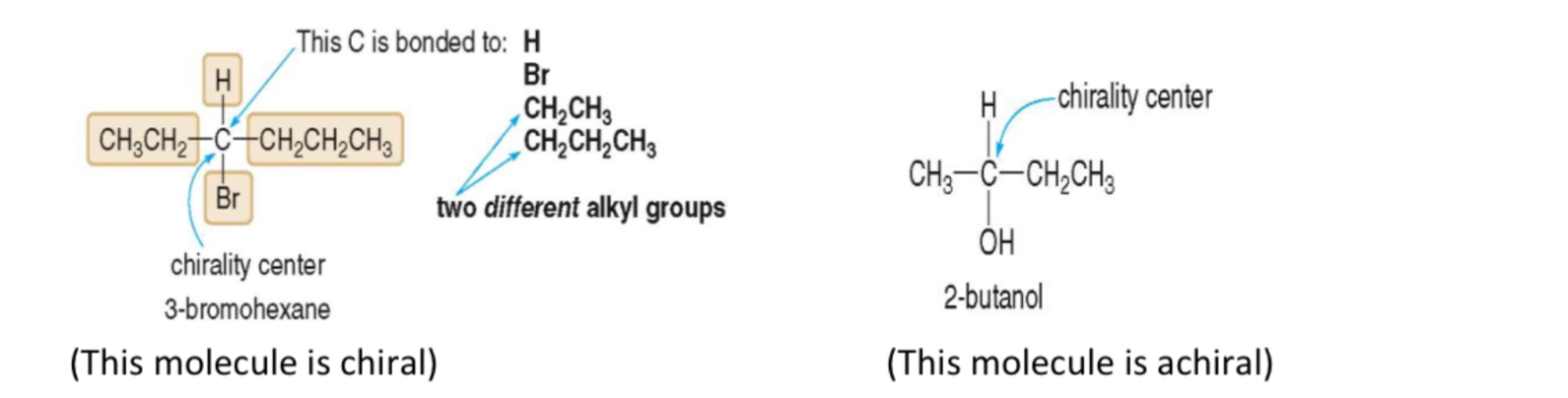
How to determine if a molecule is chiral or achiral
If it has a chiral carbon (carbon with 4 different groups bonded to it)
Why are enantiomers important in our body ?
Although they are mirror images of eachother, they are not the same! In particular, a chiral compound can “tell the difference” between the two mirror images of another chiral compound. Since most biomolecules in our body are chiral, our
bodies can “tell the difference” between the two mirror images of an enantiomer pair.
How to name alkene
Find the longest chain both carbon atoms of the double bond change suffix to ene
Number the carbon chain from the end that gives C=C the lower number
Number and name the groups
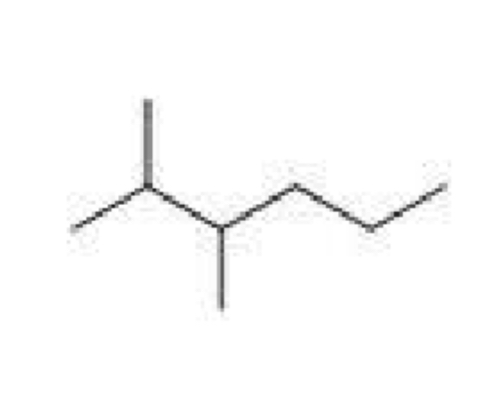
Name this
2,3-dimethylhexane
How to name alkanes
Find the longest chain both carbon atoms of the double bond change suffix to ane
Number the carbon chain from the end that gives the first side methyl group lower number
Number and name the groups
Meth
1 C
Eth-
2 C
Prop-
3 C
But-
4 C
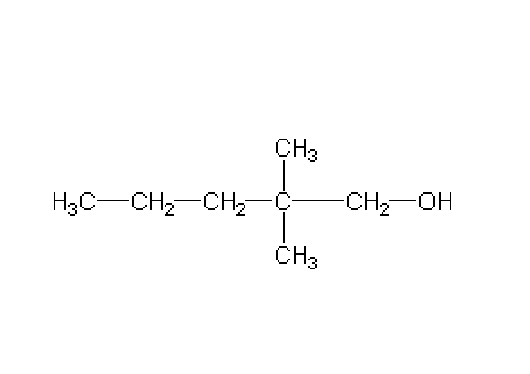
Name this
2,2- dimethyl-1-pentanol
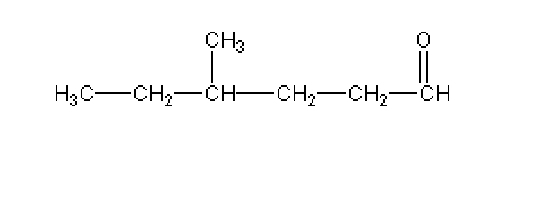
Name this
4-methylhexanal
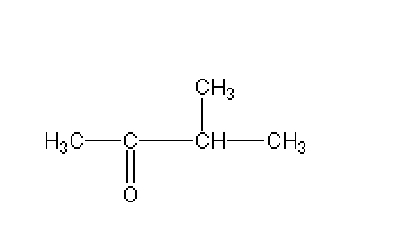
Name this
3- methyl 2- butanone
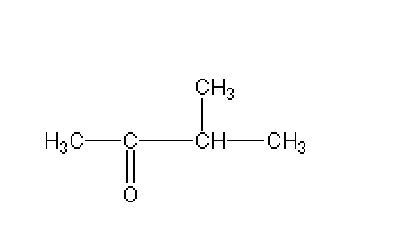
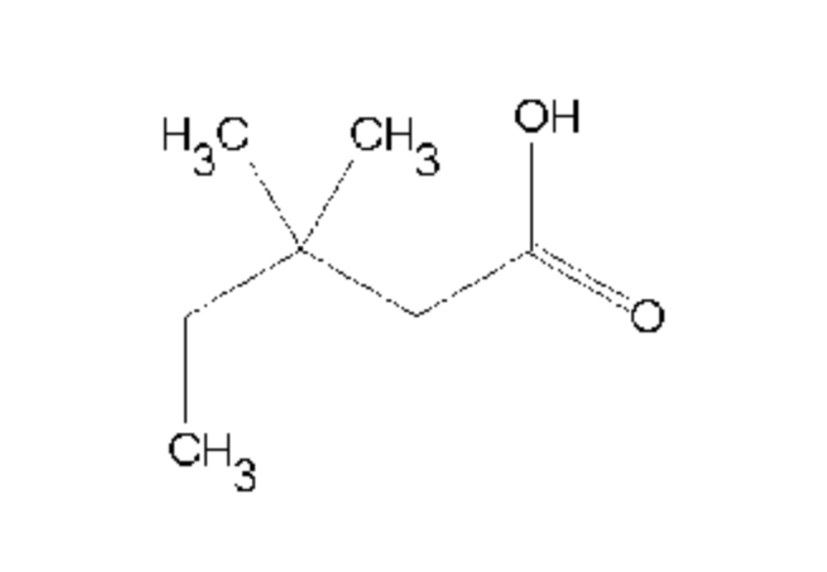
Name this
3,3- dimethyl pentanoic acid
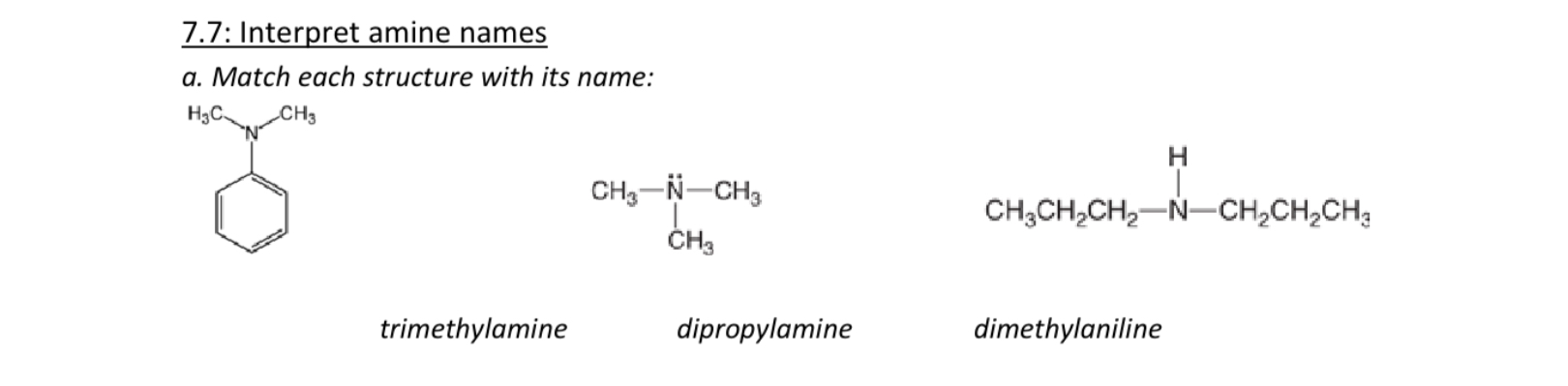
Name these
Left dimethylaniline
Middle trimethylamine
Right dipropylamine
What’s the structure for acetone
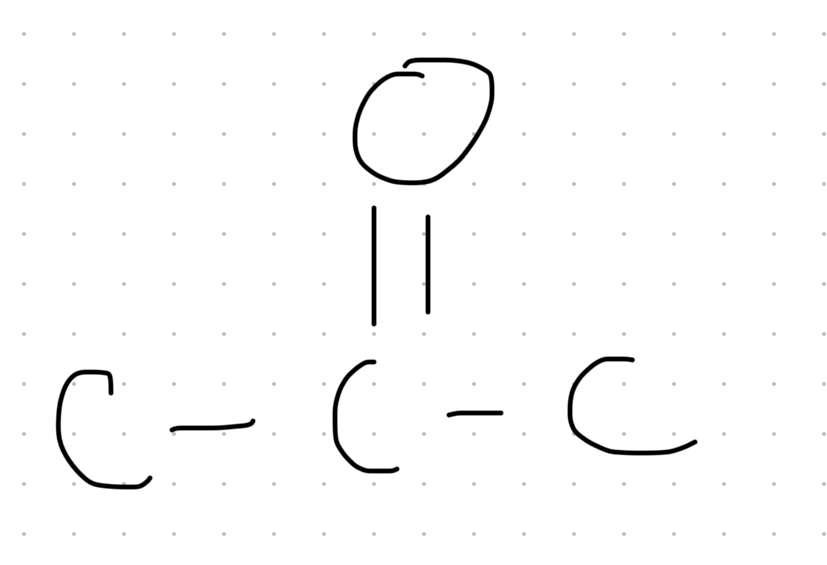
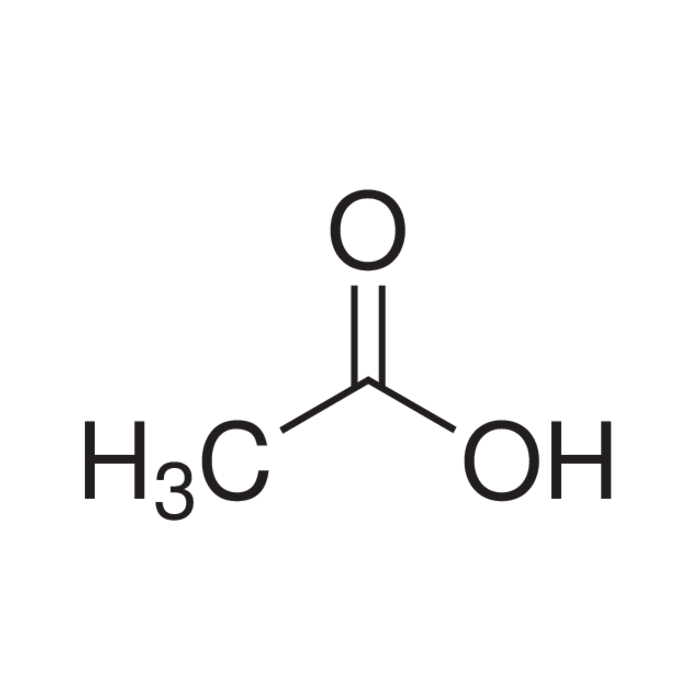
Structure for acetic acid
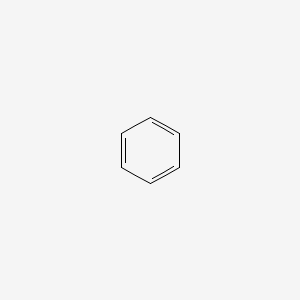
Benzene
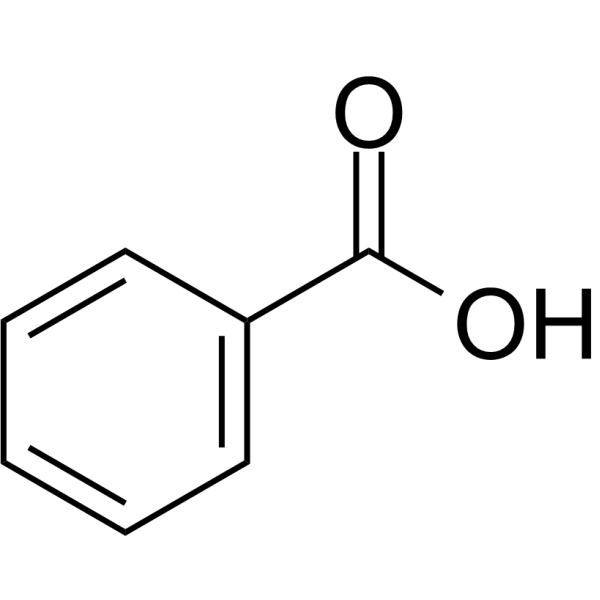
Benzoic acid
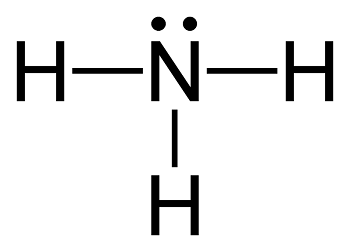
Ammonia
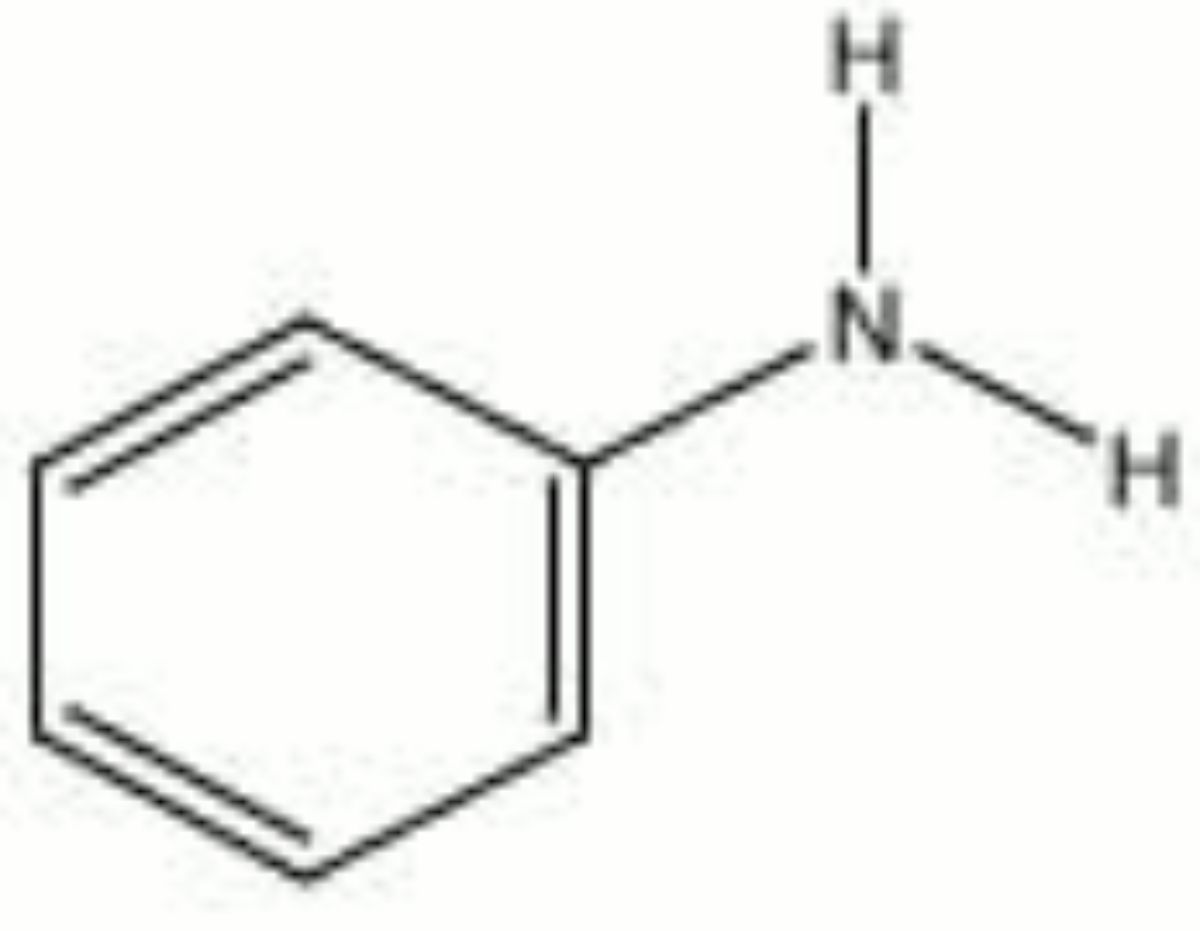
Aniline
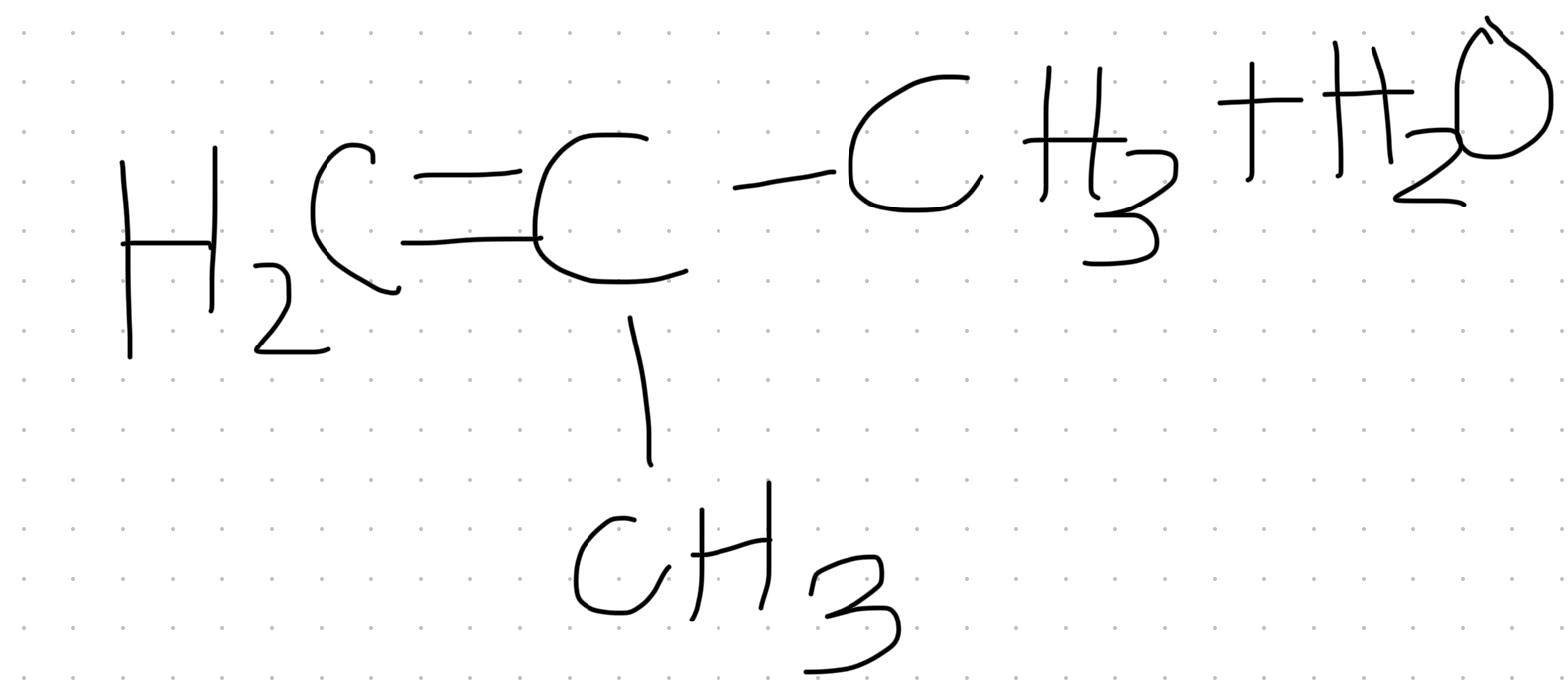
Rules for dehydrating alcohol
There must be a C (with H attached ) adjacent (next door) to alcohol group