Chemical Principles (as they pertain to microbiology) Ch. 2
1/114
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
115 Terms
What are all organisms made of?
All organisms are made up of various structures and molecules that come together to form 'life'' at the cellular level
Amino acids —> proteins
Fatty acids—> Phospholipids
Carbohydrates—>Glycerol—->Phospholipids
Carbohydrates—→Monosaccharides—→Polysaccharides
Nucleobases—→Nucleotides—→ DNA/RNA
Chemistry
the study of interactions between atoms and molecules
atom
the smallest unit of matter and cannot be subdivided into smaller substances without losing its properties
molecules
Atoms interact to form molecules
E.g O2 Oxygen gas=molecule
NH3 or ammonia =molecule
Atoms are composed of three particles:
Electrons: negatively charged particles
Protons: positively charged particles
Neutrons: uncharged particles
Protons and neutrons make up the nucleus
Electrons move around the nucleus in regions called electron shells
chemical element
Atoms with the same number of protons are classified as the same chemical element
e.g carbon, Nitrogen, phosphorus, iron, ect are all elements
Each chemical element has a different number of protons
Atomic number
number of protons in the nucleus
Atomic mass
total number of protons + neutrons in an atom
isotopes
It is possible for the same element to have atoms with different number of neutrons….if this is the case they are called isotopes.
O168 8= atomic number, 16=atomic mass
O817 8=atomic number, 17=atomic mass
Atomic mass differs between isotopes, atomic number stays the same
What are some of the important elements used by living organisms?
Hydrogen H 1
Carbon C 6
Nitrogen N 7
Oxygen O 8
Sodium Na 11
Magnesium Mg 12
Phosphorus P 15
Sulfur S 16
Chlorine Cl 17
Potassium K 19
Calcium Ca 20
Iron Fe 26
Iodine I 53
What are the most abundant elements in living organims?
Hydrogen
Carbon
Nitrogen
Oxygen
important isotopes:
C12 C13 C14
N14 N15
Why do microbiologists care about isotopes?
• Some isotopes (e.g., heavy ones are much rarer)
• Also, microbes generally prefer to use lighter isotopes.
• Heavier isotopes can be used in studies to track and enumerate fixation rates!
Electron configuration
• Electrons are arranged in electron shells corresponding to different energy levels
• Electron configuration refers to the arrangement of electrons in these shells
• Each shell can hold a characteristic number of electrons
Innermost shell: up to 2 electrons
Second shell: up to 8 electrons
valence shell
Number of electrons in outermost shell (valence shell) is what determines an atom's reactivity with other atoms
e.g carbon: Valence shell electrons=4, number of empty spaces=4
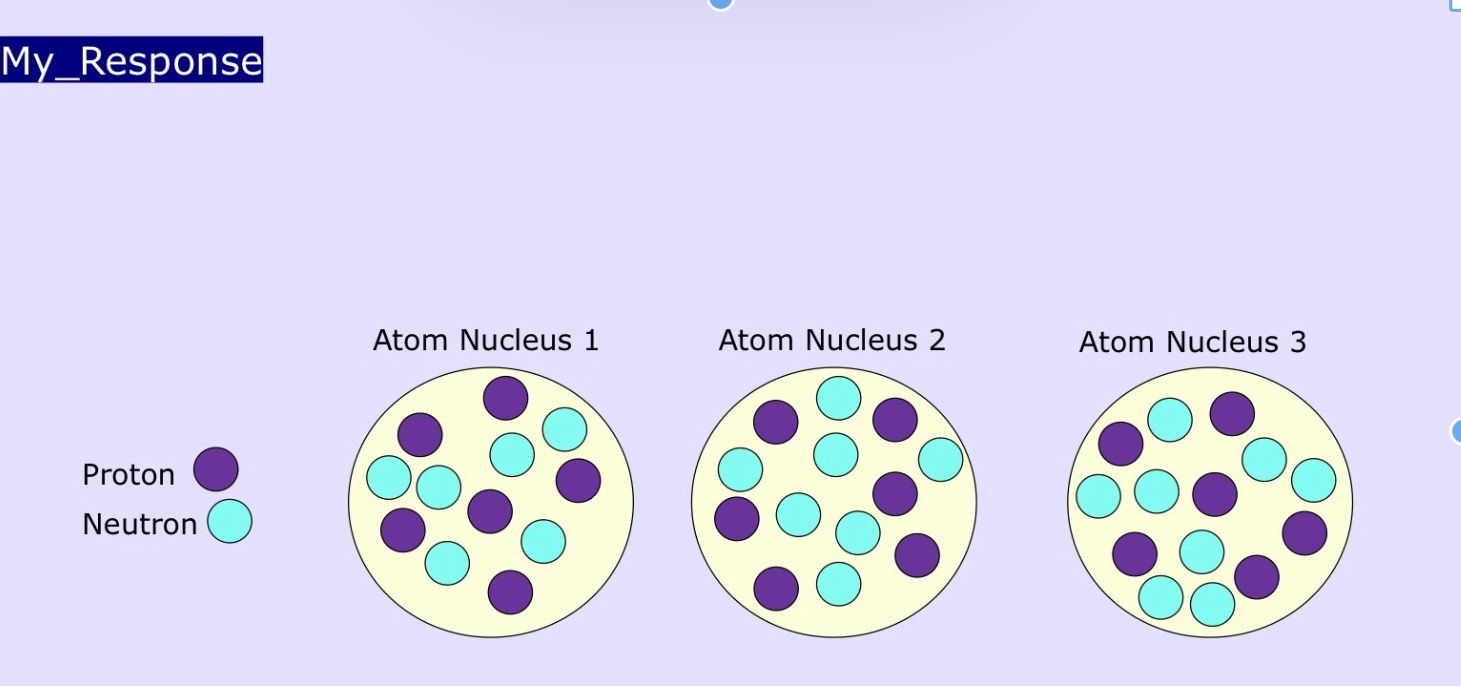
What element is this? And what is the heaviest isotope? What is the isotopes name?
This is Carbon: count protons, 6 each=carbon
Heaviest isotope? count Protons+neutrons, Atomic nucleus 3 has 14 so is heaviest
Name =C14
How to form molecules?
• Atoms form molecules by combining with other atoms to fill their outermost shells
• The number of missing or extra electrons in the outermost shell is known as the valence
• This is also considered the combining capacity of an atom
• Molecules hold together because the valence electrons of the combining atoms form attractive forces, called chemical bonds, between the atoms
compound
A compound is a molecule that contains two or more kinds of atoms
e.g H2O, two hydrogens and one Oxygen
Ionic Bonds
• Involve ions.
• Ions specifically are charged atoms that have gained or lost electrons
• Ionic bonds are attractions between ions of opposite charge
Cations
Atoms that lose electrons and become positively charged ions
K+, Na+, Ca2+
Anions
Atoms that gain electrons and become negatively charged ions
I- , Cl- , S2-
Covalent bonds
Formed when two atoms share one or more pairs of electrons.
stronger and more common in organisms than ionic bonds.
E.g. H2 gas (two hydrogens sharing an electron pair)
CH4 methane (carbon atom sharing an electron pair with each hydrogen atom)
Hydrogen bonds
Form when a H atom that is covalently bonded to an O or N atom is attracted to another N or O atom in another molecule
Are weak attractions, that do not bond atoms into molecules
Serve as bridges between different molecules or between different regions of the same molecule
Break and reform readily
Multiple hydrogen bonds stabilize large molecules
Ionic Bond importance
Weaker ionic bonds are important in biochemical reactions such as antigen–antibody reactions.
Covalent bond importance
Most common type of chemical bond in organisms and are responsible for holding together the atoms of most molecules in organisms
Hydrogen bond importance
bridges between different molecules or different portions of the same molecule, for example, within proteins and nucleic acids
Chemical reactions
involve the making or breaking of bonds between atoms
A change in chemical energy occurs during a chemical reaction
Activation energy
Breaking bonds requires Activation energy = energy needed to break a bond
Endergonic reactions
absorb energy
Exergonic reactions
release energy
• 3 Types of Chemical Reactions that are especially important in microbes (and organisms for that matter):
• Synthesis Reactions
• Decomposition Reactions
• Exchange Reactions
Synthesis Reactions
Occur when atoms, ions, or molecules combine to form new, larger molecules
A+B→ AB
Anabolism
the synthesis of molecules in a cell
Decomposition Reactions
Occur when a molecule is split into smaller molecules, ions, or atoms
AB → A+B
Catabolism
includes the decomposition reactions in a cell
Exchange Reactions
Are part synthesis and part decomposition
AB+ CD → AD +BC
NaOH + HCl → NaCl + H2O
Sodium hydroxide + Hydrochloric acid → Sodium Chloride + water
The Reversibility of Chemical Reactions
• Some reactions can readily go in either direction
• Each direction may need special conditions e.g heat or water
Organic compounds
always contain carbon and hydrogen; often structurally complex
Inorganic compounds
Typically lack carbon; usually small and structurally simple E.g. water is an important inorganic compound
Why is water an important inorganic compound?
• Excellent Temperature Buffer
• Excellent Solvent (substances dissociate in water)
• Serves as Reactant or Product in Many Cellular Reactions
pH
pH, like temperature, is very important for cells (including microbes).
Various compounds will act as either acids, bases, or salts
Acids
Substances that dissociate into one or more hydrogen ions and one or more negative
ions A.k.a. Proton (H+) donors
HCl → H+ + Cl-
Bases
Substances that dissociate into one or more hydroxide ions and one or more positive ions
A.k.a. Proton (H+) acceptors
NaOH →Na+ + OH
OH was the proton acceptor
Salts
Substances that dissociate into cations and anions, neither of which is H+ or OH
NaCl → Na+ + Cl-
What is pH is determined by?
The pH is determined by the acid-base balance
pH is the concentration of H+ ions or [H+] in solution
Increasing + [H ] increases acidity, decreasing the value of pH
Decreasing + [H ] increases the alkalinity, increasing the value of pH
pH=7 Neutral
pH<7 Acid
pH>7 Alkaline (basic)
certain microbes like to live in each of these pH’s
Why is it important to consider pH?
• Microbes must maintain a fairly constant balance of acids and bases
• Biochemical reactions are extremely sensitive to even small changes in pH
• Acids and bases that are continually formed during cellular reactions must be kept in balance by the microbe.
• Most microbes grow best between pH 6.5 and 8.5.
• But some can grow at pH extremes.
Buffers
• compounds that prevent pH changes
• Often buffers are used to keep cells viable in the lab (also sometimes the microbes make their own cellular buffers)
Mg(OH)2 + 2HCl → MgCl2 H2O
a chemical reaction where an antacid is neutralizing an acid.
Organic compounds
always contain carbon (and hydrogen if a biological molecule); and they can also contain oxygen and/or nitrogen in addition.
carbon skeleton
The chain of carbon atoms in an organic molecule is called the carbon skeleton
Functional groups
Functional groups bond to the carbon skeletons and are responsible for most of the chemical properties of a particular organic compound.
E.g. the hydroxyl (OH) group of alcohols is a functional group
R-O-H
Hydroxyl
name of group:alcohol
Biological importance: lipids, carbohydrates
R-C-H=O
Aldehyde
Biological importance: reducing sugars such as glucose, polysaccharides. Donates electrons
R-CH3
Methyl group
Important in DNA; energy metabolism
R-CH2-NH2
Amino group
biological importance in proteins
R=C=O-O-R
C doubled bonded to O single bonded to O-R
Eseter
Bacterial and eukaryotic plasma memebranes
R-CH2-O-CH2-R
Ether
Archaeal plasma membranes
R-C=O-OH
Carboxyl
Organic acids, lipids, proteins
R-O-PO3
Phosphate
ATP, DNA
Functional Groups: Amino Acids
Amino acids are important building blocks of proteins
Have two functional groups: amino group (NH3) + carboxyl group (−𝐶𝑂𝑂𝐻)
Making Larger Molecules
Small organic molecules can combine into large macromolecules
Macromolecules are polymers consisting of many small repeating molecules called monomers
Monomers join by dehydration synthesis or 'condensation reactions' to form macromolecules
R-OH+OH-R’ → R-O-R’ +H2O
OH+O is the water that is released when two organic molecules (monomers) join together to form a polymer
Larger organic compounds
Larger organic compounds are critical for life.
Includes important compounds like:
Carbohydrates
Lipids and steroids
Proteins
Nucleic acids
ATP
Carbohydrates
Serve as cell structures and cellular energy sources
Include sugars and starches
Consist of C, H, and O (often in the ratio of 1:2:1)
Carbohydrate isomers
• Many carbohydrates are isomers (same formula, different structures)
• Examples include: glucose, fructose, sucrose, lactose, maltose, starch, cellulose, and glycogen.
• Categories of carbohydrates include monosaccharides, disaccharides, and polysaccharides.
Monosaccharides
simple sugars with three to seven carbon atoms
Examples: Glucose Fructose(isomer of glucose) Deoxyribose(important in DNA)
Glucose (C6H12O6) is the most important monosaccharide in nature; it may occur as a chain or in alpha or beta ring configurations.
Glucose is the main source of energy for body cells.
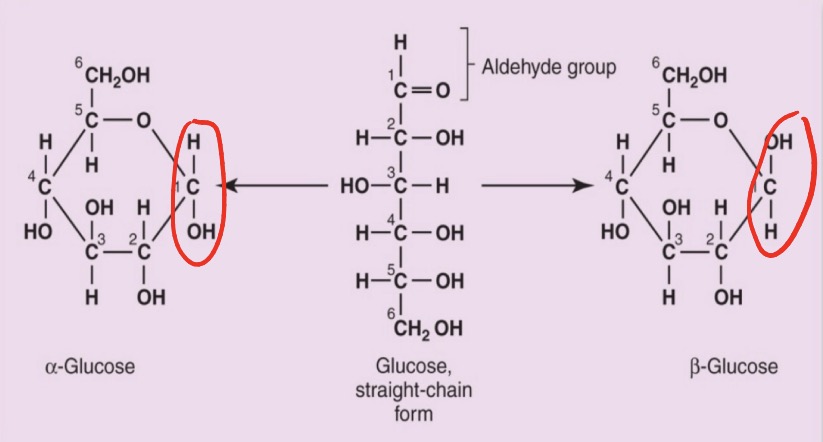
alpha and beta glucose difference
In alpha-glucose, the -OH group on C1 points down (below the ring), whereas in beta-glucose, it points up (above the ring).
Disaccharides
Disaccharides are formed when two monosaccharides are joined in a dehydration synthesis reaction
Examples:
Maltose: disaccharide of two glucoses
Sucrose: disaccharide of glucose and fructose
Lactose: disaccharide of glucose and galactose
Disaccharides can be broken down by hydrolysis reactions
Polysaccharides
Polysaccharides consist of tens or hundreds of monosaccharides joined through dehydration synthesis
Examples (made from glucose molecules) that differ in their bonding and function:
Starch (important in plant cells)
Glycogen(Important in animal cells)
Cellulose(important in many algae and plant cells)
chitin
Polysaccharides can also combine with other chemical groups to form even more complex macromolecules. – For example, chitin, the main component of the hard outer covering of insects, spiders, and crabs, is also found in the cell walls of fungi.
Lipids
Consist of C, H, and O
Are nonpolar and insoluble in water
They include:
Simple lipids [(fats and oils) triglycerides, and also waxes]
Complex lipids
Two main lipid functions:
1. They are the primary structural component of cell membranes
2. They can supply energy storage
Simple Lipids
Fats/oils or triglycerides
Contain glycerol and fatty acids; formed by dehydration synthesis
Trigerides(fats/oils): Glycerol + 3 fatty acids
Glycerol attached through carboxyl group to hydrocarbon chain of fatty acid
Saturated fats vs unsaturated fats
• Saturated fats have no double bonds in the fatty acids
• Unsaturated fats have one or more double bonds in the fatty acids
Examples of Types of Fatty Acids (Saturated vs. Unsaturated)
Saturated (single bonds between carbons)
Monounsaturated (Double bond in the carbon chain)
Polyunsaturated (Two or more double bonds in carbon chain)
Has to be in the carbon chain, the double bond is carboxyl group doesn’t count
Complex Lipids
• Contain C, H, and O AND P, N, and/or S
• Include phospholipids and steroids
• Cell membranes are made of complex lipids called phospholipids – Glycerol + two fatty acids + a phosphate group
• Phospholipids have polar as well as nonpolar regions
Phospholipids
Phosphate group, attached to glycerol, that is attached to two fatty acids
Polar head (loves water)
non polar tails (hates water)
cell membrane composed of phospholipids
Structure of Cell Membranes
A cell membrane is a lipid bilayer, consisting of two rows of phospholipids, arranged tail-to-tail.
proteins/ protein channels present
Lipids and other macromolecules
Lipids can also combine with other macromolecules to give even further molecules.
Lipopolysaccharides (LPS) come from lipid + piece of polysaccharide
Lipoproteins come from lipid + protein
Both LPS and Lipoproteins are important in the outer membrane of some bacterial cell walls.
Lipopolysaccharides
Lipopolysaccharides (LPS) come from lipid + piece of polysaccharide
E.g. if you recall lipids are generally insoluble…the LPS helps protect some bacterial cells from harsh environments (also gives more structural support)
Steroids
• Another complex lipid (also hydrophobic like other lipids).
• Contain four carbon rings
• A subset contain an –OH group attached to one ring = Sterol
Sterols are part of membranes that keep them fluid- separate fatty acid chains
Cholesterol
sterol
found in animal cell membranes and mycoplasma membranes (type of bacteria)
Ergosterol
Sterol
found in fungal cell membranes
Proteins
• Are made of C, H, O, N, and sometimes S
• Essential in cell structure and function
• Proteins include:
– Enzymes that speed up chemical reactions
– Transporter proteins that move chemicals across membranes
– Flagella that aid in movement
– Some bacterial toxins
– Structural components
• Proteins are polymers of amino acid building blocks...are typically 50 to several thousand amino acids in length
Amino Acids
• Proteins consist of subunits called amino acids
• Amino acids contain a carbon that has an attached: Carboxyl group (-COOH) Amino group 2 ( NH ) - Side group (-R group)
side groups change between amino acids
Amino Acids (microbes vs. humans)
Humans can synthesize certain amino acids, but not others. e.g his and met cannot be synthesized
Microbes typically synthesize most amino acids
Proteins…how do they form?
Are formed through the joining of amino acids via peptide bonds that are formed via dehydration synthesis reactions.
Protein Structure
• There are different levels of protein structure.
• The primary structure is a single polypeptide chain
• Is stabilized by peptide bonds
secondary structure of Protein
• The secondary structure occurs when the amino acid chain folds and coils in a helix or pleated sheet
• Is stabilized by hydrogen bonds
2= forming of helices and sheets
Tertiary structure of proteins
• The tertiary structure occurs when the helix or sheet folds irregularly into a specific 3D structure
• Tertiary structure is stabilized by: – Various types of bonds (more than only hydrogen bonds)
overall 3D shape
quaternary structure
The quaternary structure consists of two or more polypeptides
4= multiple polypeptide chains together
denaturation
• Proteins can also undergo denaturation
• Occurs when proteins encounter hostile environments (e.g. high temperature and unfavorable pH) and lose their shapes and functions
Conjugated proteins
Conjugated proteins are important and consist of amino acids and other organic molecules
– Glycoproteins (contain sugars)
– Nucleoproteins (contain nucleic acids)
– Metalloproteins (contain metal atoms)
– Lipoproteins (contain lipids)
– Phosphoproteins (contain phosphate)
Enzymes
Enzymes are specialized protein molecules produced by living cells.
They are known as biologic catalysts; that is, they speed up metabolic reactions.
Most chemical reactions in cells requires a specific enzyme
Enzymes are usually named by adding the ending “-ase” to the word. Hemolysins and lysozyme are examples of enzymes not ending in “ase.”
catalyst
A catalyst is an agent that speeds up a chemical reaction without being consumed in the reaction.
substrate
The specific molecule on which an enzyme acts is referred to as that enzyme’s substrate.
Nucleic acids
Nucleic acids are another type of large organic molecule.
• Carry genetic information
• Include DNA and RNA
• Consist of nucleotides
nucleotides contain:
– A five-carbon (pentose) sugar
– Phosphate group (nucleosides lack this)
– A nitrogen-containing (purine or pyrimidine) base
There are DNA nucleotides and RNA nucleotides One for each nitrogenous base, A, T, C, G, [U (in RNA)]
DNA
• Deoxyribonucleic acid
– Contains deoxyribose
– Exists as a double stranded helix
Nitrogenous bases from separate strands are bonded:
– Adenine hydrogen bonds with Thymine
– Cytosine hydrogen bonds with Guanine
DNA is the the “hereditary molecule”—the molecule that contains the genes and genetic code.
Missing oxygen on sugar compared to RNA
RNA
Ribonucleic acid
– Contains ribose – Is single-stranded
– Contains Uracil instead of Thymine for its nitrogenous bases
• RNA molecules participate in the conversion of the genetic code into proteins and other gene products.
There are several kinds of RNA…these play specific roles in protein synthesis