Organic Chemstry reactions
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
You have the reagents HX (could be HCl, HBr, or HI), what is this and what do you do?
This is Hydrohalogenation. You add H and X. It is Mark and has mixed steriochemistry
You have the reagent H3O+ (dilute H2So4). What do you do? What is this?
acid catalyzed hydration. You add H and OH. It is mark and has mixed steriochemistry (both syn and anti)
You have the reagent
1) Hg(OAc)2, H2O
2) NaBH4
what do you do? What is this ?
It is Oxymercuration-Demercuration. You add H and OH. It is anti-mark and has mixed steriochemsitry
You have the reagent H2, Pt.
What is this? What do you do?
This is Catalytic Hydrogenation. You add H and H. It has Syn steriochemistry.
You have the reagent Br2 or Cl2. What is this? What do you do?
This is Bromination or Chlorination.
You add Br and Br or Cl and Cl. It has anti steriochemistry
You have the reagent Br2, H2O. What is this? What do you do?
Halohydrin
You add Br and OH.
Oh goes to the most substituted. The steriochemistry is anti.
This is Halohydrin formation
You have the reagent KMnO4 or OsO4/NMO. What is this? What do you do?
This is syn dihydroxylation.
You add OH and OH. The steriochemistry is syn.
You have the reagent RCO3H (eg MCPBA). What occurs? What is this called?
This is epoxidation. This adds to one face. Forms epoxide
You have the reagent
1) RCO3H
2)H3O+
What is this called?
What does it do?
Anti dihydroxylation.
Adds OH and OH in an anti steriochemsitry
you have the reagents
1) O3
2) DMS or Zn/ H2O
What is this called? What does this do?
ozonolysis.
cleave occurs at C=C and forms 2 C=O groups.
For a terminal alkyne, Markonikov hydration yields a _____
ketone
for a terminal alkyne, anti-markonikov hydration yields an _____
aldehyde
If you have an alkyne and H2 pt, what do you get?
You get your typical 2 H’s distributed along the bond in a syn formatoin. Alkane
if you have an alkyne and H2 + lindlar’s catalyst, what will you end up with ?
cis alkene
if you have an alkyne and Na + NH3(l), what will you result with
trans-alkene
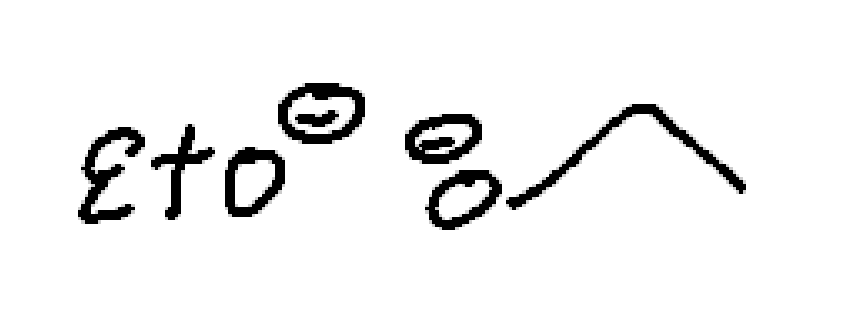
strong nucleophile
strong base
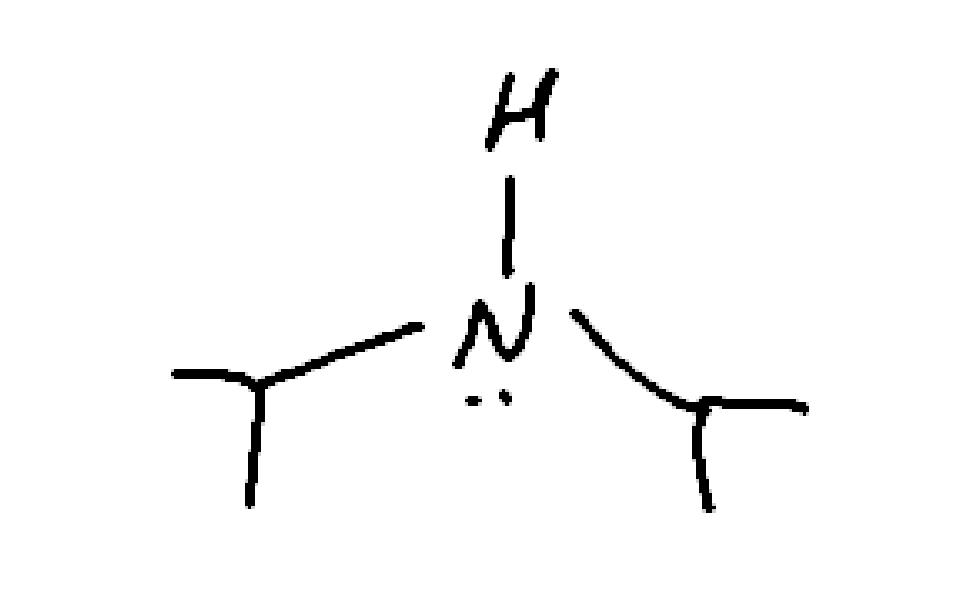
What is this
strong bulky base
diisopropylamine
If a bimolecular reaction with both nucleopphiles and bases exists and there are sterics (2 and 3) what do we have?
E2
If you have strong nucleophiles and a weak/moderate base, you will have what
SN2
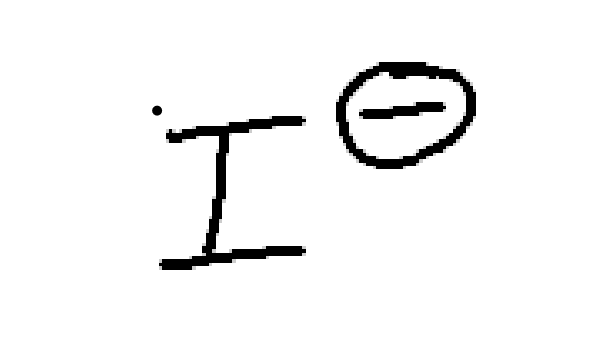
strong nuc, weak/moderate base
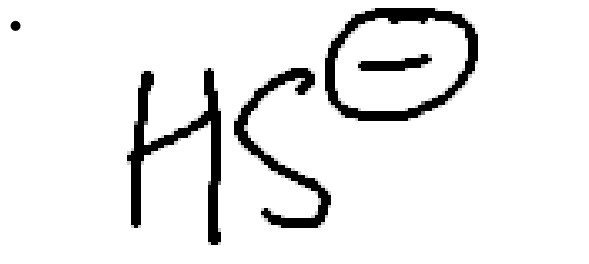
strong nucleophile, weak/moderate base
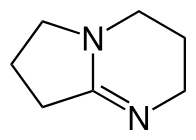
DBN?
Weak nucleophile, strong base
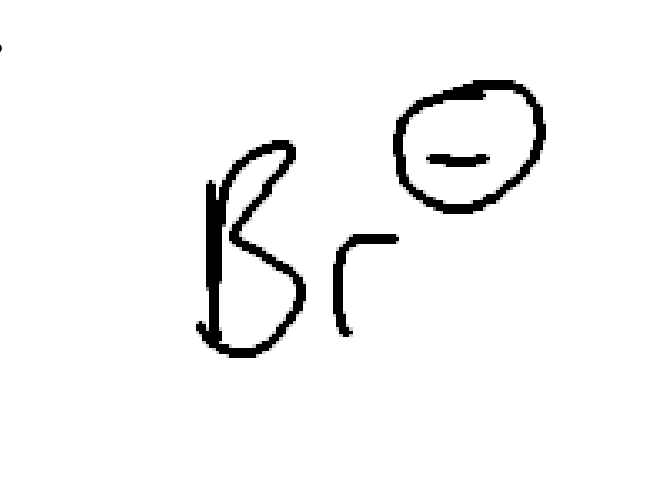
strong nucleophile and weak/moderate base
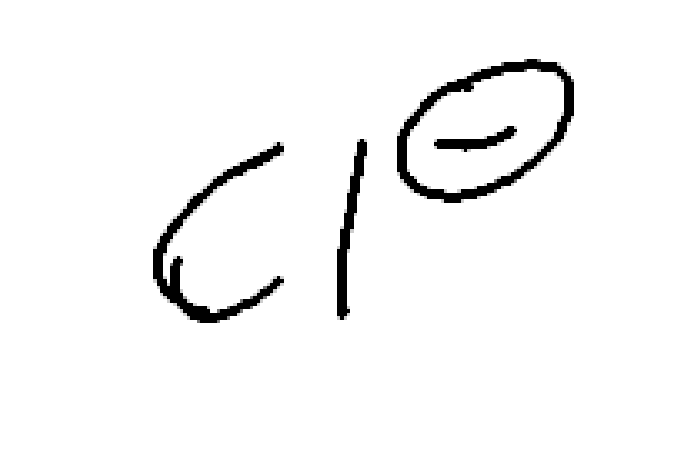
Strong nucleophile and weak/moderate base
What will donate a hydrogen in Oxymercuration Demercuration?
NaBH4
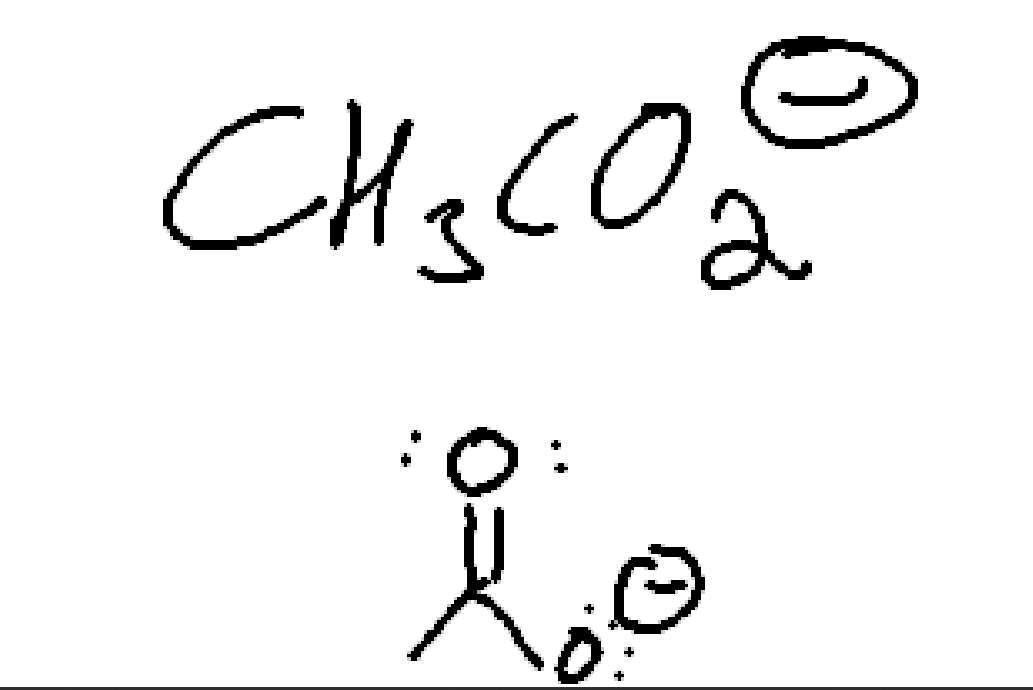
Acetate.
Strong nuc and weak/moderate base
What occurs in an anti-markovnikov situation?
H is added to the more substituted carbon. OH is added to the less substituted carbon
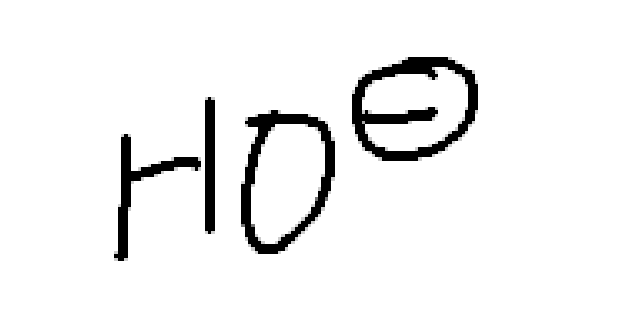
strong nucleophile, strong base
According to markovnikov, hydrogen adds to the carbon with what? halogen adds to whawt?
it adds to the carbon with more hydrogens (less substituted)
halogen adds to the more substituted carbon
If the bimolecular reaction is both nucleophilic and basic, what should we do?
look at substrates
If the bimolecular reaction is base only, what kind of reaction do we have?
E2
What does the presence of H30+, H20 {H2SO4 ——→ H20} represent in acid catalyzed hydration?
presence of water and an acid source
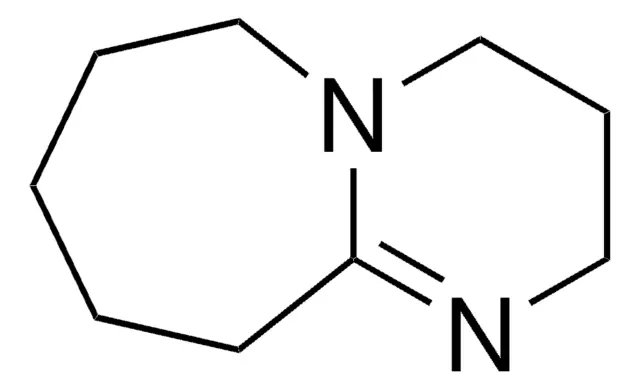
DBU
Weak nuc strong base

What is this?
Potassium tert-butoxide. Strong bulky base.
if the bimolecualr reaction is nucleophile only, what type of reaction will you have?
SN2
if a weak and stable reaction (unimolecular) exists, what should we do?
We should look at substrates
If you have a weak nucleophile and strong base you will have
E2
If you have a strong nucleophile and a storng base, you will have
SN2/E2
If a unimolecular reaction has the substrate number of 2 or 3, what do you have?
SN1/E1
If you have a weak nucleophile and wak base, you will have…
SN1/E1
What type of reaction will you have if the reaction reagent is weak and stable?
carbocation unimolecular reaction
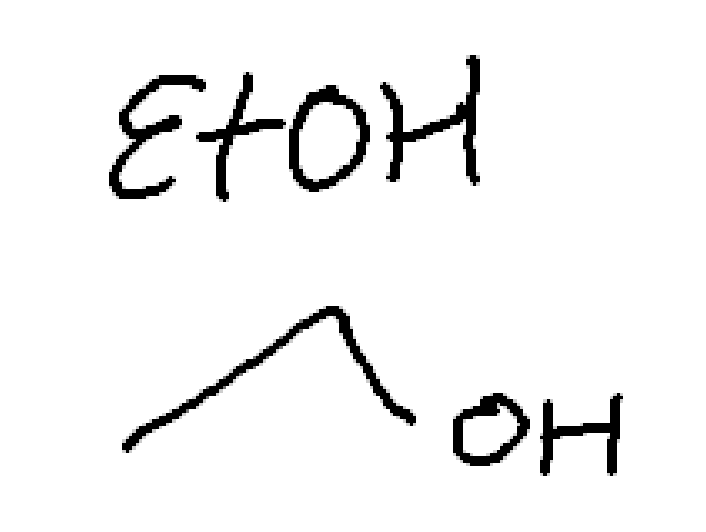
weak nuc, weak base
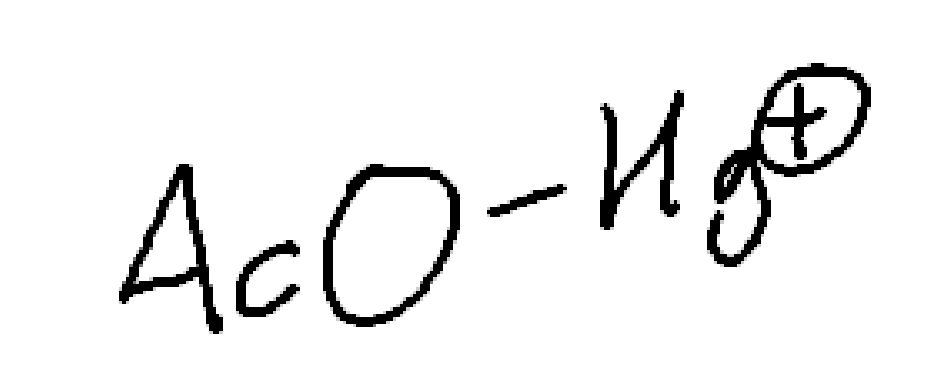
What is this
a good electrophile
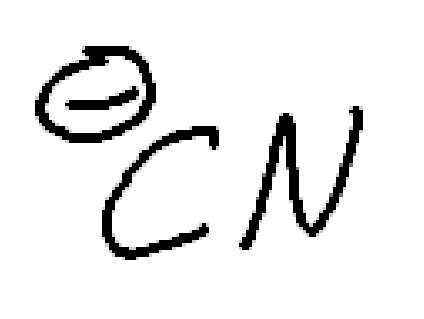
strong nucleophile, weak/moderate base
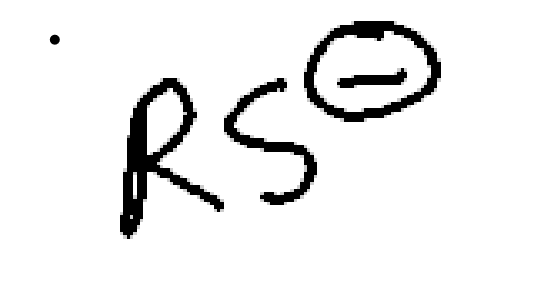
strong nucleophile, weak/moderate base
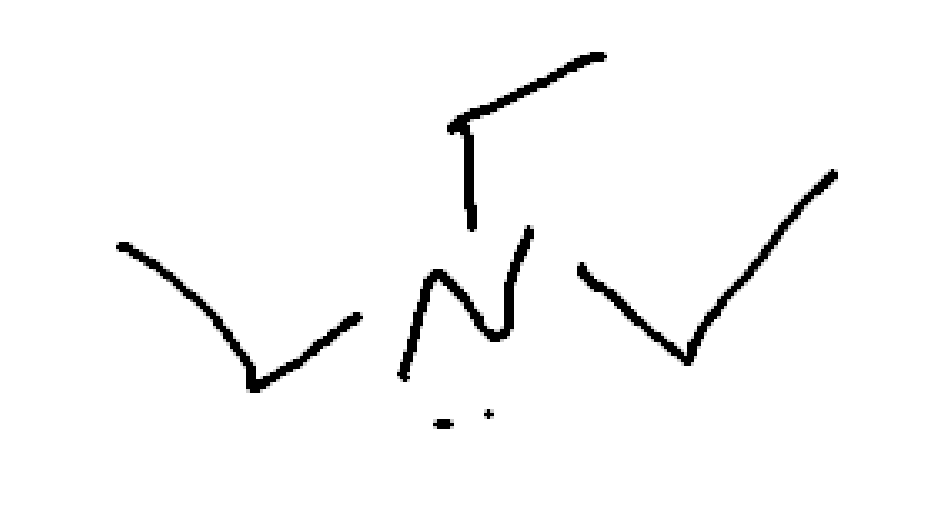
What is this?
triethyl amine
strong bulky base
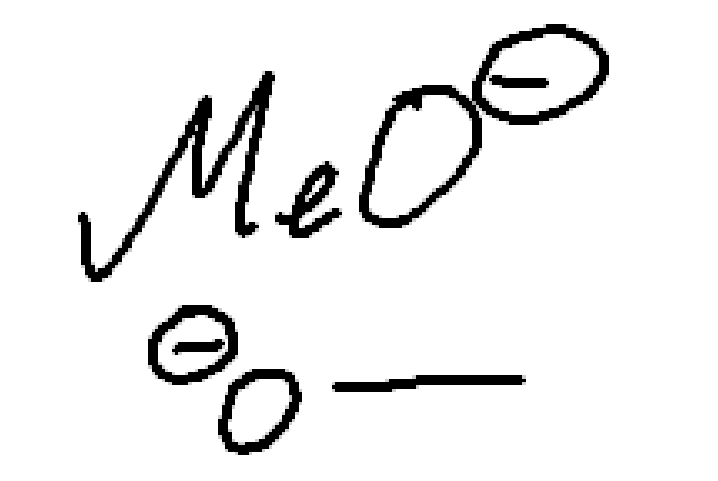
Methoxide
strong nucleophile
strong base
If a bimolecular reaction with both nucelophiles and bases exists and there are no sterics (1), what do we have?
SN2
What type of reaction do you have if the reagent is strong and reactive?
Attack
Bimoelcualr reaction
In a unimolecular reaction, if the substrate number is 1 what do we have
No reaction due to unstable C+
What is H20
weak nucleophile, weak base
What is the pattern of acid catalyzed hydration reactions
PAD
Protonate, Attack, Deprotonate
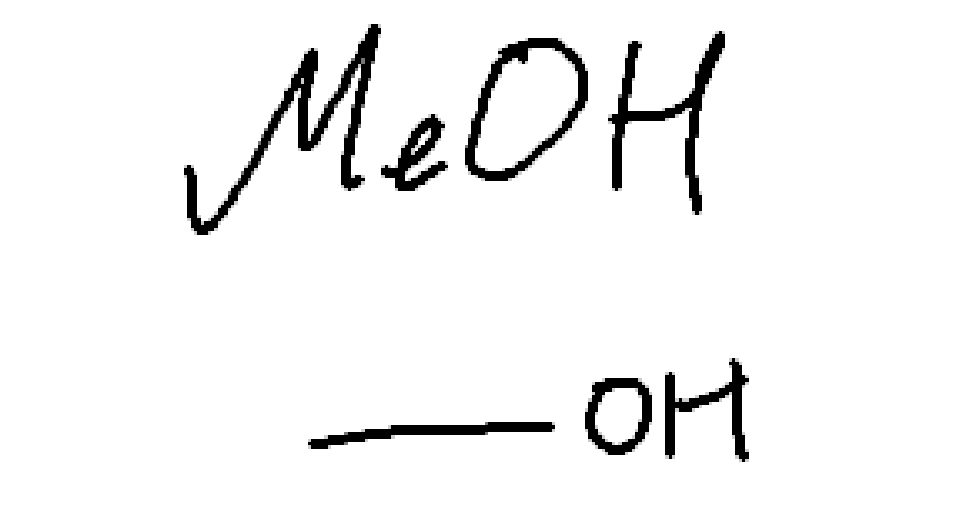
weak nuc, weak base
In acid catalyzed reactions, what do we deprotonate
OH2