Alcohol Reactants, Epoxide Reactants, Ether Reactants
0.0(0)
Studied by 22 peopleCard Sorting
1/48
Earn XP
Description and Tags
Last updated 11:54 PM on 9/27/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
1
New cards
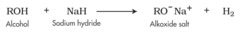
alcohol + metal hydride
forms an alkoxide
2
New cards
alcohol + active metal
forms an alkoxide

3
New cards
alcohol + H[halogen]
substitution reaction that forms an R-[halogen] and water
![substitution reaction that forms an R-[halogen] and water](https://knowt-user-attachments.s3.amazonaws.com/0fa415d98032417aad21f4a0c88ca294.png)
4
New cards
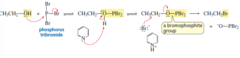
alcohol + PBr3, pyridine
substitution reaction that forms an alkane bromide by replacing -OH with -Br
CH3OH (1', 2' Alcohol only) as well as POBR2 and other salts
CH3OH (1', 2' Alcohol only) as well as POBR2 and other salts
5
New cards
alcohol + SOCL2
substitution reaction that generates an alkyl chloride

6
New cards
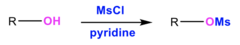
alcohol + MsCl, pyridine
substitution reaction that converts R-OH to a sulfonate; forms an alkyl nucleophile and OMs-
7
New cards
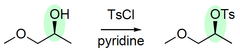
alcohol + TsCl, pyridine
substitution reaction that converts R-OH to a sulfonate; forms an alkyl nucleophile and OTs-
8
New cards
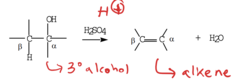
alcohol + strong acid, heat
dehydrating elimination reaction that generates an alkene
9
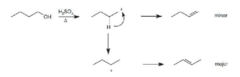
New cards
alcohol + H2SO4, heat
dehydrating elimination reaction that generates an alkene
10
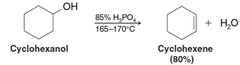
New cards
alcohol + H3PO4, heat
dehydrating elimination reaction that generates an alkene
11
New cards
alcohol + CrO3, H2SO4, Δ
Jones Oxidation reaction that results in a C=O in the form of an aldehyde or, if H2O is present, a carboxylic acid (1°) or in the form of a ketone (2°)
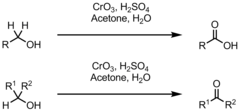
12
New cards
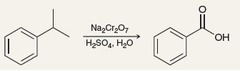
alcohol + Na2Cr2O7, H2SO4, Δ
Jones Oxidation reaction that results in a C=O in the form of an aldehyde or, if H2O is present, a carboxylic acid (1°) or in the form of a ketone (2°)
13
New cards
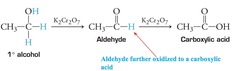
alcohol + K2Cr2O7, H2SO4, Δ
Jones Oxidation reaction that results in a C=O in the form of an aldehyde or, if H2O is present, a carboxylic acid (1°) or in the form of a ketone (2°)
14
New cards
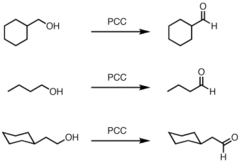
alcohol + PCC
oxidation reaction that turns 1° alcohols into aldehydes and 2° into ketones
15
New cards
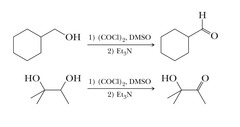
alcohol + (1) (COCL)2, DMSO (2) Et3N
Swern Oxidation reaction that turns 1° alcohols into aldehydes and 2° into ketones
16
New cards
alcohol + (1) DMP, (2) Et3N
DMP Oxidation reaction that that turns 1° alcohols into aldehydes and 2° into ketones
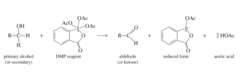
17
New cards
alcohol + HIO4 (periodic acid)
Periodic Acid oxidation reaction that splits C-C bond and replaces it with a O=C bond by splitting the reactant
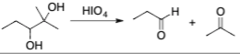
18
New cards
structure of PCC
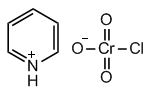
19
New cards
structure of mesyl chloride (CH3SO2Cl)
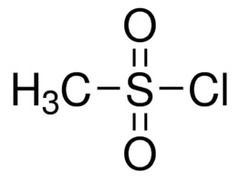
20
New cards
structure of DMP
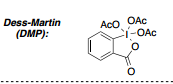
21
New cards
structure of oxalyl chloride/(COCL)2
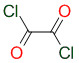
22
New cards
structure of Et3N
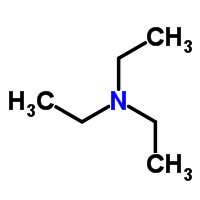
23
New cards
structure of pyridine

24
New cards
structure of a metal hydride
compounds of one or more metal cations (M+) and one or more hydride anions (H−)
25
New cards
structure of halohydrin
a halogen and a hydroxyl are bonded to adjacent carbon atoms
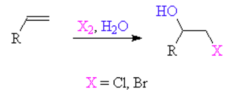
26
New cards
structure of PBR3
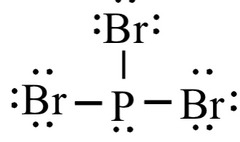
27
New cards
structure of H2SO4

28
New cards
structure of H3PO4
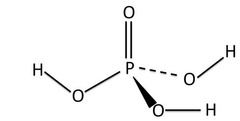
29
New cards
alkene + mCPBA
syn epoxide synthesis stereospecific rxn that generates an epoxide and a carboxylic acid
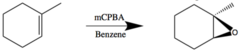
30
New cards
alkene + [R]CO3H
epoxide synthesis stereospecific rxn that generates a racemic mix of enatiomers
31
New cards
alkane + halohydrin
forms an alkoxide used to generate epoxide
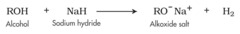
32
New cards
structure of an alkoxide
an organic group bonded to a negatively charged oxygen atom, written as RO− and often ionically bonded to a positively charged ion (like Na+)
33
New cards
structure of an epoxide
organic three-membered cyclic oxygen compounds
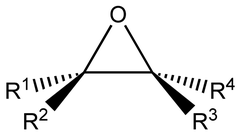
34
New cards
structure of a halohydrin
a functional group in which a halogen and a hydroxyl are bonded to adjacent carbon atoms
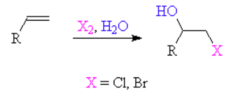
35
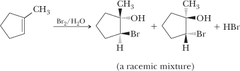
New cards
alkene + halogen, H2O
forms a halohydrin (with an OH group) to generate an alkoxide which helps generate an epoxide
36
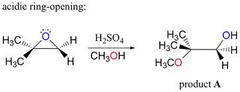
New cards
epoxide + strong acid (H+), H2O
acid catalyzed ring opening rxn in which the acid attacks the more substituted carbon
37
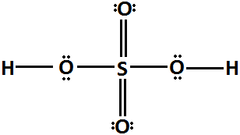
New cards
structure of H2SO4
very strong acid
38
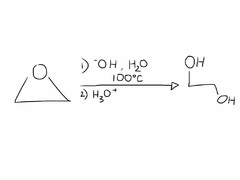
New cards
epoxide + strong base (OH-), H2O
base catalyzed ring opening rxn in which the strong nucleophile attacks the less substituted carbon
39
New cards
epoxide + alkoxide, alcohol
ring opening rxn that acts as a fully SN2 rxn
40
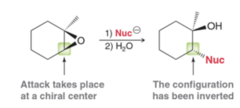
New cards
epoxide + H+, alcohol
ring opening rxn that acts as an "SN1-like" SN2 rxn
41
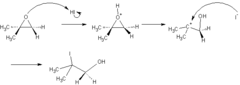
New cards
epoxide + strong nucleophile (no acid)
ring opening rxn that generates an alkyl with an alcohol group from the nucleophile attacking the less substituted carbon
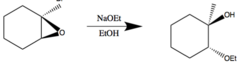
42
New cards
alcohol + H+, Δ, low heat
ether synthesis reaction that works with symmetrical ethers by adding the R groups on either side of the O
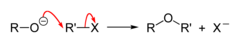
43
New cards
alkyl halide + alkoxide
Williamson Ether Synthesis that results in asymmetrical ethers

44
New cards
structure of an alkoxide
consists of an organic group bonded to a negatively charged oxygen atom and tends to form ionic bonds with positive ions, like Na+

45
New cards
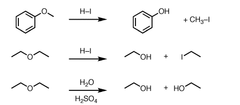
ether + HI or HBr (1 eq.)
ether cleavage resulting in an alkyl halide and an alcohol; uses SN1 unless both Cs are 1°
46
New cards
structure of an ether
R-O-R
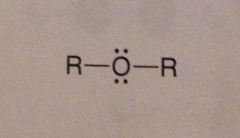
47
New cards
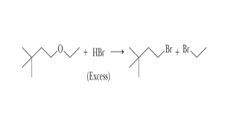
ether + HI or HBr (2+ eq.)
ether cleavage resulting in an alkyl iodide or alkyl bromide and alcohol; uses SN1 unless both Cs are 1°
48
New cards
structure of an alkyl halide
alkyl group bonded to a halogen
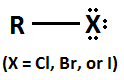
49
New cards
alkene + acid (H+), R-OH
acid catalyzed addition of alkenes to alcohols and ethers
