Exam 3 - MCB 442, ANSC - 450
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
Central vs Peripheral Tolerance
Central Tolerance - In primary or central lymphoid organs
Positive Selection - Able to weakly recognize MHC or functional
Negative Selection - Must not bind with high affinity to self peptides, induces clonal deletion
Peripheral Tolerance - Acquired by mature lymphocytes in peripheral tissues
Anergy - State of unresponsiveness, T cells that bind antigen in absence of co-stimulation
Immunoregulation - Inhibition by products of other cells
Ignorance - Sequestration of antigen(T cell is physically blocked from antigen)
B Cell Maturation
Generation of receptors(bone marrow) - Negative selection(bone marrow) - Migration to lymphoid organs and activation - Antibody secretion and memory cells(bone marrow + lymphoid tissue)
FLT3 - Drives commitment to CLP
CXCL12 - Keeps B cells in contact with bone marrow stromal cell
IL-7 receptor needed for proliferation
E2A - Transcription factor for lineage commitment, stimulates early B-cell factor(EBF)
Rearrangement of Heavy Chain Locus(Start of B cell development)
Stem cell - Early pro-B(DJ rearranging) - Late pro-B(V-DJ) - Large pre-B(VDJ)
E2A + EBF = VpreB, lambda5, RAG1/2, Pax5 = CD19,Iga
Rearranged VDJ segments combined with VpreB and lambda5 segments(surrogate light chain), Tests functionality
VpreB - Variable region substitute
Lambda5 = Constant region sub
Signaling stops rearrangement - two heavy chains could = two different receptors, Pre-B cell receptor signaling enforces allelic exclusion
Allelic exclusion = Reduces RAG1/2 expression, phosphorylation of RAG-2 for degradation, decreases access to heavy chain locus
4 possible fates for autoreactive B cells
Apoptosis(clonal deletion)
Receptor Editing - Make new receptor, stim of IgM in an immature B cell induces RAG expression = generation of new light chain
Anergy - Permanent unresponsiveness, cross-linking self antigen with low valence
Ignorance - Remain in circulation but does not see antig
Thymus
Site of T-cell maturation
Pathway: Precursor cells enter in corticomedullary junction - Migrate to the cortex, proliferate then migrate to the medulla - Migrate out of thymus to secondary lymphoid tissue
Cortex - Cortical epithelial cells, thymoctes(bone marrow origin)
Medulla - Hassal's corpsucle, dendritic cell, macrophage
Importance
T cells interact with thymic stromal cells(essential for inducing tolerance)
Mice that have undergone thymectomy are immunodeficient
Only 2-4% of cells in thymus leave, rest are dying/phagocytosed
T cell production rate is greatest before puberty and slows in adults
Removal of thymus after puberty is not associated with reduced T cell numbers
Bone Marrow Chimera studies importance of RAG and thymus
Lack ability to undergo recombination when no thymus
T cell Development
Requires Notch signaling and TXN factors
Notch Receptor Signaling - Needed for T-cell lineage commitment = TCF1 and GATA3 = RAG1 and CD3
Bcl11b induces lineage commitment
Notch Signaling = IL-7R
Similar to B cell maturation - DN stages characterized by expression of kit, CD44 and CD25
T Cell Positive Selection
CORTEX - Able to weakly recognize self MHC and peptide
Thymic cortical epithelial cells are responsible
Express both MHC 1/2 antigens
Binds with right affinity to MHC 1 = CD8 cell, MHC 2 = CD4 cell
Flow Cytometry
Determines surface expression of protein on cell, cell proliferation and cytokine production, cell viability, percent of positive cells
TXN Factors
Determine co-receptor fate
ThPOK(CD4) - Prepresses actions of Runx3
Runx3(CD8) - Represses CD4 transcription
Some CD4 cells up-regulate CD25 and express FoxP3, are natural Treg cells - Induced by binding self antigens with higher affinity than normal but not strong enough to cause deletion
T Cell Negative Selection
Deleting clones of T cells that bind too tightly to self peptide/MHC
In cortex + medulla
Likely dependent on bone-marrow APCs(more DCs than macrophage)
Thymic medullary stromal cells express tissue specific proteins that are not expressed elsewhere(AIRE - autoimmune regulator)
Affinity Hypothesis
No selection = death(low affinity)
Medium affinity = positive selection
High = Treg cells
Too high = negative selection(clonal deletion
Tregs
Naturally occurring(nTregs) = from thymus, CD25+, FoxP3+
FoxP3 - Transcription factor needed for Treg polarization
CD25 - IL-2R, on all activated T cells
Inducible Tregs are made from TGFbeta stimulation in periphery, function to maintain peripheral tolerance
Function of Suppression
CTLA-4 - Negative regulator of T-cell activation, binds CD80/86, inhibits DC and macrophage activation
Consumption of cytokines needed for T cell viability
Production of cytokines(IL-10, TGFB, IL-35) - Anti-inflammatory
Lymphocyte entry into 2nd lymphoid tissues is dependent on expression of chemokines/adhesion molecules
CD62L - Marker on naive T-cells, is needed for homing to the LN
L-selection - Binds to vascular addressins on HEV
Entry into LN parenchyma requires expression of integrins(LFA-1, ect) and chemokine receptors(CCR7)
Integrins bind to adhesion molecules(ICAM) - where T cells are needed
IFN-y, IL-2, LT-a/B
Th1 Cytokines(pro-inflammatory/Intracellular defense)
IFN-y - Macrophage activator, boosts MHC expression and helps B cells switch of IgG classes
IL-2 - T cell growth factor, needed for proliferation after antigen-T cell interaction
LT-a/B - Lymphotoxin, kills target cells during lymphoid organ development(similar to TNF)
IL-4, IL-5, IL-13
Th 2 Cytokines(Anti-parasitic/Allergy)
IL-4 - B cell switcher, tells B cells to make IgE, also helps naive T cells = Th2
Il-5 - Eosinophil activator
IL-13 - Similar to IL-4, increases mucus production in gut and lungs
IL-17A
Th 17 Cytokine
IL-17A - Recruiter of neutrophils, vital for fighting extracellular bacteria and fungi, big role in autoimmune inflammation
IL-10, TGF-Beta
Treg Cytokines
IL-10 - Brakes, inhibits Th1 cells and macrophages to prevent immune system from overreacting and damaging tissue
TGF-B - Generally anti-inflammatory but is also required to tell a T cell to become a Th17 or Treg
TNF, GM-CSF
Generally inflammatory + Growth Factors
TNF - Tumor necrosis factor, master of acute inflammation, activates vascular endothelium(leaky blood vessels, so cells can move)) and can induce fever
GM-CSF - Recruitment factor, stimulates bone marrow to produce more monocytes and granulocytes
Conventional DC vs Plasmacytoid DC
Conventional DC - T cell priming
Plasmacytoid DC - Interferon production
L-2R(CD25) alpha
On activated T cell surface, makes receptor high affinity, naive only have gamma + beta subunits
Stimulates proliferation, IL-2 acts in an autocrine + paracrine fashion
Instability sequence at 3' end makes RNA unstable
T Helper 1 Cell
Fights Intracellular infection (viral/bacterial)
Macrophage activating effector molecules, IFN-y, GM-CSF, TNF-a, IL-2
IL-12 induced STAT4 phosphorylation is required for TH1 polarization
T Helper 2 Cell
Fights Worms, part of allergic response
Barrier immunity activating effector molecules: IL-4,5,13 + IL-3,10
STAT6 - TH2 polarization
T Helper 17 Cell
Fights extracellular bacteria/fungi
Barrier immunity, neutrophil recruitment: IL-17A, 17F
IL-17A,F, IL-22, drives production of G-CSF + KC/CXCL1
G-CSF = neutrophil hematopoietic factor
KC = Neutrophil chemoattractant
IL-17 = Causes neutrophilia
Signaling for IL-17 requires Act1 and NFkB pathway
T Regulatory Cells
Immunoregulation
Suppressive cytokines: IL-10, TGFbeta
iTreg cells: TGF-β; STAT5; FoxP3; CTLA-4 expressio
Cytotoxic T cell Mediated Cell Death
CD8 attaches to target via LFA/ICAM
Recognition of MHC 1 = cytoskeletal rearrangement, directs cytotoxic granules toward cell
Release of granules induces cell death
Granzyme/perforin/serglycin complex ensures granzyme mediated cleavage of caspase 3
B-cell co-receptor Complex
CD19, 21, 81
Binding of both BCR to an antigen and CD21 or CR2 binding to complement proteins(C3d) on cell surface = B-cell proliferation and activation
Linked Recognition - B cell can only be activated by its cognate T cell that recognizes the same antigen, ensures tolerance
Because B cell activation requires linked recognition for activation, this means that both T and B-cells need to display a receptor for same antigen
B cell must come in contact with T-cells in order to receive help(otherwise they die)
Trafficking of activated B-cells toward T-cells = Upregulation of EBI2, CCR7, and continued expression of CXCR5
EBI2, CCR7, CXCR5
EBI2 - Recognizes 7a, 25-dihydroxycholesterol
CCR7 - Receptor for CCL21, CCL19(Tfh)
CXCR5 - Receptor for CXCL13(fDC)
All contribute to trafficking of activated B-cells towards T cells
Germinal Centers
Important for somatic hypermutation, affinity maturation, class switching, and formation of memory cells
Three zones(Mantle, light, dark)
Follicular dendritic cells CXCL13 to CXCR5(In light zone)
Stromal cell CXCL12 to CXCR4(in dark zone)
Dark zone is filled with proliferating B cells(stained green with (Ki67+)
Both light and dark is where somatic hypermutation occurs
Centrocytes - Reduced proliferation and CXCR4 expression, increased surface Ig
B cells can only enter dark zone if they are CXCR4 positive
ICOS and CD40 expression by Tfh cells are needed for germinal center maintenance
B-cell Pax5 and Bcl6 inhibit expression of transcription factors needed for differentiation into plasma cells
These factors are decreased prior to differentiation
Expression of BLIMP-1, transcriptional repressor is increased - Decreased CXCR5 and upregulates CXCR4 and a4B1 integrins
Some B cells differentiate into memory cells which divide slowly but maintain BCR specificity
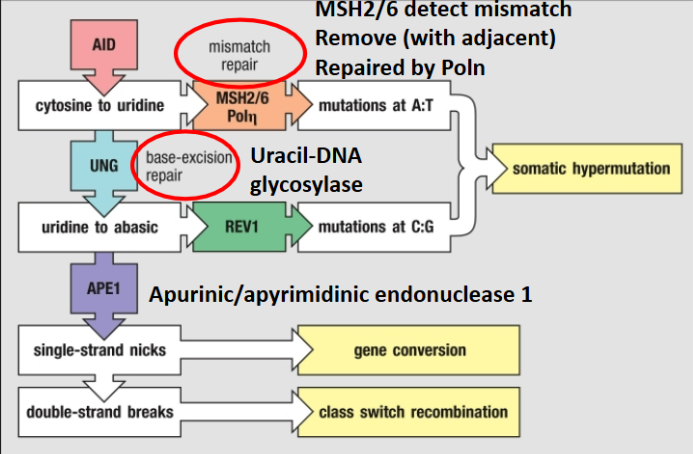
Activation Induced Cytidine Deaminase(AID)
Expressed by germinal center B-cells, causes random mutations in V genes
B cells with successful mutations can move to dark zone and undergo rounds of division
If mutations weakens affinity for antigen it will no longer receive T-cell help(Dies)
If mutation strengthens affinity, it receives T cell help and will proliferation and differentiate
Is positive selection, ensures only a few B-cell clones are highly responsive to the antigen
AID Mechanism:
Switch regions = G-rich, as transcription occurs G-rich RNA invades DNA duplex and binds to C-rich template strand creating an R-loop, this R loop provides a binding site
AID = C to U mutations, processed by Uracil DNA Glycosylase(UNG) and APE1 creating DNA nicks on both strands
DNA is cut and sewed to different antibody constant regions = Class switching
Just AID or just UNG mechanism = somatic hypermutation
Class Switching(B cell)
Directed process as the isotype that the antibody switches to is determined by the type of Th-cell cytokines are made
Cytokine-specific receptor = which isotype is created by dictating which C gene segments are transcriptionally expressed
Class switching only occurs after stimulation with antigen
IL-4 = IgG1, IgE
IFN-y = IgG3, IgG2a
TGF-B = IgG2b, IgA
IL-21 = IgG3, IgG1, IgA
IL-5 = IgG1, IgA
Humoral Response: TI antigens
BCR-activation is first signal for antibody production
For thymus independent antibody generation costimulation is from activation of PRRs by PAMPs
Thymus Independent Antigens
TI-1 - Activate B-cells without antigen specificity
Polyclonal Activation - Activation of more than one B-cell clone - leads to B-cell mitogens: Induce mitotic division; LPS
TI-2 - Activate only mature B-cells, have highly repetitive structures
Infants do not have mature B-cells so cannot elicit an antibody response to polysaccharides
Made predominantly by B-1 cells
Provide a prompt antibody response against pathogens(IgM) but can undergo class switching
Follicle Periphery
Antigen recognition - Antigen presentation - Proliferation/effector function, cytokines circle back and activate B cells(same as antigen recognition)
IL-21 promotes B cell proliferation
IL-4 + IFN-y will influence class switching
IgM
First antibody that is made
Largest antibody
Highest avidity but lowest affinity for antigen(10 binding sites)
Most effective at complement activation
Largely made by B-1 cells in spleen
Effective at complement activation(C1q binds to one Fc region)
IgA
Made after class switching, IL-5 enhances IgA production
In secretions, two subclasses(IgA1,2)
Has neutralizing capacity(IgA1)
Not a good opsonin or good at complement activation
Can form dimers(IgA2)
Dimeric IgA can bind to polymeric immunoglobulin receptor(plgR)
Transcytosis - Process of transporting IgA(and IgM to lesser extent) across the epithelial barrier
IgG
Made after class switching
5 subclasses(IgG1, IgG2a, IgG2b, IgG3, IgG4)
Has neutralizing capacity
Principal antibody in blood and extracellular fluid
Can activate complement(worse than IgM)
IgE
Made after class switching(IL-4)
Potent inducer of Mast cell activation
Also binds FceRII on eosinophils and basophils
Is monomeric
Mast Cells
Cross linking two IgE molecules on surface causes degranulation
3 Functions
Histamine = vasodilation
Immune cell attraction - Lipid inflammatory mediators, prostaglandin D2, leukotriene C4, cytokine production(TNF)
Muscle contraction - Physical expulsion
Active vs Passive Immunization
Active immunization
Require generation of adaptive immune responses
Long lived = memory
Passive Immunization
Do not need to generate an immune response
Short lived
Some toxins are too toxic to vaccinate against
Inject small amounts into larger animals, generate antibody
Injection of antivenin(purified IgG) into person that has been bitten
Referred to passive immunization
B cell and natural killer cell positive feedback loop
Binding of Fc receptors on NK cells promotes perforin and granzyme secretion
B cell secrete IgG3 = activates NK cells = IFN-y = activates B-cell
Innate Lymphoid Cells
Shape effector functions of adaptive immunity
4 Groups(found mostly at epithelial barriers) - Cytotoxic ILC, group 1, group 2, group 3
Cytotoxic ILC(NK) - Viruses, activated by IL-12,15, makes IFN-y
ILCs 1 - Type 1 immunity(intracellular bacteria) - IFN-y, Il-2, TNF, Macrophage activation, granuloma formation, IgG2a, IgG3
ILCs 2 - Type 2 immunity(Helminths) - IL-4,5,13, Mucous production, SM contraction, IGE, IgG1, Eosinophil and mast cell activation, granuloma formation
ILCs 3 - Type 3 immunity(Extracellular bacteria) - IL-17A, IL-17F, IL-22, Neutrophil attraction
NK cells vs ILC1
Both innate immune cells, both produce IFN-y
ILC1 progenitor is different
NK cells present in blood and spleen, ILC reside in tissue
NK cells express cytotoxic granules
CXCL10, CCL 5,17,22,20
CXCL10, CCL5 will attract Th1 cells
CCL17, CCL22 will attract Th2 cells
CCL20 will attract Th17 cells
Naive Cells express CD62L(LFA) and CCR7
Granuloma Formation
Granuloma formation is dependent on Th1 responses
Microbes are walled off from surrounding tissue by macrophages and T cells
IFN-y enhances macrophage killing of pathogen by causing upregulation of iNOS and production of ROS and RNS
Comes at a cost - Tissue in center is destroyed(caseous necrosis) by intense ongoing immune reaction
ILC1 + Th1 = IFN-y = Inducible nitric oxide synthase(M1), arginine reacts with iNOS = NO = Toxic to intracellular microbes
IL2 + Th2, functions
= IL-4,13 = Arginase 1, Arginine + arginase 1 = Ornithine and proline = Toxic to worm, SM contraction, tissue remodeling
Immunological Memory
Maintenance is dependent on IL-7, IL-15
Memory T cells express many markers of activated T cells but not others
Important Marker: IL-7Ra(CD127)
IL-7 needed for CD4/8, IL-15 needed for CD8 maintenance
Memory Cells
Classified by surface markers that control localization
Naive T cells - CCR7(localization to LN) and CD45RA
Central Memory T Cells - CD45RO
Effector T Cells - Cytokines and CD69(Lack CD127)
Effector Memory T Cells - CD45RO and non-lymphoid homing receptors
Resident Memory T Cells - CD69 and CD127(IL-7Ra)
B Cell Immunological Memory
Memory B cells respond more rapidly than naive B cells
Usually have undergone class switching and affinity maturation
Distinguished by surface markers - Naive = IgM, IgD - Memory = CD27, slightly higher MHC 2 and CD80 able to re-enter germinal center
Original Antigenic Sin
From Thomas Francis, Tendency to make antibodies against epitopes expressed on first exposure to flu even if exposed to different variants with highly immunogenic epitopes
Absorptive Enterocyte
Uptake of nutrients/fluids, microbial and metabolic sensing, transport of secretory immunoglobulins
Microfold(M) cell
Antigen uptake, bacterial translocation
Goblet Cell
Production of mucins, antigen uptake
Paneth Cell
Has lysozyme, secretion of antimicrobial peptides - Support for intestinal stem cells
Tuft Cell
Il-25, sensing of helminthic odorants and succinate, mobilization of ILCs 2 via IL-25 and eicosanoid release
Peyer’s Patch
Surveillance and antigen sampling in intestines
Traffics antigen to mesenteric lymph nodes
Consists of T,B, M, and dendritic cells
Cryptopatches
Functions like a smaller Peyer’s patch
ILC 3 instead of B and T cells
Function on tissue repair and precursor for larger follicles
Isolated Lymphoid Follicle(ILF)
Antibody factory in intestines
Mainly makes IgA
Ensures that sections far away from Peyer’s Patches still have antibodies
Gut-specific homing by antigen-stimulated T and B cells
Gut lymphocytes express CCR9 which binds CCL25(epithelial cells in small intestine) or CCL28(colon, mammary/salivary glands)
Integrin a4:B7 on lymphocytes binds MAdCAM-1 on gut endothelium
Antigen encounter outside gut induces a4:B7 binds VCAM1 and CCR4 expression = licenses cells to skin
Encountering antigen IN gut induces expression of a4:B7 which directs cells to mucosa
Intestinal Macrophages
Replenished from blood monocytes
Highly phagocytic
Do not readily respond to PAMP by making proinflammatory cytokines
Product large amounts of IL-10 - ACT AS IMMUNE SUPPRESSORS TO PREVENT DAMAGE