Group 7-Halogens
1/110
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
111 Terms
Fluorine gas colour
Yellow
Test for ammonia
EQ
Add NaOH
Hold damp red litmus paper at the mouth of the tube
9o8
Test for carbonate what acids can you add
EQ
Nitric acid
Hydrochloric acid
Test for sulfate ions
EQ
Add barium chloride
Sulfate gives white precipitate
What do you have to remember when doing half equations?
EQ
BALANCE FIRST
With both compounds on either side
Not last or whole equation is wrong
Similarity between sodium chlorides reaction with concentrated sulfuric aid and sodium bromides
EQ
Both form hydrogen halides
Difference between sodium chlorides reaction with concentrated sulfuric aid and sodium bromides
EQ
Bromide ions reduce sulfur in sulfuric acid
Oxidising agent definition
EQ
Electron acceptor
Give an equation to show the reaction between iodide ions and concentrated sulfuric acid to form the yellow solid
EQ
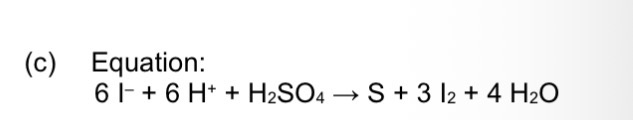
What is a sulfur containing foul smell in gas?
EQ
Hydrogen sulfide
Name the compound/element that is a yellow solid
EQ
Sulfur
Explain this observation
Effervescence when adding sodium carbonate
EQ
Shows contains hydrogen ions
So it is acidic
If a white precipitate is formed when adding silver nitrate to a sample what does that show
EQ
Chloride ions are present

Second mass is smaller/mass after step 4 is smaller that mass after step 2
AgCl dissolves in dilute ammonia
State why the amount of chlorine added to water must be controlled
EQ
Poisonous
Give an equation for the reaction of chlorine with water to form a solution containing 2 acids
EQ
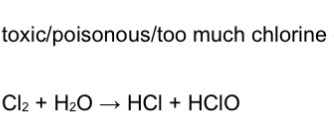
Observation when solid iodine is formed in a solution as a ppt
EQ
Brow solution
Black solid
Give an equation for the formation of sulfur dioxide when concentrated sulfuric acid is added to sodium iodide
EQ
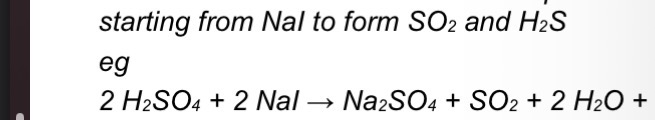
Give an equation for the formation of hydrogen sulfide when concentrated sulfuric acid is added to sodium iodide
EQ

Observation of sodium fluoride with acidified silver nitrate
EQ
No visible reaction
Solution remains colourless
Ionic equation for the formation of sodium carbon dioxide
EQ
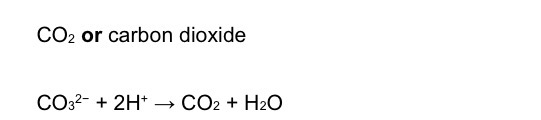
True or false
Sodium bromide forms bromine when added to concentrated sulfuric acid
EQ
True
True or false
Boiling points decreases from HCl to HI
False
Reducing ability increases down the group so is bromine a better oxidising agent or chlorine
EQ
Bromine
True or false
Chlorine can react with water to form an alkaline solution
False
Which is the best reducing agent
Cl2
Cl-
I2
I-
I-
Which species is the best oxidising agent
Cl2
Cl-
Br2
Br-
EQ
Cl2
Sodium chloride with acid base reaction-only one sulfur product
Sodium iodide-3 sulfur products
Sodium bromide- 2 sulfur products-1 redox reaction-with sulfur
What are the main products of these with sulfuric acid
EQ
H2
What can iodine reduce h2SO4 t?
EQ
Hydrogen sulfide
Sulfur
Sodium hydrogen sulfate
Dilute sulfuric acid with sodium iodide non redox reaction products
EQ
Sodium hydrogen sulfate
Iodine water
Concentrated sulfuric acid+ sodium iodide products
EQ
Sodium hydrogen sulfate
HI -hydrogen iodide
Concentrated sulfuric acid+ sodium chloride
EQ
HCl
Sodium hydrogen sulfate
Concentrated sulfuric acid+ sodium bromide products
EQ
HBr
Sodium hydrogen sulfate
Why does sodium fluoride not react with an acid?
EQ
It is a dilute not concentrated acid
Must be concentrated to produce F2
Is this too much or less for a boiling tube?
EQ
Too much so use large beaker
How do you maximise AgCl production?
EQ
Use excess HCl
What does decanting mean
EQ
A wet solid
Or some solid lost
Method of filtration
EQ
Filter
Wash
Dry
Lattice so there are —— strong bonds
EQ
Many
Disadvantage of treating water with chlorine
EQ
Some people find the taste unpleasant
Why does sodium bromide react with concentrated sulfuric acid in a different way to sodium chloride
EQ
Bromine ions are bigger than Cl- ions
How do you dissolve AgCl
EQ
Add excess dilute ammonia
Formula for dissolving silver chloride in dilute ammonia
EQ
AgCl+NH3 —> Ag(NH3)2 ^+ + Cl-
Ionic equation for the reaction between chlorine and cold dilute sodium hydroxide solution
EQ
Cl2 + 2HO- —> OCl- + Cl- + H2O
Equation for sodium chloride and concentrated sulfuric acid
EQ
NaCl +H2SO4 —>NaHSO4 + HCl
What is the role of sulfuric acid in the reaction fo sodium chloride with sulfuric acid
EQ
Proton donor
What is the role of sulfuric acid in the reaction of sodium bromide with concentrated sulfuric acid
EQ
Oxidising agent
If you have a solid sample and have to conduct tests on it what is the first thing you have to do?
EQ
Add water to make it a solution
What test would you do for sodium carbonate?
EQ
Add an acid
Fizzes
What happens if you add an acid or silver nitrate to sodium fluoride?
EQ
No change
What is formed when a metal carbonate reacts with a base?
EQ
Salt
Carbon dioxide
Water
What is produced in the redox reaction of concentrated sulfuric acid and sodium iodide?
EQ
Sulfur
Hydrogen sulfide
iodine
Give an equation for the reaction of solid bromide with concentrated sulfuric acid to form bromine
EQ
NaBr + H2SO4 —>SO2 +Br2 +Na2SO4 + H2O
Whenever bromine is formed in a product what colour is the solution
EQ
Orange brown solution
Explain how the forces between molecules of chlorine arise
EQ
Movement of electrons in one molecule
Induces a dipole in a neighbouring molecule
Temporary dipoles attract
Give an equation for the reaction of chlorine with water
EQ
Cl2 +H2O —>HCl +O2
Give an equation for the reaction of cold aqueous sodium hydroxide in the manufacture of bleach
EQ
Cl2 + NaOH —> NaCl +NaClO +H2O
What colour is the product when bromine the product itself is formed along with a salt
EQ
Brown solution

D
Iodide solution brown as well

A
Give an equation for the reaction of solid sodium bromide with concentrated sulfuric acid
EQ
NaBr + H2SO4 >Na2SO4 +SO2+Br2 +H2O
Why is HNO3 added to test for halides?
EQ
Removes carbonate ions that may give other precipitates with AgNO3
Why is AgNO3 added to test for halides?
EQ
Produces precipitates with chloride
Why is dilute aqueous ammonia added in halide test between 2 halides?
EQ
It dissolves one of them leaving the other
Formula of silver chloride with ammonia
EQ
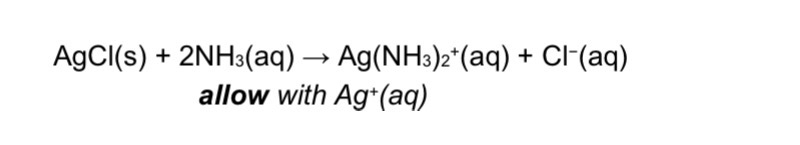
Explain why chlorine is added to water even though it is toxic
EQ
Health benefit outweighs risk
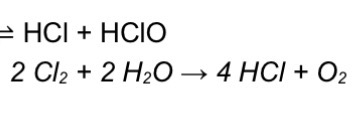
Equation for reaction of chlorine with cold water
EQ
Bottom EQ
Suggest why an excess of silver nitrate is used in test
EQ
To ensure that all the halide ions are removed from the solution
State what is observed when silver nitrate solution is added to sodium fluoride solution
EQ
Colourless solution
State one observation when solid sodium chloride reacts with concentrates sulfuric acid
EQ
Misty
Role of chloride ions in the reaction of sodium chloride with concentrated fluoride solution
EQ
Proton accept on
Redox equation of sodium bromide with concentrated sulfuric acid
EQ
NaBr +H2SO4 —>Na2SO4 +Br2 +SO2 +H2O
What are the major products formed when chlorine reacts with cold dilute aqueous sodium hydroxide?
EQ
NaCl and NaClO
What products are for me when iodine reacts with sulfuric acid
H2S
SO4 2-
S
SO2
What are the products of Br- and sulfuric acid
Hydrogen bromide and sulfur dioxide
What is formed when chlorine reacts with water
HClO
HCl
Chlorine gas colour
Green
Bromine gas colour
Red brown
Iodine gas colour
Grey
Describe boiling points down group 7.
Increases down the groups
Why does the boiling point increase down the group?
Increases strength of VDW as size and Mr of molecules increases
Physical state of fluorine
Gas
Physical state of chlorine
Gas
Physical state of bromine
Liquid
Physical state of iodine
Solid
Trend in electronegativity down the group
Decreases down the group
Why does electronegativity decrease down the group?
Outer electrons are further from the nucleus and are more shielded as they have more inner electrons
Are all halogens oxidising agents?
Yes
Why are all halogens oxidising agents?
They gain an electron
What happens when chlorine is added to potassium chloride solution?
No reaction
What happens when chlorine is added to potassium bromide?
Orange solution
Because Br2 is formed
What happens when chlorine is added to potassium iodide?
Brown solution
Because iodine is formed
What happens when bromine is added to potassium chloride?
No reaction
What happens bromine is added to potassium bromide?
No reaction
What happens when bromine is added to potassium iodide?
Brown solution
Iodine
What happens when iodine is added to potassium chloride?
No reaction
What happens when iodine is added to potassium bromide?
No reaction
What happens when potassium iodide is added to iodine?
No reaction