GCSE Chemistry: C7 - Energy changes
1/108
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
109 Terms
Describe what happens in exothermic reactions
- exothermic reactions transfer energy from the reacting molecules to the surroundings
- this means temperature of the surroundings increases in an exothermic reaction
- in other words, the surroundings get hotter
Name some examples of exothermic reactions
Examples of exothermic reactions:
combustion
certain oxidation reactions
neutralisation
What does combustion mean?
Combustion means burning, and this is an example of an exothermic reaction.
How do scientists represent energy changes taking place in reactions?
Scientists represent energy changes taking place in reactions by drawing an energy profile diagram.
Interpret a diagram of the energy profile for an exothermic reaction
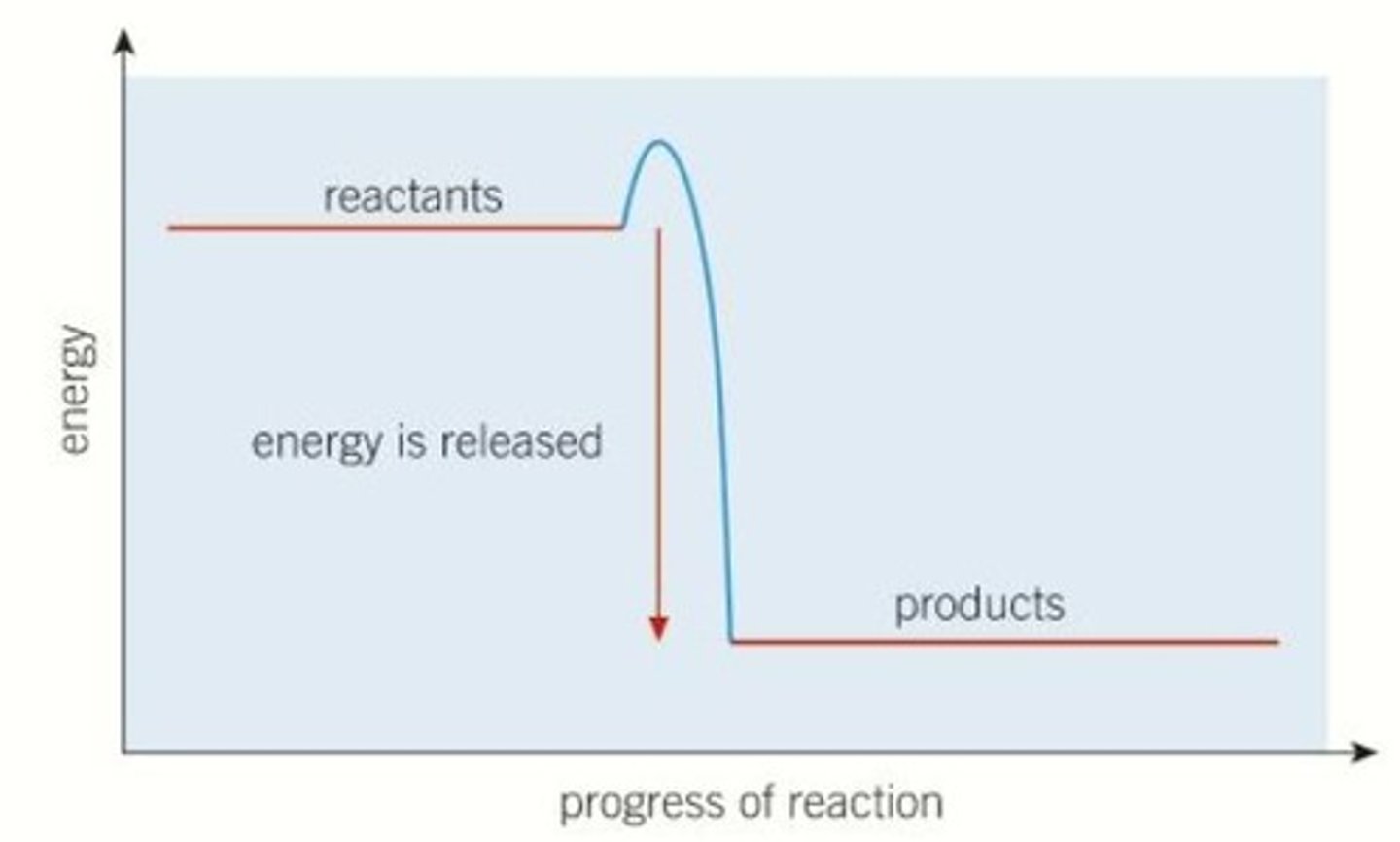
Describe what the energy profile for an exothermic reaction tells us
- in an exothermic reaction, we can see from the energy profile diagram that the products have less energy than the reactants
- the products have less energy as energy has been released from the reacting molecules into the surroundings
- difference between energy of reactants and energy of products tells us the energy that has been released to the surroundings
What is important to bear in mind for an exam question on energy profile diagrams?
In the exam, you could be asked to label an energy profile showing the energy change.
How can we label the energy change on an energy profile diagram?
Draw a dotted line from the end of the reactants line across to the right.
Then draw a dotted line from the end of the products line.
Draw a downwards arrow from the reactants dotted line to the products dotted line.
Name some uses of exothermic reactions
Uses of exothermic reactions:
- hand warmers
- self-heating cans, e.g. for food and drink
Describe what happens in endothermic reactions
- in endothermic reactions, the reacting molecules take in energy from their surroundings
- this means, in endothermic reactions, the temperature of the surroundings decreases
- in other words, the surroundings get colder
Name an example of an endothermic reaction
An example of an endothermic reaction is thermal decomposition.
Name a use of endothermic reactions
Sports injury packs are a use of endothermic reactions.
What does it mean if the products of a reaction store more energy than the reactants?
- if the products of a reaction store more energy than the reactants, it is an endothermic reaction,
- as the reacting molecules have taken in energy from the surroundings
- so the products store the same amount of energy as the reactants, but plus the energy taken in during the endothermic reaction
Explain why melting is an endothermic process
- melting is an endothermic process,
- because a substance in the liquid state stores more energy than a substance in the solid state,
- so energy must have been taken in during melting for the reacting molecules to have gained energy
Explain why freezing is an exothermic process
- freezing is an exothermic process
- as a substance in the solid state stores less energy than a substance in the liquid state
- so energy must have been transferred to the surroundings during freezing for the reacting molecules to have lost energy
What is meant by spontaneous reactions?
Spontaneous reactions are reactions that can occur without any external input.
Suggest why most spontaneous reactions are exothermic
- in endothermic reactions, energy is taken in, so this means most endothermic reactions need the reactants to be heated so they have the activation energy needed for this reaction to take place
- this means endothermic reactions are less likely to be spontaneous than exothermic reactions
Interpret a diagram of the energy profile for an endothermic reaction
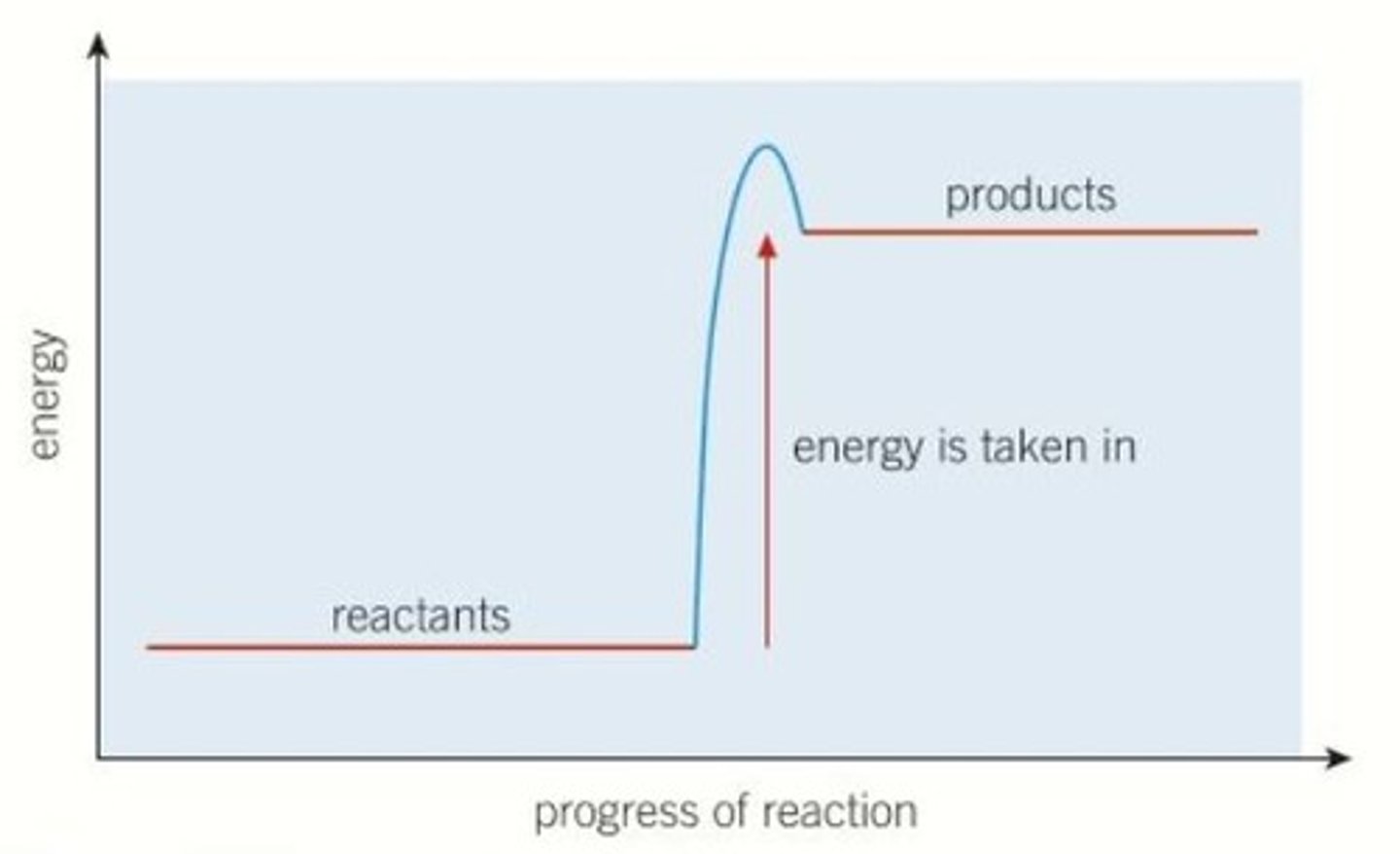
Describe what we can tell from the energy profile for an endothermic reaction
- in an endothermic reaction, the products have more energy than the reactants
- the products have more energy as energy has been taken in from the surroundings in the endothermic reaction
The difference in energy between the reactants and products tells us the amount of energy that has been taken in by the reaction.
What happens to the energy at some point in both the energy profiles for both exothermic and endothermic reactions?
The energy rises to a peak in the energy profile diagrams for both exothermic and endothermic reactions.
What must happen for a reaction to take place?
Reactions can only take place when the particles collide with each other with sufficient energy.
What is activation energy?
activation energy is the minimum amount of energy that particles must have in order to react
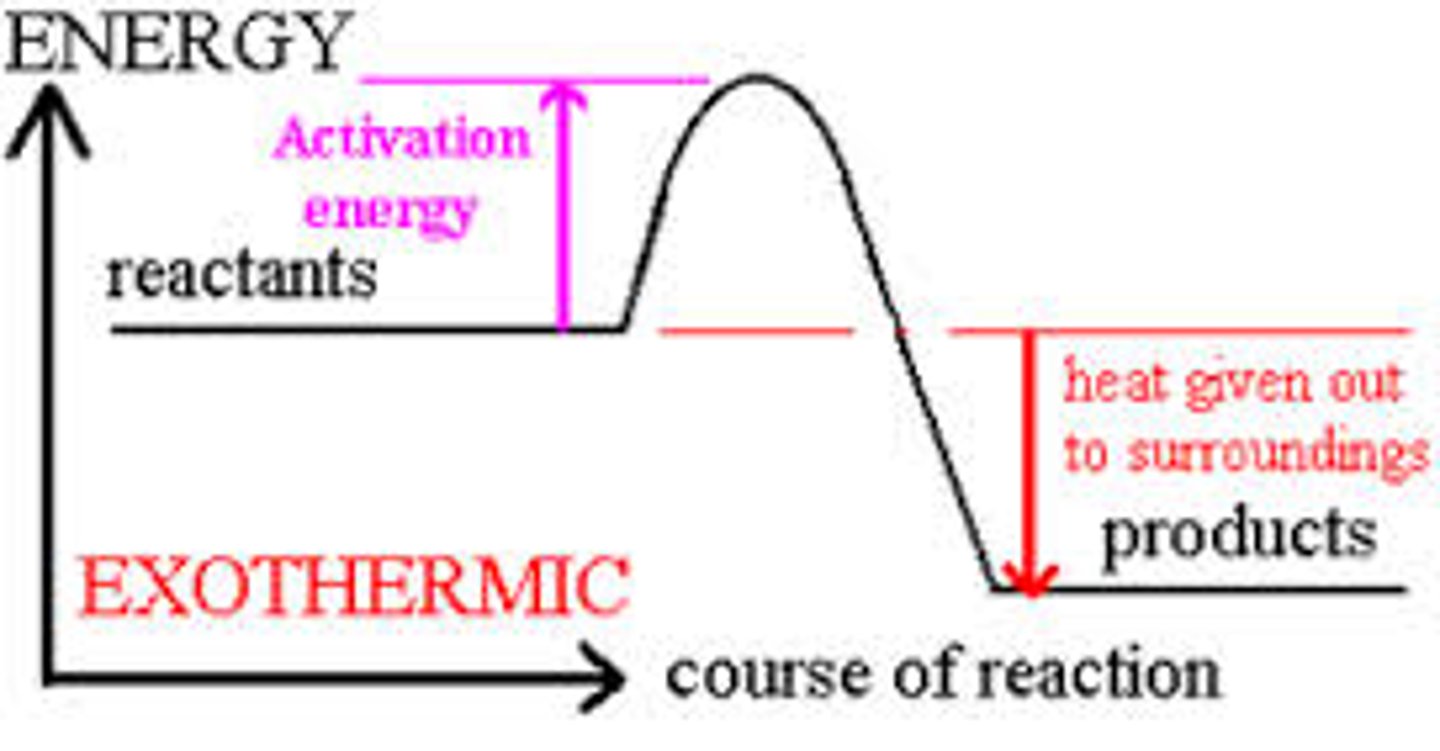
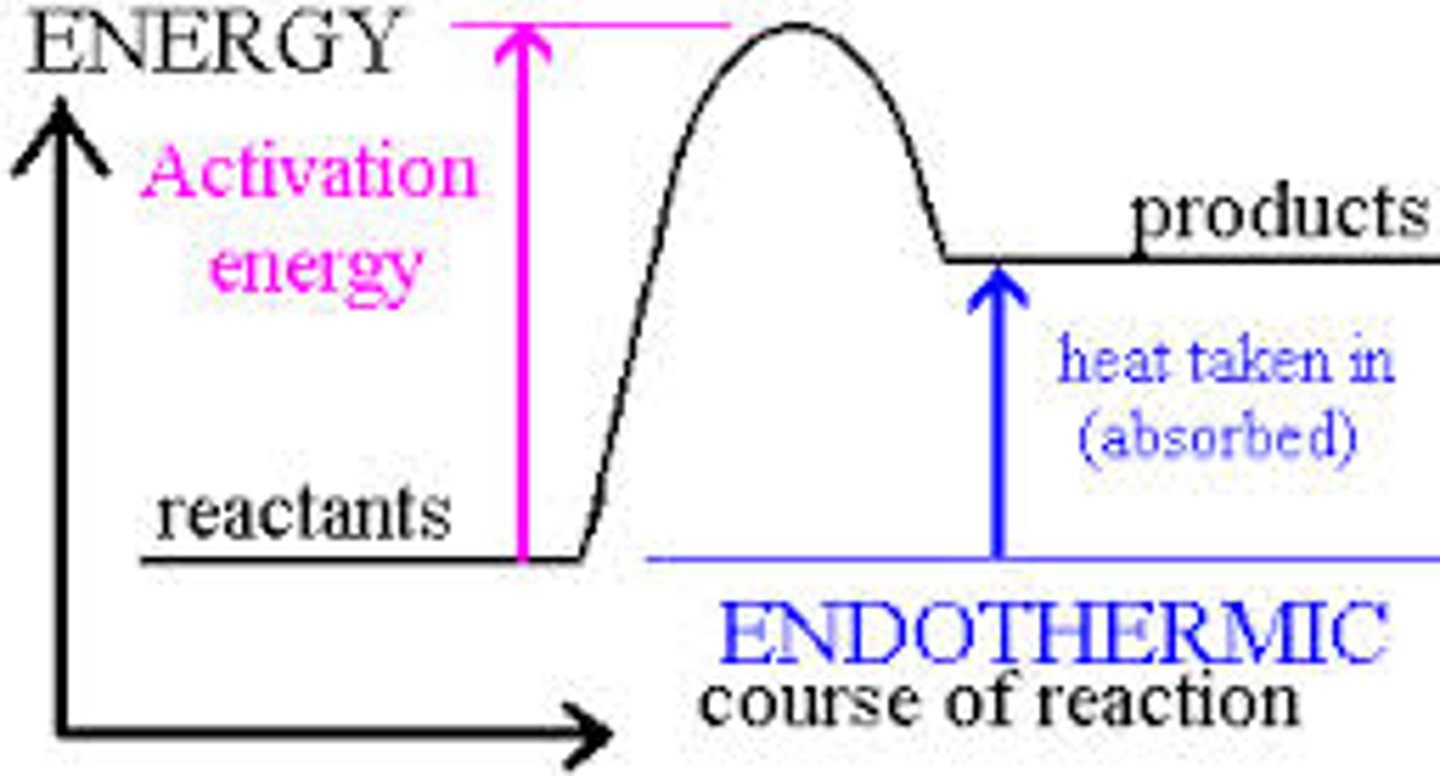
Where is the activation energy located on a energy profile diagram?
The activation energy is the difference in height from the energy of the reactants to the peak of the curve when it is represented on an energy profile diagram.
Interpret a diagram of activation energy on an exothermic energy profile
Interpret a diagram of activation energy on an endothermic energy profile
What are exothermic energy changes shown as?
Exothermic energy changes are shown as negative, because energy has been transferred from the reacting chemicals to the surroundings.
This means the reacting molecules have lost energy.
What are endothermic energy changes shown as?
Endothermic energy changes are shown as positive, because energy is taken in from the surroundings by the reacting molecules.
This means the reacting molecules have gained energy.
What type of reaction is breaking chemical bonds?
Breaking chemical bonds requires energy, so is endothermic.
What type of reaction is making chemical bonds?
Making chemical bonds releases energy, so is exothermic.
Will the overall change in bond energy for an exothermic reaction be positive or negative?
- for an exothermic reaction, the overall change in bond energy would be negative,
- as more energy is released from the reacting molecules into the surroundings in order to form bonds in the products,
- than the amount of energy taken in by the reactants
- so the reacting molecules will have lost energy and the change in bond energy for an exothermic reaction will be negative
Explain why the total bond energy does not just depend on the number of bonds
- the total bond energy does not just depend on the number of bonds
- as different bonds have different bond energies depending on the strength of the bond
This means a small number of strong bonds could have a greater total bond energy than a large number of weak bonds
What information will the exam question give for a question using bond energies to calculate the energy change for a chemical reaction?
The exam question will give us information on the structure of the molecules, so we can see all the different parts.
E.g. H₂ becomes H-H
What is the unit for the energy value of a chemical bond?
The energy value for each chemical bond is measured in kJ/mol
In a body energy calculation, what is happening to the reactants?
Where the reactants are we are breaking the chemical bonds.
Breaking bonds is endothermic, so we add a positive + symbol before the energy value for each bond value of the reactants.
We add the bond energy values for the reactants together, and add in the positive + symbol before the number again.
In a bond energy calculation, what is happening to the product(s)?
On the right hand side of a bond energy calculation, where the product is, we are making chemical bonds.
Making bonds releases energy, so is exothermic.
Exothermic reactions are negative, as the reacting molecules lose energy to they release it to the surroundings,
This means we add a negative - symbol before the bond energy value for the product.
What is the final energy change in a bond energy calculation measured in?
The final energy change is measured in kJ
How do we calculate the final energy change for the bond energy reaction?
To calculate the final energy change for the reaction, we put the bond energy value of the product under the total bond energy value for the reactants.
Then we complete the sum.
E.g. +678
-862
= 184
final energy change = 184 kJ
How can we tell whether the bond energy calculation has been exothermic or endothermic?
If the reaction has a negative symbol, we know it is exothermic.
If the reaction has a positive symbol, we know it is endothermic.
Required Practical 4: Temperature Changes
Which exothermic reaction do we use in this practical?
In the practical investigating temperature change in an exothermic reaction, the exothermic reaction we use is the neutralisation reaction between hydrochloric acid and the alkali sodium hydroxide.
How does the amount of sodium hydroxide solution we add change over the course of the practical investigating temperature change in an exothermic reaction?
We add increasing volumes of sodium hydroxide solution to the hydrochloric acid.
What will we measure in each experiment investigating temperature change in an exothermic reaction?
We will measure the maximum temperature reached in each experiment.
In the practical investigating temperature change in an exothermic reaction, what is the independent variable?
The independent variable is the volume of sodium hydroxide solution.
In the practical investigating temperature change in an exothermic reaction, what is the dependent variable?
The dependent variable is the maximum temperature reached.
In the practical investigating temperature change in an exothermic reaction, what are the control variables?
Control variables:
- volume of hydrochloric acid
- concentrations of both the hydrochloric acid and the sodium hydroxide solution
- the amount of insulation used
Required Practical 4: Temperature Changes
Describe a practical investigating the temperature change in an exothermic reaction
1) Start by using measuring cylinder to measure 30 cm³ of dilute hydrochloric acid
2) Then transfer acid into polystyrene cup
3) Stand polystyrene cup inside beaker to stop cup from falling over
4) Use thermometer to measure temperature of acid and record this in a table
5) Now use measuring cylinder to measure 5 cm³ of sodium hydroxide solution and transfer this to polystyrene cup
6) Next, fit a plastic lid to cup, and place thermometer through hole in the lid, where the bulb of the thermometer is certainly in the solution
7) Use thermometer to gently stir the solution
8) The reaction is exothermic, so releases energy, causing temperature of solution to increase
9) Carefully look at temperature rise on thermometer, and when the reading stops changing, record highest temperature reached
10) Rinse out and dry polystyrene cup
11) Then repeat experiment using 10 cm³ of sodium hydroxide solution and carry out experiment several more times, with 5 cm³ increases in volume of sodium hydroxide
12) Stop adding increasing volumes of 5 cm³ when we reach maximum of 40 cm³ of sodium hydroxide solution
13) Finally, repeat entire experiment once more so we have 2 sets of results to calculate mean value for temperature reached for each volume of sodium hydroxide solution
What volume of hydrochloric acid do we use?
30 cm³ of hydrochloric acid
Why do we stand the polystyrene cup in the beaker?
We stand the polystyrene cup in the beaker to stop it from falling over.
By how much do we increase the volume of sodium hydroxide solution added in each experiment?
We use a 5 cm³ increase in the volume of sodium hydroxide added in each experiment.
When do we stop adding increasing volumes of sodium hydroxide solution to the hydrochloric acid?
We continue adding increasing 5 cm³ volumes of sodium hydroxide solution to the hydrochloric acid until we reach a maximum of 40 cm³ of sodium hydroxide solution.
Why do we repeat the entire experiment again?
We repeat the entire experiment investigating temperature change in an exothermic reaction again, so we have two sets of results.
We use these two sets of results to calculate a mean value for the maximum temperature reached for each volume of sodium hydroxide solution.
How can we represent the results of the practical investigating the temperature change in an exothermic reaction?
We can plot a graph to show our results.
The graph will show that, as we increase the volume of sodium hydroxide solution, the maximum temperature reached increases
Why does the maximum temperature reached increase when we increase the volume of sodium hydroxide solution?
- maximum temperature reached increases as we increase volume of sodium hydroxide solution
- because, as we add more particles of sodium hydroxide, they react with hydrochloric acid
- and as it is an exothermic reaction, more energy is released and maximum temperature reached increases
What will happen for the maximum temperature reached to decrease instead?
- at a certain volume of sodium hydroxide solution, maximum temperature reached starts to decrease
- because we are adding so much sodium hydroxide that there is not enough hydrochloric acid
- this means some of the sodium hydroxide is unable to react, so the amount of energy released by the reaction has reached a maximum
Explain why the maximum temperature decreases when the increasing volumes of sodium hydroxide added cause the hydrochloric acid to be completely used up
- as we repeat the experiment with a greater volume of sodium hydroxide solution each time,
- the energy released is now spread out into a greater volume
- so when we add large volumes of sodium hydroxide solution, the maximum temperature reached actually decreases
In the practical investigating temperature change in an exothermic reaction, why do we use a polystyrene cup with a lid?
The polystyrene cup with a lid is used as we want to measure the temperature by reducing any heat losses.
Polystyrene is a good thermal insulator, so is used to reduce heat loss through the sides and the bottom, as it insulates the reactants as they are mixed.
The lid reduces heat loss to the air.
When the temperature change of a reaction where the temperature increases is measured, why is the value measured usually less than the actual temperature change?
Some energy is always lost to the surroundings as heat.
What are electrolytes?
electrolytes are liquids and solutions that can conduct electricity as their ions are free to move
What happens when two different metals are placed into an electrolyte?
If we place two different metals in an electrolyte, then we can produce electricity.
Name a general example of an electrolyte
A solution of an ionic compound would be an electrolyte, as the ions are now free to move and conduct electricity.
What would happen if we placed strips of two different metals into an electrolyte solution and connected these to a voltmeter?
- If we placed e.g. a strip of copper and a strip of magnesium into an electrolyte solution
- and connected these two metals to a voltmeter,
- we would see a potential difference between them
- also, an electrical current will flow through the wire
If an electrical current flows through the wire and we see a potential difference between the strip of copper and strip of magnesium when they are connected to a voltmeter, what does this tell us?
The electrical current flowing through the wire tells us a chemical reaction is taking place on the surface of the two metals, and these reactions are producing electricity.
Scientists call this set up a cell.
What are cells used for?
Cells can be used to generate electricity.
What property of metals makes them suitable for use as electrodes?
Metals are good conductors electricity, and all parts of the cell must be able to conduct electricity in order for charge to flow.
Interpret a diagram of what the set up of a simple cell could look like
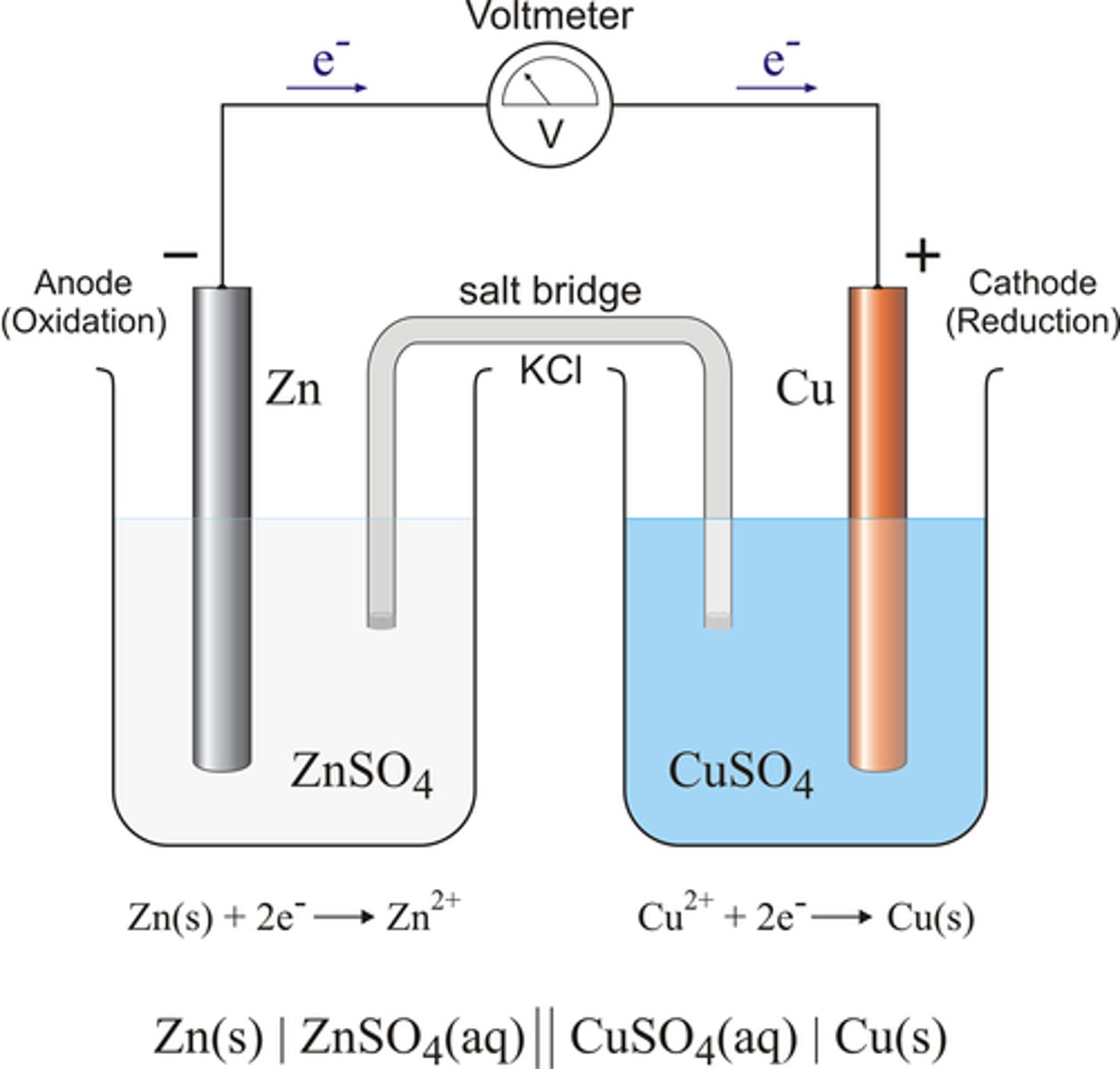
What will the set up of a simple cell include?
Set up of a simple cell:
- strips of two different metals
- an electric current flowing through the wire
- a voltmeter which causes a potential difference between the two strips of metals
- an electrolyte solution
Explain how an electrochemical cell produces electricity
- in an electrochemical cell, chemical reactions between the electrodes and the electrolyte sets up a potential difference between the electrodes
- if the electrodes are connected by a wire, charge flows and electricity is produced
Name two factors that affect the voltage produced by a cell
- temperature
- the type of electrode
What type of ion do alkaline fuel cells contain?
Alkaline fuel cells contain OH⁻ ions
When can cells produce electricity?
A cell can only produce electricity for a certain period of time. Eventually, the chemicals in the cell run out and the reaction stops
What types of metals do cells have to use in order to produce electricity?
Cells only produce electricity if we use metals with different reactivities.
We can work out whether two metals have different reactivities by comparing their places on the reactivity series of metals.
What is a cell?
a cell is a system made up of a single pair of electrodes connected by a wire and in contact with an electrolyte
How can we select the metals for an electrical cell that we want to produce electricity?
We can use the reactivity series of metals to select the metals for an electrical cell.
This cell will only produce electricity if two metals with different reactivities are selected.
What does the size of the potential difference between the two metals depend on?
The size of the potential difference between the two metals depends on the difference in the reactivity between the two metals.
What will the potential difference be like for a cell containing two metals with very different reactivities?
A cell containing two metals with very different reactivities will have a large potential difference.
E.g. a cell containing magnesium and copper.
What will the potential difference be like for a cell containing two metals with similar reactivities?
A cell containing two metals with similar reactivities will have a smaller potential difference, as there is a smaller difference in reactivity between the two metals.
E.g. a cell containing zinc and tin.
The greater the difference between the reactivity of the metals, the ....
The greater the difference between the reactivity of the metals, the greater the potential difference produced by the cell.
What else does the potential difference depend on?
The electrolyte also affects the potential difference.
What is a battery?
a battery contains two or more cells connected in series to produce a greater voltage
How does the potential difference of cells connected in series together relate to the potential difference of the battery these two cells form when connected?
E.g. if we had two cells in series, each with a potential difference of 2.5 V, and we connected them in series together, this would mean the battery would have a potential difference of 5 V.
What is the difference between a cell and a battery?
A cell is made up of a single pair of electrodes connected by a wire and in contact with an electrolyte, while a battery contains two or more cells connected in series.
Are alkaline batteries rechargeable?
Alkaline batteries are non-rechargeable.
Explain why alkaline batteries are non-rechargeable
- in alkaline batteries, the reactants are used up at some point and no more electricity/charge is produced
- this is because alkaline batteries are not rechargeable
- the chemical reactions are irreversible, so alkaline batteries are non-rechargeable batteries
Describe why rechargeable batteries can be recharged
- rechargeable batteries can be recharged
- because we can reverse the chemical reactions when we apply an electrical current
What is a fuel cell?
a fuel cell is an electrochemical cell in which a fuel donates electrons at one electrode and oxygen gains electrons at the other electrode
What are the reactants in a fuel cell?
In a fuel cell, we react fuel such as hydrogen with either oxygen or air.
What is the fuel in a hydrogen fuel cell?
Hydrogen is the fuel in a hydrogen fuel cell.
What happens inside the fuel cell?
Inside the fuel cell, a chemical reaction takes place, producing an electric current.
Describe how fuel cells work
fuel cells work by reacting a fuel with oxygen to produce energy
What waste product is produced for a hydrogen fuel cell?
For the hydrogen fuel cell, the only waste product is water.
Interpret a simplified diagram of a hydrogen fuel cell
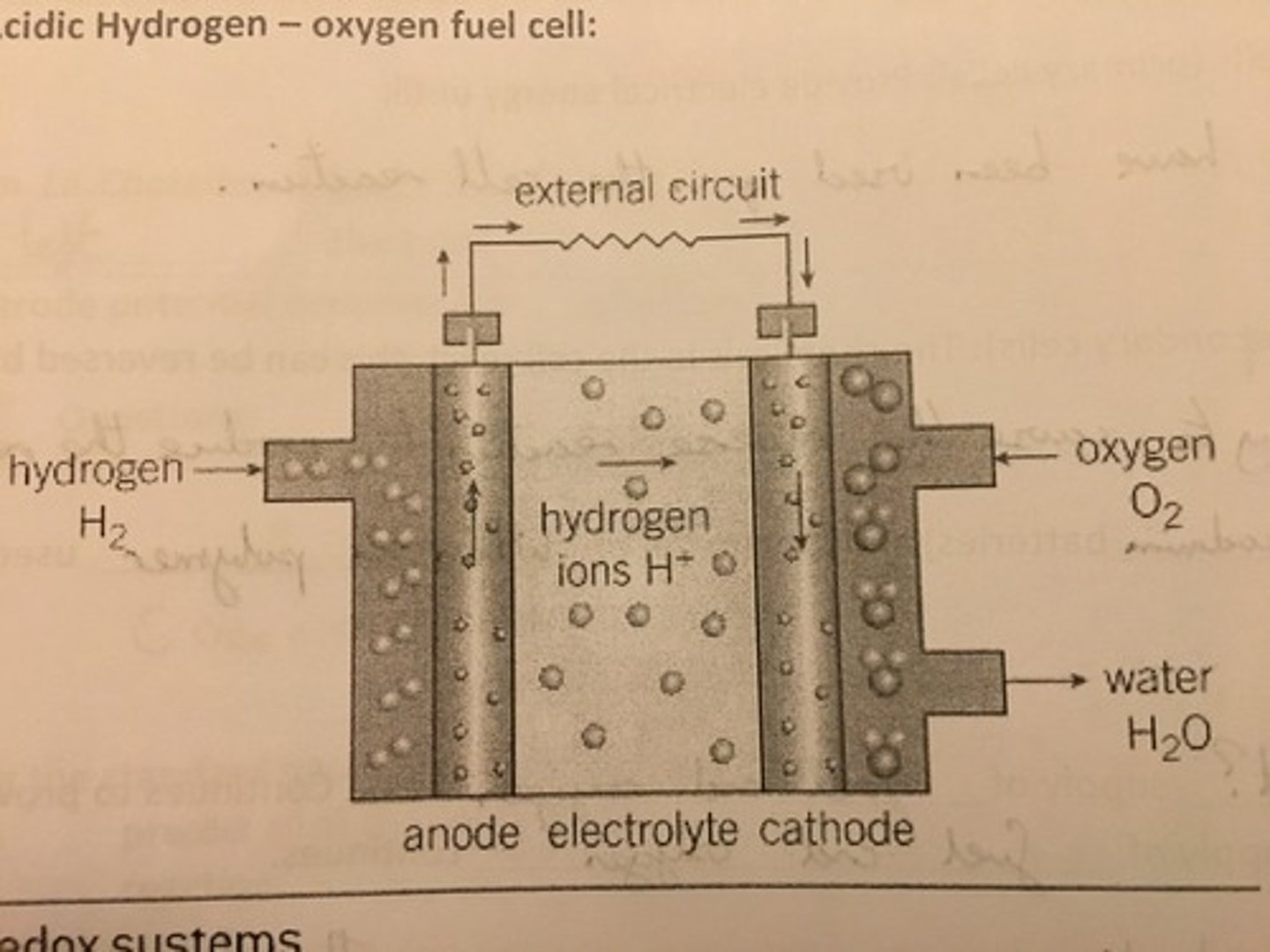
What happens at the negative electrode in a hydrogen fuel cell?
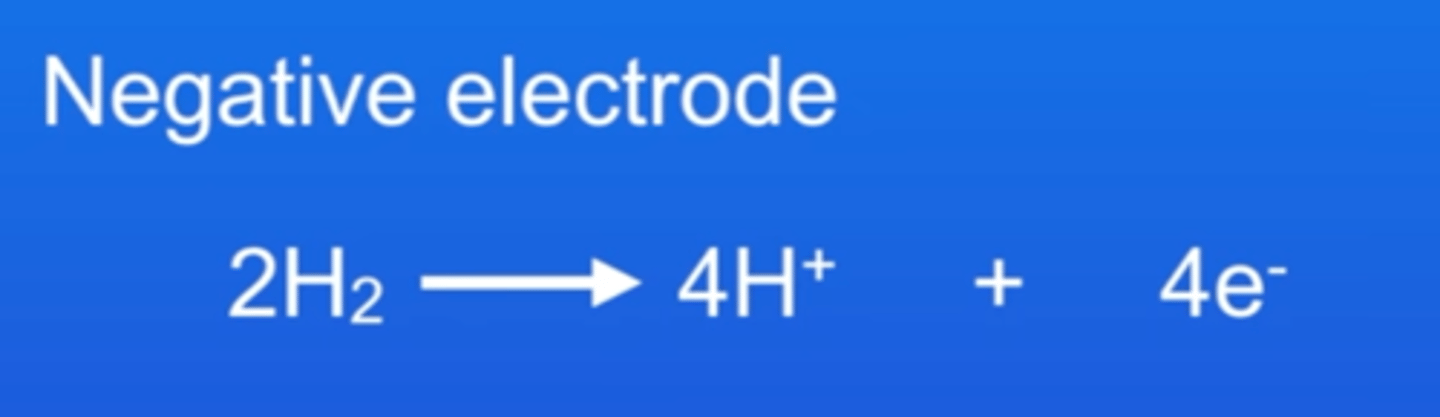
At the negative electrode in a hydrogen fuel cell, hydrogen is produced as hydrogen ions combine with electrons, which pass through the wire as the electrical current.
State the half equation at the negative electrode of a hydrogen fuel cell
What happens at the positive electrode in a hydrogen fuel cell?
At the positive electrode, oxygen molecules combine with the electrons and hydrogen ions produced at the negative electrode to produce water.
State the half equation at the positive electrode in a hydrogen fuel cell

What is essentially happening between hydrogen and oxygen in a hydrogen fuel cell?
Essentially, in the hydrogen fuel cell, hydrogen is combining with oxygen, so the hydrogen is being oxidised.
What process takes place to create a potential difference in the fuel cell?
The fuel is oxidised, so this means it gives up electrons /loses electrons, and this creates a potential difference in the cell.
What is oxidation in terms of electrons?
Oxidation in terms of electrons is a loss of electrons
State the overall equation of a hydrogen fuel cell
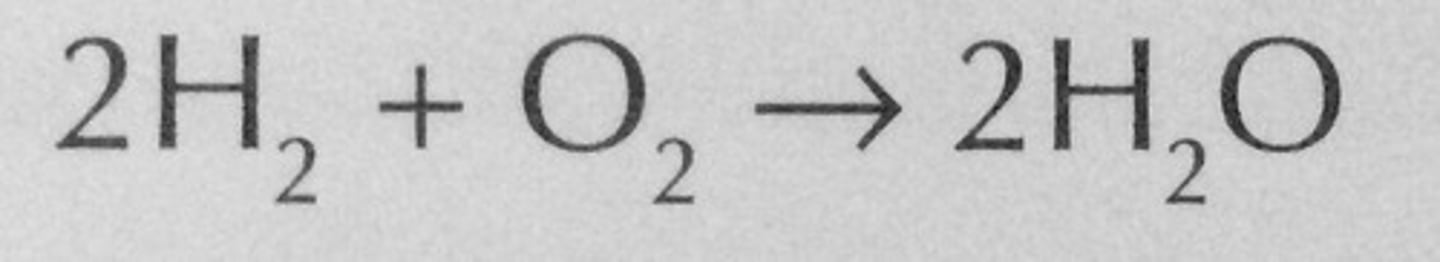
In the equation for a hydrogen fuel cell, what is happening to the hydrogen?
In the equation for a hydrogen fuel cell, the hydrogen is being oxidised.
This means the hydrogen is gaining oxygen when it combines with oxygen to produce water.
Describe what happens overall in a hydrogen fuel cell
- so in a hydrogen fuel cell, two simple gases, hydrogen and oxygen, combine to produce electricity
- the only waste product of this is water