AROMATIC HYDROCARBONS
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
What is an aliphatic organic chemical?
straight or branched chain organic substances
What is an aromatic organic chemical?
includes one or more ring of 6 carbon atoms with delocalised bonding
What is benzenes structure?
C6H6
6 carbon atoms in a hexagonal ring with 1 hydrogen atom bonded to each carbon atom
each carbon atom is bonded to 2 other carbon atoms and 1 hydrogen atom by sigma bonds, leaving a delocalised electron in the p orbital
What is the structure and bonding found in benzene?
planar molecule
C-C bonds are all the same length and have bond energy between a C-C single and C=C double bond
bond angle is 120
What is the enthalpy of hydrogenation of benzene?
theoretically as there are 3 double bonds the enthalpy of hydrogenation should be -360
actually benzenes is less exothermic
benzene is more stable due to the delocalised electrons, leading to the delocalisation energy
Why is cyclohexa-1,3-diene more stable than cyclohexa-1,4-diene?
as the pi electrons overlap
What type of reactions does benzene usually undergo?
not normally addition reactions as these would involve breaking the delocalised system
electrophilic substitution is usually preferred
What’re the issues with benzene?
benzene is a carcinogen and is banned in schools
Why is methylbenzene used instead of benzene?
less toxic
reacts more readily than benzene as the methyl side group releases electrons into the delocalised system making it more attractive to electrophiles
What occurs in the nitration of benzene?
forms nitrobenzene
reagents - concentrated nitric acid in the presence of concentrated sulfuric acid (catalyst)
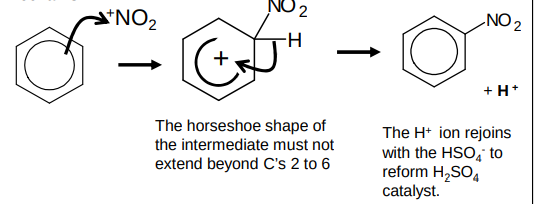
mechanism - electrophilic substitution
Why is the nitration of benzene important?
it synthesises useful compounds e.g. TNT and is used in the formation of amines
What is the overall equation for the nitration of benzene?
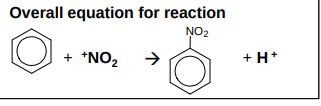
How is the NO2+ electrophile created?
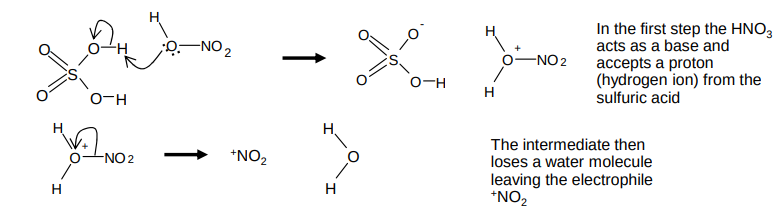
in an acid base reaction
HNO3 + 2H2SO4 → +NO2 + 2HSO4- + H3O+
What is the mechanism for the formation of the electrophile +NO2?
What is the mechanism for the nitration of benzene?
What is friedel crafts acylation?
forms a phenyl ketone
reagents - acyl chloride in the presence of anhydrous aluminium chloride catalyst
conditions - heat under reflux
mechanism - electrophilic substitution
What is the equation for the formation of the electrophile?
AlCl3 + CH3COCL → [CH3CO]+ + [AlCl4]-
What is the mechanism for friedel crafts acylation?
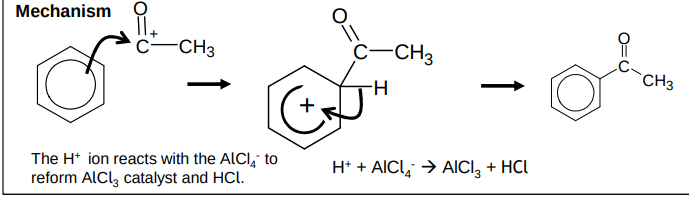
How does the AlCl4- act as a catalyst?
H+ ion reacts with it to reform AlCl3 catalyst and HCl
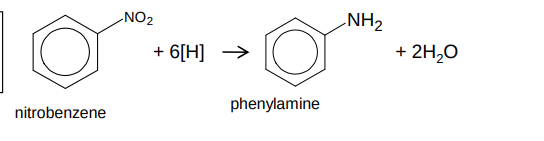
How do you reduce a nitroarene into aromatic amines?
reagent - Sn and HCl or Fe and HCl
conditions - heating
mechanism - reduction
or via catalytic hydrogenation
What occurs in the reduction of nitroarene into an aromatic amine?
What is the effect of having a chlorine atom attached to the benzene ring?
makes it stronger
halogenoalkane substitution and elimination reactions do not occur
electron rich benzene will repel nucleophiles
What is the effect of a OH group attached to the benzene ring?
delocalisation makes the C-O bond stronger and the O-H bond weaker
phenol does not act as an alcohol, it is more acidic and doesn’t oxidise
What is the effect of having a NH2 group attached to the benzene ring?
less basic than aliphatic amines as lone pair is delocalised
less available for accepting a proton