15. Heavy Metals II
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
How does mercury (Hg) exemplify environmental movement of heavy metals?
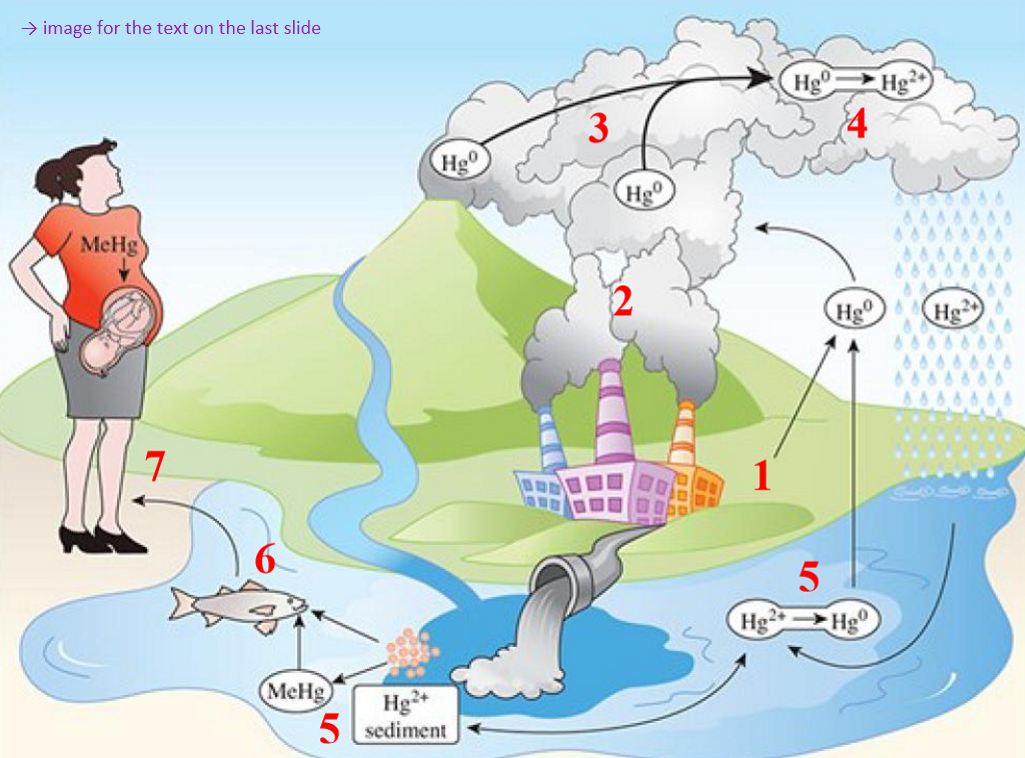
1) atmospheric Hg comes from natural degassing of crust, volcanic eruptions, & evaporation from oceans & soils
2) human sources (anthropogenic) → 2/3 total atmospheric Hg → coal combustion, smelting, etc.
3) Hg vapour is monatomic gas, retained 1 yr in atmosphere (time to distribute around the world)
4) oxidizes to Hg2+ → returns to earth dissolved in water as rain
5) either reduce & vaporized or incorporated into organic compounds by microorganisms producing methylmercury (MeHg)
6) MeHg enters trophic levels starting w plankton, herbivorous fish, carnivorous fish, sea mammals
7) bioamplification up higher trophic levels (1800-80000x the concentration in surrounding water
Toxicokinetics of Hg Absorption

Toxicokinetics of Hg Distribution
into kidneys as cysteine S-conjugate or from the basolateral membrane through OATs & excreted in urine & feces
bind to thiol in cysteines & mimics methionine for transport into the brain & placenta via neutral aa carriers (e.g. LAT1)
binds to sulfur-rich keratin-containing proteins & accumulates in hair → indicator of exposure

Toxicokinetics of Hg Elimination
conjugation to glutathione → excretion via MRP2 by kidney & liver
Dose-Dependent Methylmercury Toxicity