research methods I: unit 2 exam
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
between-subjects design
research between different groups of participants who are tested on different conditions or levels of an independent variable
between-subjects design: disadvantages
may require larger sample sizes, differences may have occurred due to individual differences, and groups must be equivalent
creating equivalent groups: random assignment
every participant have the same chance of being distributed into a random group
creating equivalent groups: blocked random assignment
participants are distributed evenly into groups to ensure there an equal number of participants in each group
creating equivalent groups: matching
participants are grouped into some subject variable of interest and are dispersed evenly across the experimental groups to ensure these variables are evenly distributed
→ these subjects variables of interest are often confounds that trying to be controlled for
conditions for matching
small sample sizes are not ideal, variable targeted must be related to the dependent variable, and measuring the matching variable must be feasible logistically
within-subjects design
the same participants experiences all of the conditions of a study and the difference is measured
within-subjects design: when to use
for smaller sample sizes, when individual differences may affect the results, and for when the differences of the same individual is essential (medication trials)
within-subjects design: order effects
when the order of specific sequences affect participant performance
progressive order effects: practice
when a sequence of conditions make proceeding trials easier from practice
progressive order effects: fatigue
when a sequence of conditions make proceeding trials worse from fatigue
carryover order effects
a specific order of a condition that may affect proceeding conditions than any other condition
→ e.g., participant who did AB did better than BA
controlling order effects: counterbalancing
altering the order of experimental conditions to reduce order effects
complete counterbalancing
every possible combination of conditions are tested
→ may not be plausible if there are many conditions
partial counterbalancing
a subset of all potential combinations of conditions are tested to ensure equal distribution of conditions across each position in a sequence
→ best for smaller sample sizes
latin square
table or square that ensures sequences of conditions are equally distributed across all columns and rows
reverse counterbalancing
reversing the order of a sequence of conditions and repeating it
→ e.g., ABCDDCBA
block randomization counterbalancing
assigning conditions in a sequence without repeating any conditions and starting a new sequence; ensures conditions are distributed evenly across all positions in a combination
e.g., ABCD → BADC → CDAB → DCBA
cross-sectional design
study design where different groups of participants are tested at one point in time
→ may be affected by cohort effects
→ less likely of loss due to attrition
→ requires larger sample sizes
longitudinal design
study design where the same group of participants are examined over multiple points in time
→ less likely to be affected by cohort effects
→ loss of participants due to attrition
→ a small sample is sufficient
cohort effects
people from peer/generational groups may have cultural/inherent differences from other groups
attrition
loss of participants who withdrew from the study
cohort sequential design
combination of longitudinal and cross-sectional design
follows different cohorts over a smaller period of time
→ minimizes attrition and cohort effects
experimenter bias
biases from the researcher that affect data
controlling experimenter bias: automated procedures
procedures that are automated by machinery or other means that do not involve the researcher
controlling experimenter bias: double blind procedures
neither the participant or the researcher know which group is the true IV and not the placebo
subject bias: hawthorne effect
participants will inherently behave differently when they are aware they are a part of research
subject bias: “good” subjects
subjects act purposefully/subconsciously act more cooperatively with researchers
subject bias: evaluation comprehension
participants are apprehensive when under research and tries to act ideally
subject bias: demand characteristics
when a participant slowly becomes aware of the study’s true hypothesis and begin to act differently in respect to the true hypothesis
controlling for subject bias: effective deception
proper deception to ensure participants don’t understand the true intention of the study
controlling for subject bias: manipulation checks
checking after the study (sometimes before) with participants to see if the deception was effective
controlling for subject bias: field research
conducting field research to avoid subject bias (observational studies help participants act naturally)
descriptive statistics
summarizes sample data into understandable metrics
inferential statistics
uses statistical methods to make inferences about the greater population given a sample
descriptive statistics: mean
measure of central tendency of the average of all points in a data
pros: representative of ALL data
cons: not resistant to outliers and skewness
descriptive statistics: mode
measure of central tendency of the most commonly occurring value in a data set
pros: useful for categorical data and is resistant to outliers
cons: not useful for continuous data and in small samples
descriptive statistics: median
measure of central tendency that finds the center value of a data set
pros: resistant to outliers in the data set
cons: not truly representative of data; not useful for inferential statistics
implications of outliers
outliers can skew measures of descriptive statistics and indicates there may be confounding factors
descriptive statistics: range
the minimum and maximum of a data set
pros: tells a researcher the range of a data set
cons: not resistant to outliers and overall not very useful
descriptive statistics: standard deviation
average distance of data points from the mean
pros: best measure of variability
cons: not resistant to outliers
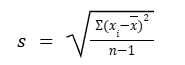
descriptive statistics: variance
standard deviation squared
pros: essential for ANOVAs
cons: not very useful outside of that
descriptive statistics: interquartile range
middle 50% of data
pros: resistant to outliers and useful when there is a lot of spread
cons: not truly representative of the other 50% of data
skewness
a data’s uniformity and where the data tends to drag toward
positive skew
tail of the data is dragged to the right
negative skew
tail of the data is dragged to the left
normal curve
uniform bell curve of a distribution
kurtosis
weight of the tails of a distribution and how the data is distributed there
platykurtic
the distribution is fairly flat with the tails and center of the distribution being fairly close in value
mesokurtic
the distribution is balanced across its tails and center
leptokurtic
the distribution is heavily centered around the middle and the tails are light
null hypothesis
there is no difference or effect between an IV and DV
→ not what the researchers want
alternate/research hypothesis
there is an effect between an IV and DV
→ what the researchers want
alpha level
chance of committing a type I error
→ proportion of times where the null hypothesis was true and the differences occurred by chance
type I error
rejecting the null hypothesis when it’s true
type II error
failing to reject the null hypothesis when the alternate is true
systematic variance
variance that have occurred due to the IV affecting the DV
error variance
variance that have occurred randomly unrelated to the IV
→ confounds
file drawer effect
studies where the null hypothesis was failed to be rejected and kept away from publishing because of insignificant results
importance of replicating failed to reject null hypotheses
replicating the study could refine the measure methodology and obtain an effect from the IV
→ type II error
effect size
magnitude of the effect of the IV and not simply whether it had a significant effect
confidence interval
interval in which the true population statistic is estimated to fall within given a level of confidence
power
chance of correctly rejecting a null hypothesis in research
→ 1 - beta
tables
useful for when multiple precise measurements and data are to be reported
bar charts
useful to illustrate the difference between levels of an independent variable
histograms
useful to illustrate the distribution of data points
pie charts
useful for categorical data and when data adds up to a proportion
line graphs
useful for illustrating the interaction between two IVs
scatterplots
useful for illustrating the correlation between two variables
first code of 1953: hobbs committee
gathered to create guidelines for psychological research using input from previous incidents and researchers
first code of 1953: critical incidents procedure
surveyed psychological researchers for input on ethical guidelines to put into place for research
APA ethical principles of psychologists and code of conduct
many revisions; most recent being in 2002
addresses guidelines for practicing, researching, and teaching of psychology
principles of ethics: beneficence and non-malevolence
weighing risks and benefits of research, protecting participants from risks, and produce research for greatest good
principles of ethics: justice
ensure everybody is able to benefit from research and be treated fairly
principles of ethics: integrity
meticulously transparency and honesty
principles of ethics: respect for people’s rights and dignity
respecting people’s rights and their rights to confidentiality and anonymity
principles of ethics: fidelity and responsibility
professionalism and responsibility of actions
ten standards of ethics
resolving ethical issues
competence
human relations
privacy and confidentiality
advertisements/public statements
recordkeeping and fees
education and training
research and publication
assessment
therapy
institutional review board (IRB)
board of individuals that oversees research approval in terms of their ethics, risk to participants, and benefits
IRB reviews: exempt
studies that pose no discomfort to individuals at all
→ survey studies about economics, educational purposes, literature reviews, training purposes, etc.
IRB reviews: expedited
studies that pose minimal risk to participants or be subject to manipulation
→ deception studies, surveys about mental illness, etc.
IRB reviews: full
studies that are beyond minimal risk for participants in some way
→ medication trials
IRB reviews: no risk
no physiological or psychology harm or discomfort
IRB reviews: minimal risk
minimal physiological or psychological harm or discomfort
IRB reviews: at risk/beyond minimal risk
beyond minimal risk of physiological or psychological harm or discomfort
elements of consent
→ sufficient details about the study and what participants will be expected to do and how long of a commitment they will have to make
→ assurance of confidentiality, anonymity, and voluntary leave if one wishes to do so
→ contact information of researchers and whether participants would like to receive the results of the study
→ signatures
deception
manipulation of participants to falsely believe something to have them act naturally
special populations in consent
prisoners, institutionalized individuals, pregnant women, children, etc.
debriefing
after (sometimes during) a study is complete, a researcher informs a participants of the true nature of the study
dehoaxing
the true hypothesis and intent for the study is revealed
desensitization
negative feelings and/or stress caused by manipulation is accounted for toward the end of research
concerns with internet research
difficulties properly obtaining consent, difficulties properly debriefing individuals, and concerns over privacy and confidentiality in internet spaces
concerns with animal use in research
concern of animal welfare in research and whether or not their use is necessary
institutional animal care and use committee (IACUC)
IRB-like committee that sets ethical guidelines for animal research and determines whether or not research using animals are ethical/permissible
→ benefits vs. risks
→ some benefits to animals as well
requirements for animal care: expertise
all researchers and assistants must undergo training before handling or caring for animals
requirements for animal care: veterinarian care
animals must have access to humane care such as medical care, housing, food, water, etc…
requirements for animal care: euthanasia
animals who are in any amount of pain or suffering as a result of research should be euthanized for their own wellbeing
requirements for animal care: wild/lab animals
animal research with wild or lab animals must undergo their specific procedures
wild: ethical catch/release, treatment, and insuring minimal impact to environment
lab: ethical treatment and humane living conditions
scientific fraud
committing acts of fraud and malicious doing in scientific research with the intent of deception
plagiarism
purposefully stealing others work and claiming it as ones own