Lab 6: Determination of the Ratio of Product Alcohols in the Luche Reduction of (-)-Menthone
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
what is the Luche reduction used for
for the regioselective reduction of alpha enones
what is the term to describe the catalyst needed (what does it do)
Lanthanide catalyst (it activates the alcohol solvent)
in what formation does Luche reduction occur in?
1, 2 fashion
what is the purpose of this experiment?
The Luche reduction can increase the stereoselectivity of chiral ketones
what does the stereoselectivity of chiral ketone depend on?
the concentration of catalyst
reagents used
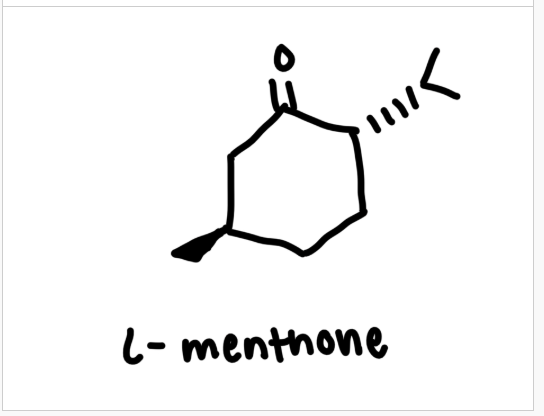
L-Menthone (structure, MW, BP, density)
Cerium chloride heptahydrate (structure, MW, MP)
Sodium borohydride (structure, MW, MP)
Ethanol (structure, MW, BP, density)
Diethyl ether (structure, MW, BP, density)
Magnesium sulfate (structure, MW, MP)
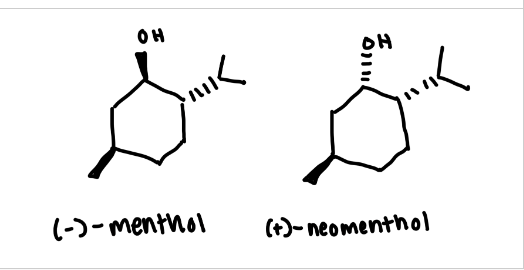
(-)-Menthol (structure, MW, BP, density, α°)
(+)-Neomenthol (structure, MW, BP, density, α°)
broadly describe the protocol
dissolve menthone into ethanol into small beaker
place in water bath then add NaBH4 and let the H2 bubbles go away. stir for about 5-10 mins
dilute in DI water and extract with diethyl ether (aka add diethyl ether to aqueous)
combine the organic layers and wash with (1) water then (2) brine (add these to organic)
dry with magnesium sulfate and remove solid with vacuum filtration (and any other remaining solvent with rotovap)
take IR
dissolve samples in 15 mL 95% ethanol and transfer to polarimetry tube. get the degree of rotation
what is the difference between luche and standard reduction reaction
Luche adds cerium catalyst to a standard sodium borohydride reaction. the purpose is the allow for a more regio and stereoselective reduction reaction. also, standard adds in 1,4 addition while luche does 1,2 addition
what kind of addition to Luche reaction favor
1,2 addition
normal sodium borohydride reactions will produce a racemic mixture. what will luche do?
it favors one an will produce enantiomeric excess of one
what position does Luche favor the most?
equatorial alcohols when reducing substituted, cyclic ketonesh
how do you find enantiomeric excess ratio?
find aº of mixture
then plug into second equation to find x (make sure you have the aº for methanol and ethnaol)
once you have x, (aka it will be x:1) ad one then find % methanol by taking the x value of methanol (numerator) over the total
what does x represent
methanol: neomethanol
polarimetry procedure
see photo

what dextrorotatory mean
when the darker half circle is to the right
what does levorotatory mean
darker half circle is on the left
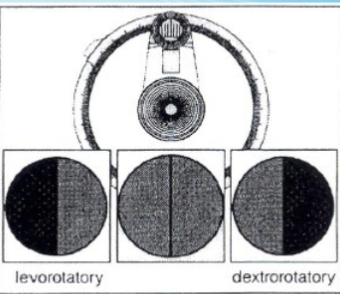
what instrument will you be using
SR-6 polarimeter
what are the alcohols formed in this experiment
menthol (wedge) and neomenthol (dash)
what is the reactant
L-menthone
When both the organic and aqueous layers have been transferred into the separatory funnel in the Luche reduction experiment, which layer will be on the bottom?
aqueous
How will the residual water remaining in the organic layer be removed prior to distillation in the Luche reduction experiment?
magnesium sulfate
What solvent will your sample be dissolved in for determining the optical rotation?
95% ethanol
general balanced equation
see photo
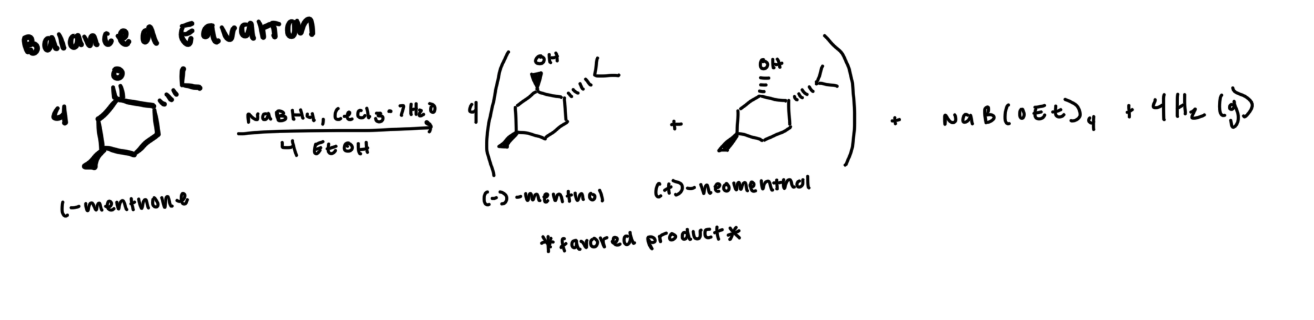
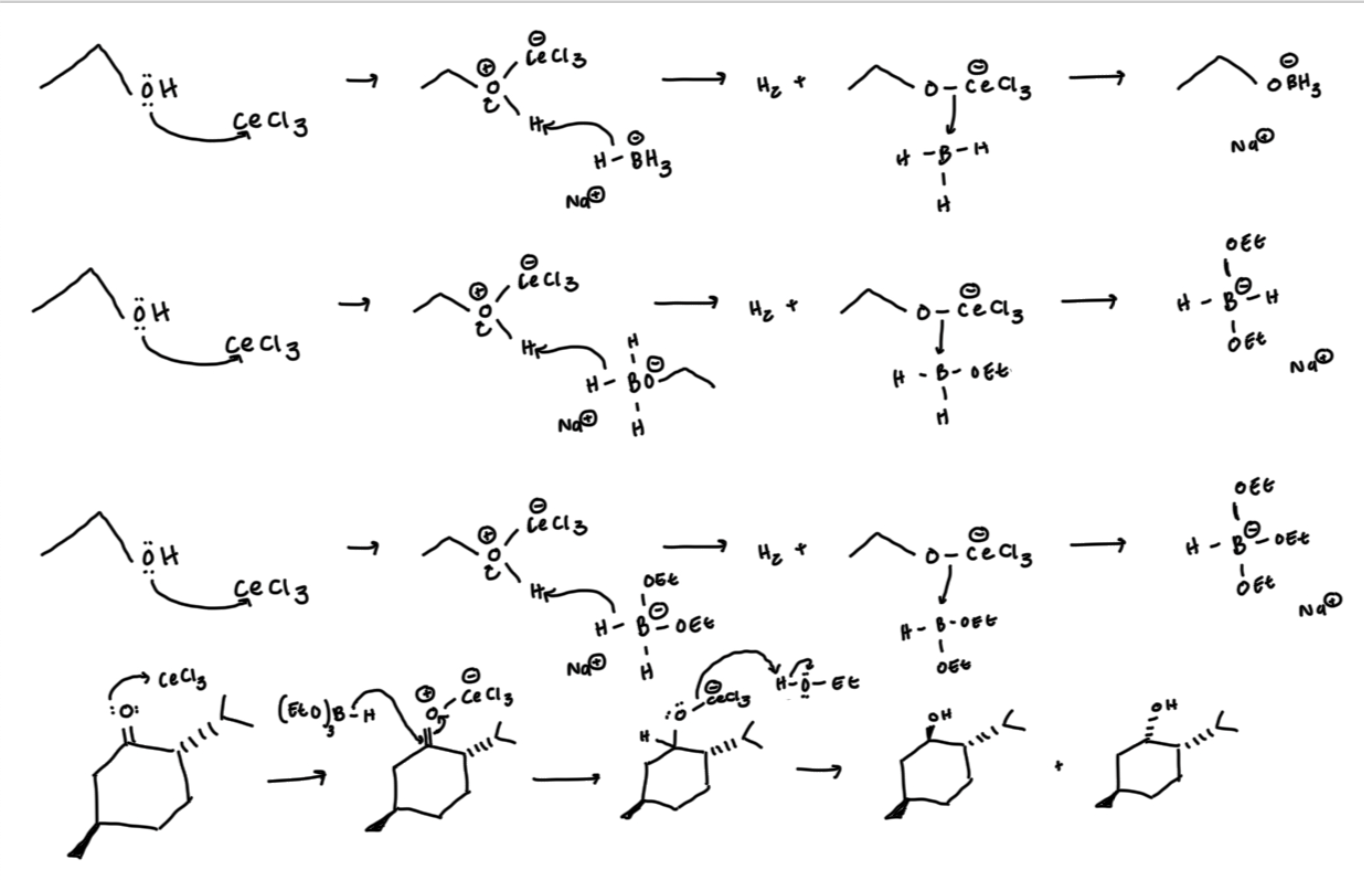
general reaction mechanism
you have to form your B(OEt)3H (aka the first three steps get the OEt group to connection with B
the bottom is the O-Ce, nuc attack by B(OEt)3H to push electrons from bond to O. then HOEt will protonate to form hydroxyl
limiting reagent
L-menthone
did i use IR to find final product? or the ratio? what about to tell if reaction went to completion
no you cannot find the ratio because they have the same functional groups
you will see the disapperance of the ketone group and the formation of the broad intense peak for OH
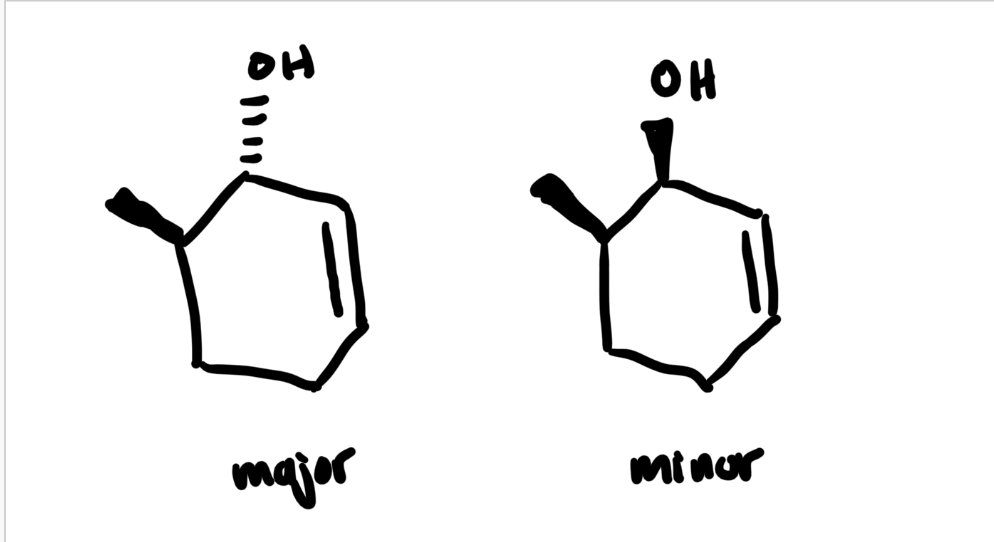
predict major/minor reactions
1, 2 addition is the major
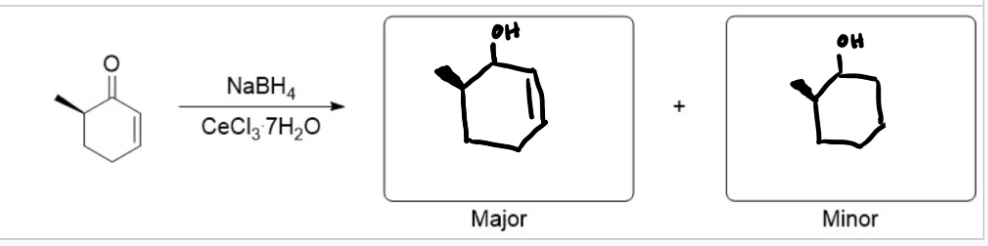
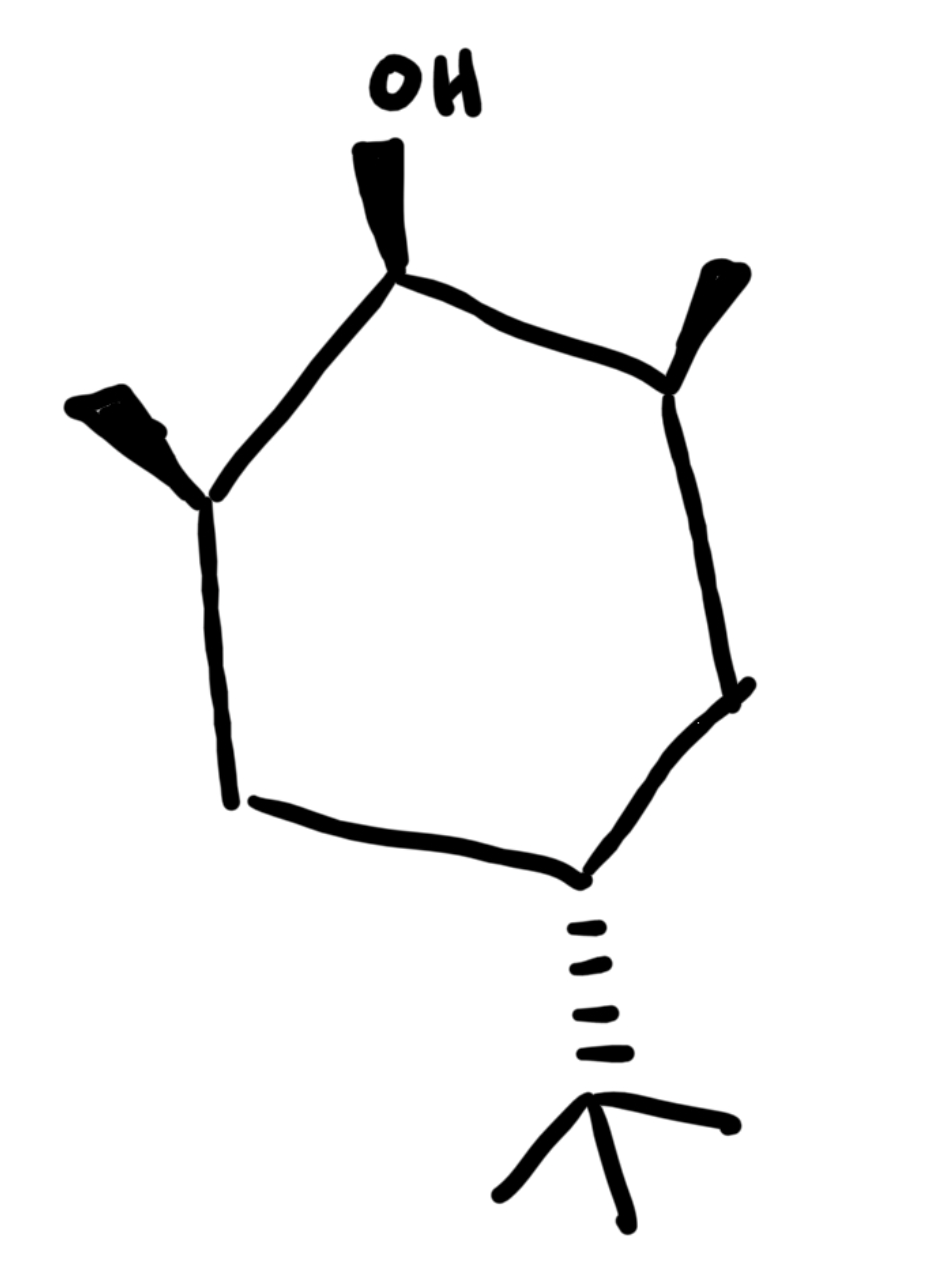
The Luche reduction can also be stereoselective, as you saw in this lab which gave you a mixture of diastereomers. Predict the major and minor reduction products considering only the stereochemistry (for this question, you may disregard regioisomers).
see image
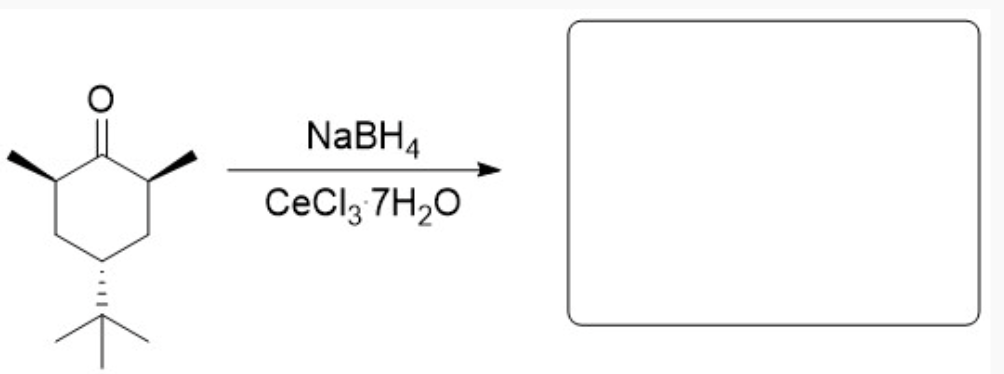
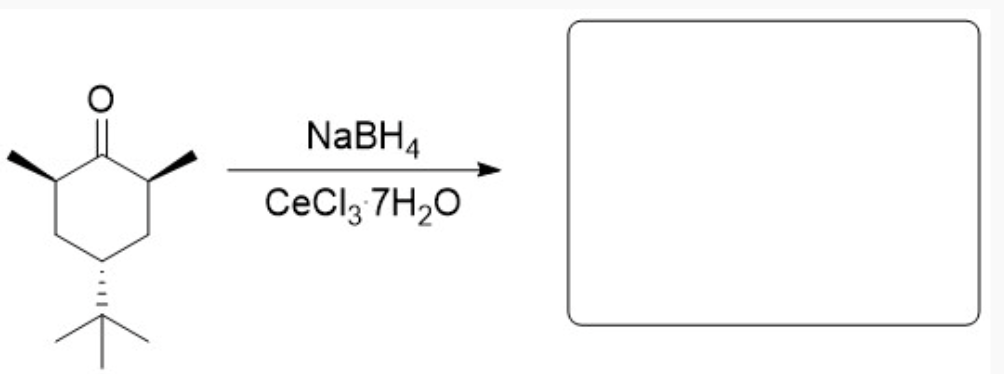
Why is only one product observed from this reaction? Explain your answer in complete sentences and include an image of the chair structure that adequately shows any relevant steric or electronic interactions that impact this reactivity.
Here, the tert-butyl group is bulky, so it needs to be in the equatorial position to minimize steric hindrance. Putting it in the axial positions results in overcrowding and steric hindrance, even if it is just two adjacent hydrogen groups. Having it in the equatorial position will result in a more stable, lower-energy chair configuration. Additionally, because the front side of the carbonyl is crowded by the two wedge methyl groups, the hydride shift must occur on the backside—the least sterically hindered face. The hydride will attack from the back, and ultimately, after protonation, the OH will end up on the opposite side, ending up on the front side. This results in the OH being in the equatorial position in the chair configuration.
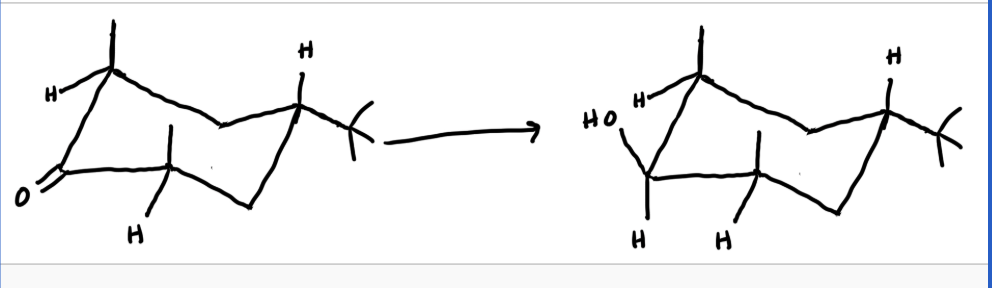
draw the non chair configuration with this
see photo