GCSE Chemistry: Quantitative Measurements, Equations, and Calculations
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What is the law of conservation of mass?
No atoms are lost or made during a chemical reaction; the mass of the products equals the mass of the reactants.
What does a balanced chemical equation ensure?
It ensures that the number of atoms of each element is the same on both sides of the equation.
How is relative formula mass (Mr) calculated?
It is the sum of the relative atomic masses of the atoms in the numbers shown in the formula.
What happens to mass during a chemical reaction?
Essentially, no mass is lost during a reaction; the sum of Mr of reactants equals the sum of Mr of products.
What should you check if a reaction appears to involve a change in mass?
Check if a reactant or product is a gas and if its mass has not been accounted for.
What is an example of mass change in a reaction?
When a metal reacts with oxygen, the mass of the metal oxide product is greater than the mass of the metal.
What is the unit for measuring chemical amounts?
The unit is moles, symbolized as 'mol'.
How is the mass of one mole of a substance related to its relative formula mass?
The mass in grams of one mole is numerically equal to its relative formula mass.
What is the Avogadro constant?
6.02 x 10^23 particles per mole.
How can you convert between moles and grams?
Using the equation: mass = moles x molar mass.
What does the equation Mg + 2HCl -> MgCl2 + H2 represent in terms of moles?
It shows that 1 mole of Mg reacts with 2 moles of HCl to produce 1 mole of MgCl2 and 1 mole of H2.
What must be the same on both sides of a balanced chemical equation?
The total moles of one element.
What steps are involved in balancing a symbol equation using masses?
Convert the masses in grams to amounts in moles.
What is a key consideration when making chemical measurements?
There is always some uncertainty about the result obtained.
What should you be prepared to do regarding uncertainty in measurements?
Make estimations of uncertainty.
What is a limiting reactant?
The reactant that is used up in a chemical reaction, limiting the amount of products formed.
What happens if a limiting reactant is used in a reaction?
The amount of product produced is restricted to the amount of the limiting reactant that reacts.
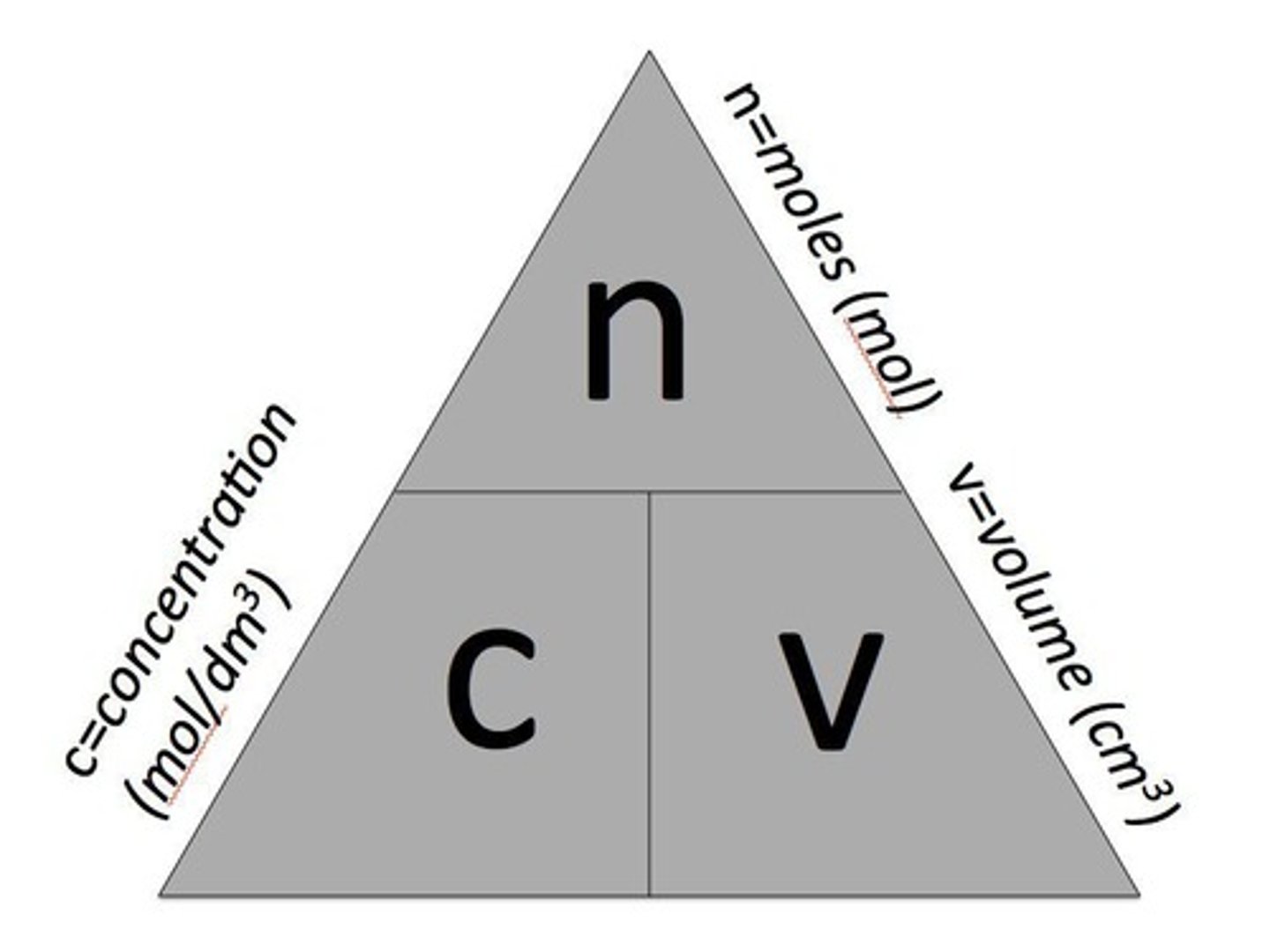
How is the concentration of a solution measured?
In mass per given volume, e.g., grams per dm³ (g/dm³).
What is the formula to calculate the mass of solute in a solution?
mass = concentration x volume (g = g/dm³ x dm³).
What effect does a smaller volume of solution have on concentration?
A smaller volume or larger mass of solute gives a higher concentration.
What is percentage yield?
Percentage yield = (Amount of product produced / Maximum amount of product possible) x 100.
What are three reasons why the actual yield may be less than the theoretical yield?
1. Reaction may not go to completion (reversible). 2. Product may be lost during separation. 3. Reactants may react differently than expected.
What is the yield in a chemical reaction?
The amount of product obtained from a reaction.
How do you calculate the theoretical mass of a product?
1. Calculate moles of reactant (mol = mass / molar mass). 2. Use balancing numbers to find moles of product. 3. Calculate theoretical mass (mass = moles x molar mass).
What is atom economy?
A measure of the amount of starting materials that end up as useful products.
Why is high atom economy important?
It is important for sustainable development and economic reasons.
What is the formula for calculating atom economy?
Atom economy = (Mr of desired product / Sum of Mr of all reactants) x 100.
What is the relationship between moles of gases and volume at RTP?
Equal amounts (in mol) of gases occupy the same volume under the same conditions of temperature and pressure.
What is the volume of 1 mole of any gas at RTP?
24 dm³.
How do you calculate the volume of gas produced at RTP?
Volume of gas (dm³) at RTP = Moles x 24.
What is the significance of using the limiting reactant for calculations?
Calculations should be based on the limiting reactant, not the one in excess.
What happens to the concentration if the mass of solute decreases?
The concentration of the solution decreases.
What is the effect of increasing the volume of a solution on concentration?
A larger volume or smaller mass of solute gives a lower concentration.
What is the first step in calculating the theoretical mass of a product?
Calculate the moles of the reactant using the formula mol = mass / molar mass.
What should be considered when choosing a reaction pathway?
Factors such as atom economy, yield, rate, equilibrium position, and usefulness of by-products.
What is the relationship between moles and concentration in solutions?
Moles = concentration x volume.
What is the importance of knowing the concentration of one solution in a reaction?
It allows for the calculation of the concentration of another solution when volumes are known.
What is the effect of a reversible reaction on yield?
It may not go to completion, affecting the yield of the product.
How does the loss of product during separation affect yield?
It decreases the actual yield compared to the theoretical yield.