Exam 3 prep - N cycle!
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
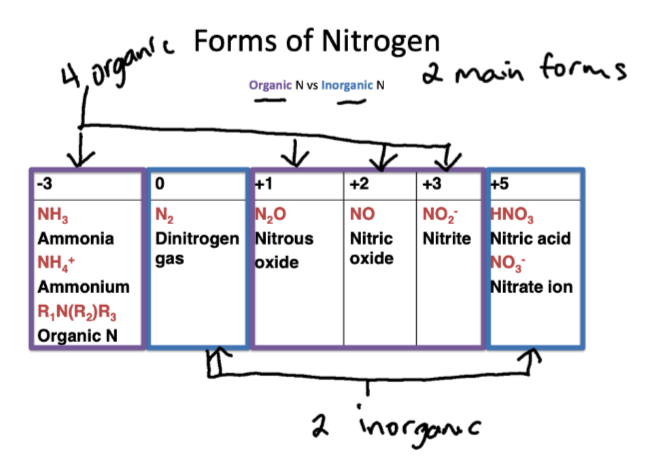
General forms of N
inorganic and organic N
Forms of inorganic N
dinitrogen gas (N2)
Nitric acid (HNO3)
Nitrate ion (NO3-)
Forms of organic N
ammonia (NH3)
ammonium (NH4+)
organic N
Nitrous oxide (N2O)
nitric oxide (NO)
nitrite (NO2-)
oxidation reactions
have increasing oxidation number

reduction reactions
have decreasing oxidation number

Microbes driving the N cycle - N2 in atmosphere
fixation by bacteria and archaea
to form organic compounds with amino (NH2) groups that are used by plants and animals (decomposition of proteins and nucleic acids)
Microbes driving the N cycle - ammonia
nitrification by bacteria into nitrite (NO2-)
Microbes driving the N cycle - nitrite
nitrification by bacteria to nitrate (NO3-)
Microbes driving the N cycle - nitrate (NO3-)
denitrification by bacteria and archaea to N2 in atmosphere
N Reservoir
most in the atmosphere — but it is NOT biologically available
lots in sediments and rocks too but not in available organic pools
plants and soils are the next largest pool
Dinitrogen gas (N2)
relatively inert and inactive
most stable form
~78% of the atmosphere and is the largest reservoir of N
SLOWLY used
microbes and humans use it
via N fixation
Name the steps of the N cycle
N fixation
N assimilation
Ammonification
Nitrification
Denitrification
Sedimentation
Nitrogen fixation
the conversion of free N of atmosphere into the biologically acceptable form or into nitrogenous compounds
3 ways to convert N2 into more chemically reactive forms via:
1) biological N fixation
2) physiochemical N fixation
3) industrial N fixation
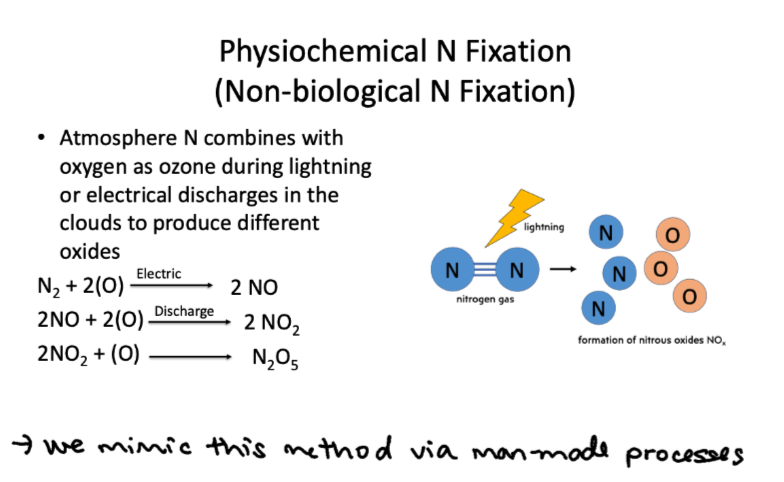
Nitrogen fixation - physiochemical N fixation
atmosphere N combines with oxygen as ozone during lightning or electrical discharges in the clouds to produce different oxides
Nitrogen fixation - industrial N fixation
man-made process that converts N from the atmosphere into a form that can be used to make fertilizer for plants
via Haber-Bosch method
Haber-Bosch process
produces fertilizer by combining hydrogen and nitrogen under high pressure and temperature.
• Fritz Haber (1918) and Carl Bosch (1931) = Nobel prizes
• “Most influential persons of the 20th century” (Nature, 1999)
• Combine N (air) with H at high temps (>400C) and pressure (>200 atm) to get NH3
VERY energy intensive
3 CH4 + 6 H2O → 3 CO2 + 12 H2
4 N2 + 12 H2 → 8 NH3
• “detonated the population explosion” through the “Green Revolution”
Nitrogen fixation - Biological N fixation
process by which certain bacteria convert inorganic atmospheric nitrogen into organic N, making it accessible to plants.
– N2 + 6H+ + 6e- → 2NH3– Requires energy (ΔG = 630 kJ/mole)
discovered by Beijernick (bacteria had already been doing this for a while, he just described the process)
performed by symbiotic bacteria, symbiotic blue-green algae, and free-living bacteria
many bacteria and archaea
diazotrophs
cyanobacteria, rhizobia, frankia
nitrogenase enzyme
very O2 sensitive
Strategies for protecting nitrogenase
avoid (escape) - move to lower O2
protect - make proteins to protect nitrogenase under O2 exposure
O2 scavenging - increase O2 update by increasing respiration
separation in space - form special cells to perform N fixation
heterocysts in cyanobacteria
nodules in plants
separation in time - fix at night when O2 is lower due to decreased photosynthesis
Types of N fixers - Symbionts
rhizobia and legumes
Actinomycetes (aka Frankia) and woody plants
Types of N fixers - Associative
Azotobacter
live in rhizosphere
Types of N fixers - Free-living
cyanobacteria
also in soils