Literally everything from all of gen chem
1/377
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
378 Terms
Solute
The substance that is dissolved in a solution (e.g., salt in saltwater).
Solvent
The substance that dissolves the solute (e.g., water in saltwater).
Solutions
one compound (solute) is dissolved in another (solvent)
aqueous solution
when the solvent is water
electrolytes
substance which dissolves in water to give an electrically conducting solution
solubility
the amount of a substance that dissolves in a given quantity of solvent
saturated solution
solvent holds as much solute as is possible at that temperature
unsaturated solution
less than the maximum amount of solute for that temperature is dissolved in the solvent
supersaturated solution
solvent holds more solute than is normally possible at that temperature
miscible
when two liquids create a homogenous mixture
immiscible
when two liquids create a heterogeneous mixture
the 4 conditions that determine solubility/miscibility
intermolecular forces, entropy change, temperature, pressure
Solvation
the process where solute particles (ions or molecules) become surrounded and stabilized by solvent molecules
What does each variable mean in Henry’s Law: Sg = kPg
Sg: the solubility of the gas; k: Henry’s law constant for that gas in that solvent; Pg: The partial pressure of the gas above the liquid
percent mass
Mass % of A = (mass of A in solution/ total mass of solution) x 100
mol fraction
XA = moles of A/ total moles in solution
molarity
M = mol of solute/ L of solution
molality
m = mol of solute/ kg of solvent
Units to express solubility
g/L, mol/L, mol/kg
factors affecting solubility:
temperature, pressure, Henry’s law, molecular size and shape, polarity
grams to moles
moles= mass (grams)/ molar mass (g/mol)
What does each variable mean in Raoult’s law: Psolvent= Xsolvent x P°solvent
Where, Xsolvent is the mole fraction of the solvent and P0solvent is the normal vapor pressure of pure solvent at that temperature.
Vapor pressure of a solution is ? than that of the pure solvent.
LOWER
Positive and negative deviations, what intermolecular forces leads to each?
For a positive deviation, the vapor pressure is greater than expected.
For a negative deviation, the vapor pressure is lower than would be expected
Define colligative properties
Properties of solutions that depend only on the NUMBER of solute particles present, not on the identity of the solute particles.
List the 4 colligative properties
vapor pressure lowering, boiling point elevation, melting point depression, osmotic pressure
Give formulas for ΔTb and ΔTf
ΔTb =__
ΔTf =__
What concentration units are used in the above formulas?
DTb = Kb x m; DTf = Kf x m; C, molal, and C/mol
Are the ΔT values to be added or subtracted?
Boiling point: ADDED
Freezing point: SUBTRACTED
Which of the following solutions would not be expected to exist?
NaCl in water
MgCl2 in CCl4
Methanol, CH3OH, in water
Pentane, CsH12, in CCl4
Methanol, CH3OH, in water
Does molarity or molality change with temperature?
molarity
Solubility of a gas increases as ? and ? increases, and decreases as ? is increased
pressure and size, temperature
Physical properties that depend on the ? but not the ? of solute particles in a given amount of solvent are called colligative properties.
number ; kind
Which of these aqueous solutions would be expected to have the lowest freezing point energy?
0.100 m KCl
0.200 m CH3OH
0.100 m NaOH
0.075 m K2SO4
0.075 m K2SO4
The Tyndall effect describes ?
the scattering of light by colloidal particles
Which one of the following statements about soaps and soap molecules is false?
They are often sodium salts of long chain fatty acids.
The hydrophilic end of a soap molecule is attracted by grease.
They have a hydrophobic end.
They have a polar end.
The hydrophilic end of a soap molecule is attracted by grease.
Which statement is incorrect?
The Law of Conservation of Energy is another statement of the First Law of Thermodynamics.
The energy of the universe is constantly increasing.
Energy is the capacity to do work or to transfer heat.
Potential energy is the energy that a system possesses by virtue of its position or composition.
The energy of the universe is constantly increasing.
Which term is not correctly matched?
universe / system plus surroundings
exothermic / energy is released
endothermic / energy is absorbed
state function / property dependent on how the process takes place
state function / property dependent on how the process takes place
Which statement concerning sign conventions for DE = q + w is false?
For heat absorbed by the system, q is positive.
For work done by the system, w is negative.
When energy is released by the reacting system, DE is negative.
If DE is positive, products are at a lower energy than reactants.
If DE is positive, products are at a lower energy than reactants.
Lattice Energy
how much energy you need to break a bond
hydration energy
Energy released when water molecules surround ions, like salt, as they dissolve
State Raoult’s law and the equation: P(solution) = ________*_________
The vapor pressure of a solvent in an ideal solution is directly proportional to its mole fraction in the solution. Xsolvent x P0solvent
vapor pressure ? as temperature increases, and ? as the strength of the intermolecular forces increases.
increases; decreases
as vapor pressure increases, boiling point ?
decreases
For an ideal mixture of 2 liquids, the total vapor pressure in terms of the mol fractions and partial pressures is expressed as
XA x P0A + XB x P0B
A positive deviation from Raoult’s law implies that
o the vapor pressure of the solution is ? than predicted by ideal
behavior
o the boiling point of the solution is ? than predicted by ideal behavior
o the intermolecular forces in the solution is ? than that of the
individual solvents.
greater; lower; weaker
A negative deviation from Raoult’s law implies that
o the vapor pressure of the solution is ? than predicted by ideal
behavior
o the boiling point of the solution is ? than predicted by ideal behavior
o the intermolecular forces in the solution is ? than that of the
individual solvents.
lower; higher; stronger
define reaction rate
Change in concentration of a reactant or
product per unit time
Name four factors which affect the rate of a chemical reaction
temperature, surface area, concentration, catalysts
For the chemical reaction F → G, the initial concentration of F is 2.7 M and its concentration after 2.0 hours is 1.3 M. What is the average rate of this reaction?
-0.7 M/hr
What happens to the rate of the reaction as
the reaction progresses and why?
The rate decreases because as time progresses, the more the two reactants become one.
define rate law
An equation that expresses reaction rate as a function of reactant
concentrations
define rate constant (k)
A proportionality constant in the rate law.
units of first order
s⁻¹
units of second order
M⁻¹ s⁻¹
units of third order
M⁻² s⁻¹
The reduction of bromate ions, BrO3−, by bromide ions in acidic solution has the rate law:
Rate = k[BrO3−][Br−][H+]2 What are the orders with respect to the reactants and the overall order?
first, first, second, and fourth
The decomposition of ozone follows the equation: 2O3(g) → 3O2 (g)
The following rates were measured at 71C:
Initial rate, M/s [O3], M
1.55 x 10-8 ,1.25 x 10-4
3.06 x 10-8 ,2.50 x 10-4
6.15 x 10-8 ,5.00 x 10-4
a. Express the rate of change of [reactant] and [product] in the correct relationship to each other.
b. Write the rate law for the reaction.
c. Calculate the initial rate of the reaction when [O3] is 7.50 x 10-4 mol/L.
d. what does k=
a) rate = -1/2 x d(O3)/dt = 1/3 d(O2)/dt
b) rate=k(O3)^1
c) 9.30 × 10^-8 M/s
d) 1.24 × 10^-4 M-1 s-1
The following rate data, obtained at 250 K, describe the reaction: F2 (g) + 2 ClO2 (g) → 2 FClO2 (g)
Initial rate, M/s [F2] [ClO2]
a. 1.2 x 10-3 0.10 0.010
b. 1.9 x 10-2 0.10 0.040
c. 2.4 x 10-3 0.20 0.010
d. ? 0.30 0.020
Determine the rate law and rate constant for the reaction. Determine the initial rate expected for (d).
rate law= k[F2][ClO2]^2; k=120M^-2 s^-1; 0.0144M/s
Examine the following reaction and rate data: 2 SO2 + O2 → 2 SO3
Initial rate, M/s [SO2] [O2]
1.12 x 10-7 0.03 0.04
2.25 x 10-7 0.015 0.16
9.01 x 10-7 0.06 0.16
Determine the rate law and rate constant for the reaction.
rate law=k[SO2][O2]; k=9.33 × 10^-5 M-1s-1
NO2 + CO → NO + CO2
Initial rate, M/s [NO2] , M [CO] , M
4.0 x 10-6 2.0 x 10-3 3.0 x 10-3
1.6 x 10-5 4.0 x 10-3 3.0 x 10-3
1.0 x 10-6 1.0 x 10-3 2.0 x 10-3
What is the rate law? What is the rate constant?
rate law=k[NO2]^2; k=1 M^-2 s^-1
One of the reactions that is used to produce gaseous hydrogen commercially follows. A proper expression for the rate of this reaction could be ____.
H2O(g) + CO(g) → H2(g) + CO2(g)
-d(H2O)/dt
In the following reaction, d(B)/dt = -0.89 mol/L x s . What is the value of the rate of the reaction at this time? A + 3B → AB3
0.30 mol/L x s
A reaction A + 2B ® C is found to be first order in A and first order in B. What are the units of the rate constant, k, if the rate is expressed in units of moles per liter per minute?
M^-1/min^-1
Consider the following rate data for the reaction below at a particular temperature.
2A + 3B ® Products
Experiment | Initial [A] | Initial [B] | Initial Rate of Loss of A |
1 | 0.10 M | 0.30 M | 7.20 ´ 10-5 M·s-1 |
2 | 0.10 M | 0.60 M | 1.44 ´ 10-4 M·s-1 |
3 | 0.20 M | 0.90 M | 8.64 ´ 10-4 M·s-1 |
The reaction is ____ order in A and ____ order in B.
second; first
A second order reaction will be (faster/slower) than a fifth order reaction
faster
Determine the overall rate order
rate = k[H2SeO3][H+]2[I-]3
6
units of zero order
mol L^-1 s^-1
reaction order when n=1 in rate=(a)1
first order
reaction order when n=1 in rate=(a)2
second order
Give the mathematical expression for the concentration-time relationships for rate laws obeying 1st order and 2nd order kinetics. (rate law & integrated rate form)
1st: rate=k(A); ln(A)=ln(A)0-kt. 2nd: rate=k(A)^2; 1/A=1/(A)0 + kt
The integrated form of the first order rate law in a (positive/negative) slope
negative
The integrated form of the second order rate law in a (positive/negative) slope
positive
Which of the following straight line plots correspond to a first order reaction?
A. (A) vs. t with a slope of -k
B. ln(A) vs t with a slope of +k
C. 1/(A) vs t with a slope of +k
D. ln (A) vs t with a slope of -k
E. (A) vs t with a slope of +k
d
The following data were obtained for the gas-phase decomposition of nitrogen dioxide at 300C, 2 NO2 → 2 NO + O2
Time (s) [NO2,], M
0.0 0.0100
50 0.0079
100 0.0065
200 0.0048
300 0.0038
Is the reaction first or second order in NO2? (Use graphical methods)
second
Butadiene reacts to form its dimer according to the equation: 2C4H6 (g) → C8H12 (g)
The following data were collected for this reaction at a given temperature.
[C4H6],M Time, s
0.01000 0
0.00625 1000
0.00476 1800
0.00370 2800
0.00313 3600
0.00270 4400
0.00241 5200
0.00208 6200
Is the reaction first or second order in C4H6 ? Write a rate law for the reaction.
second order; rate=k[C4H6]²
Dinitrogen pentoxide, N2O5, decomposes by 1st order kinetics with a rate constant of 0.15 s-1 at 353 K.
a) What is the half-life of this decay at 353 K ?
b) If [N2O5]o is 2.33 x10-2 M, what will the [N2O5] concentration be after 2.0 s ?
c) How much time will elapse before the concentration of [N2O5] decreases from that in part (b) to 1.56 x10-3 M?
4.62 s; 0.01726 M; 16 sec
An oil painting, supposed to be the work of Rembrandt (1606-1669 AD), is checked by 14C dating. In 1997, the 14C content (t1/2 = 5720 yr) of the canvas was measured to be 0.961 (A/Ao) times that in a living plant.
How long ago was the painting created, could the painting have been by Rembrandt?
328.28 years ago; yes
The radioactive isotope 32P decays by first-order kinetics and has a half-life of 14.3 days. How long does it take for 95.0% of a sample of 32P to decay?
61.82 days
The gas phase reaction A + B → C has a reaction rate which is experimentally observed to follow the relationship rate = k[A]2[B]. If the concentration of A is tripled and the concentration of B is doubled, the reaction rate would be increased by a factor of ____.
18
A plot of ln[D] vs. time is linear for the reaction D→E. What is the kinetic order of the reaction?
first
The rate constant for the second order reaction below is 2.79 L/mol ·min at 48°C. If the initial concentration of NO2 is 1.05 M , what is the half-life?
2NO2 → N2O4
[t(1/2) = 0.693/ak for first order reaction; t(1/2) = 1/ak[A0} for second order, where a = coefficient of A in the balanced chemical reaction]
10.3 s
Which of the following statements concerning graphical methods for determining reaction order is false?
A) For a first-order reaction the slope of the straight-line graph equals -k.
B) For a first-order reaction the plot of ln[A] vs. time gives a straight line.
C) For a second-order reaction the plot of [A]2 vs. time gives a straight line.
D) For a second-order reaction the intercept of the straight-line graph equals 1/[A]0.
C
The reaction below has the following rate law: rate = 0.011 L/mol·s [SF4]2.
SF4 ← SF2 + F2
How many minutes will it take for the concentration of SF4 to be reduced from 2.5 M to 0.25 M?
5.5 minutes
Discuss reaction rate in terms of the Collision Theory. What three criteria must be met for a reaction to occur between molecules?
collision, energy, orientation
Discuss reaction rate in terms of Transition State Theory.
Transition State Theory describes the reaction process as passing through a high-energy
transition state or activated complex. Reactants must absorb enough energy to reach this
transition state before proceeding to products.
The heat of formation of HCl(g) is exothermic by -90 kJ/mol and the activation energy for the reaction is 150 kJ/mol. Calculate the activation energy for the reaction: HCl(g) → 1/2 H2(g) + 1/2 Cl2(g)
Illustrate this result on an energy vs. reaction coordinate diagram.
The reverse activation energy = 240 kJ/mol and the graph would be exothermic because there is a release of 90kJ/mol
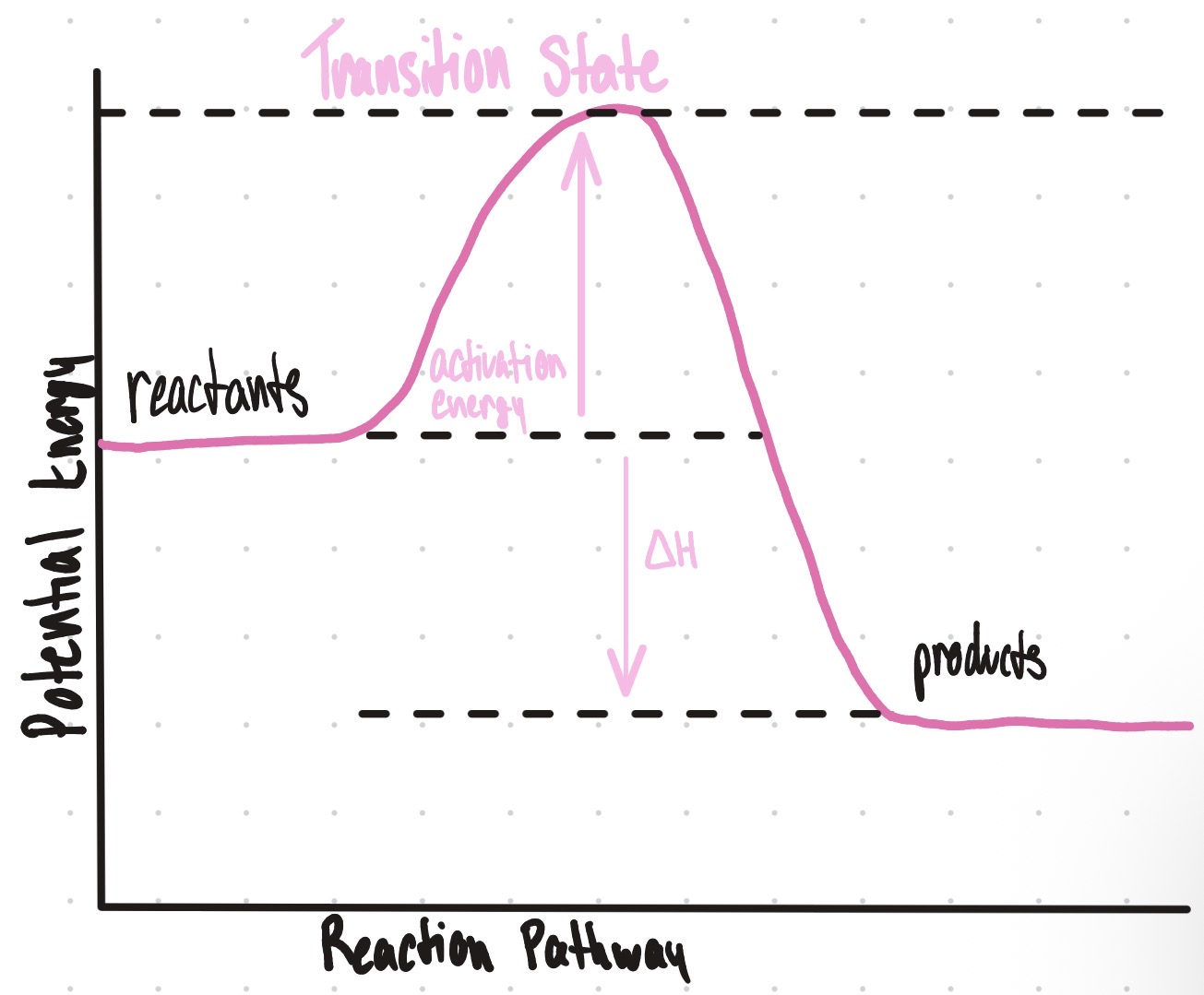
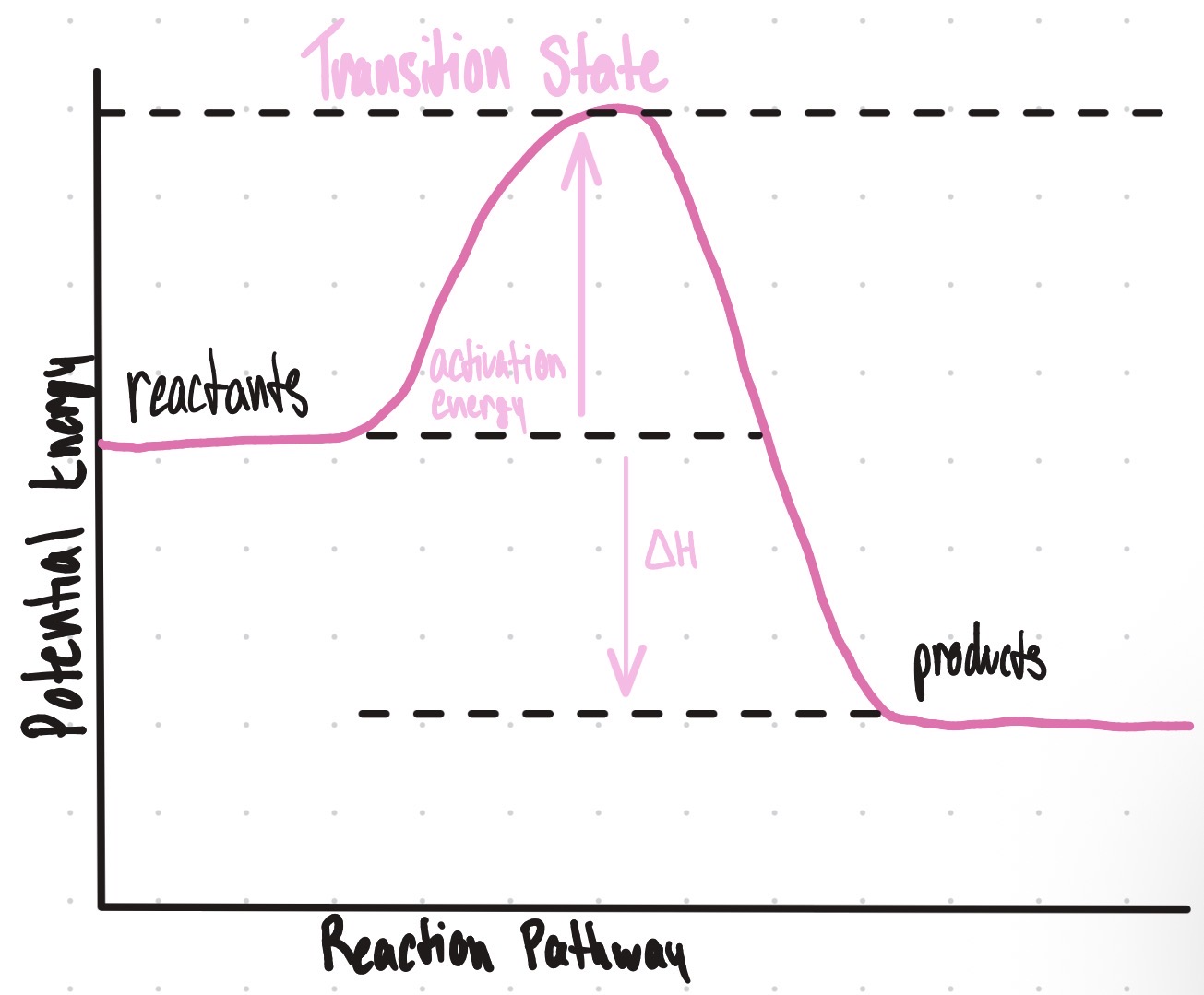
Is this an endothermic or exothermic reaction?
Endothermic
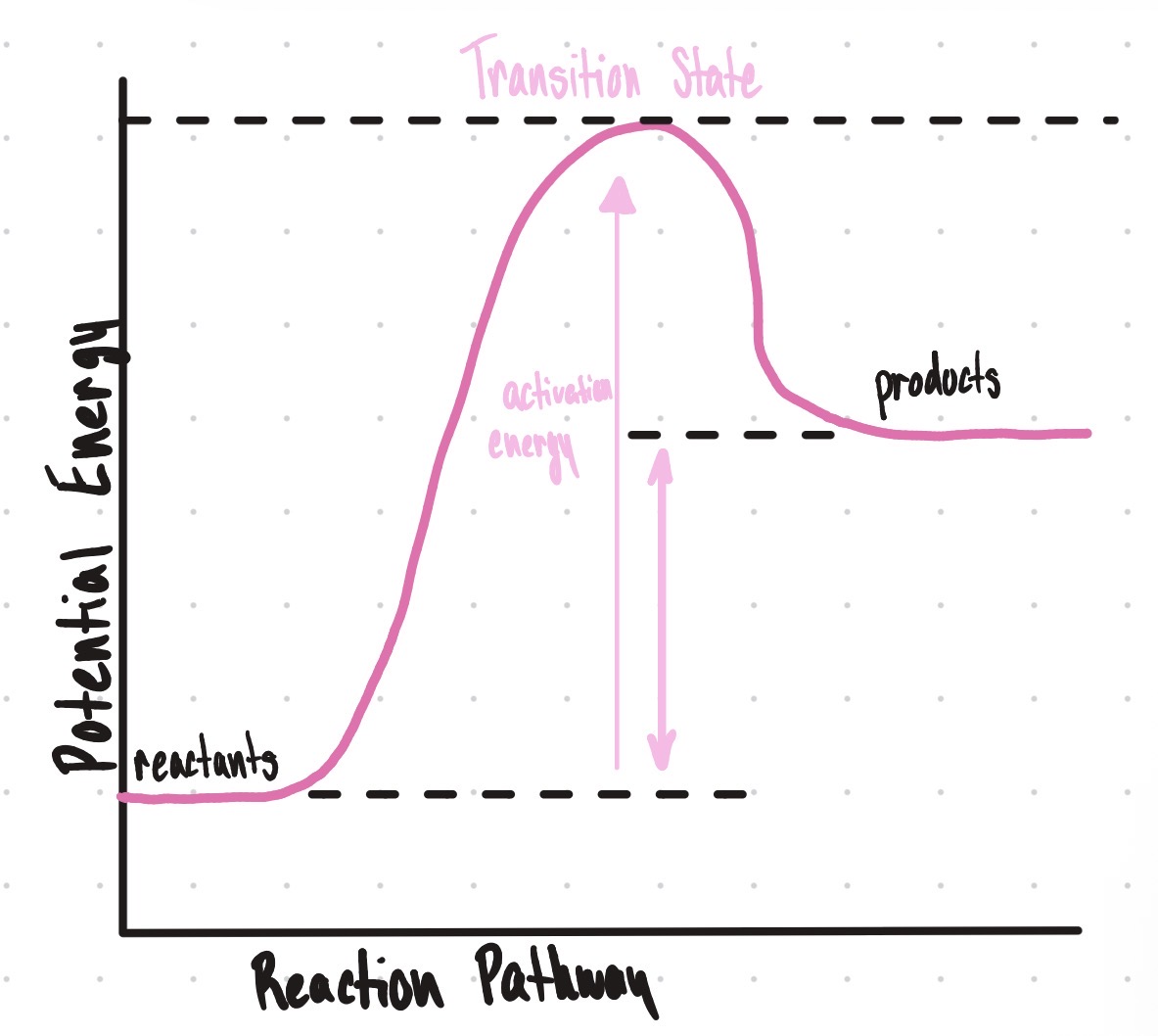
Is this an endothermic or exothermic reaction?
Exothermic
Why does adding heat increase the rate of a reaction?
Because it causes more frequent collisions among molecules
Define all the symbols in the Arrhenius equation (k = A * e^(-Ea/RT)) and show how it can be arranged into the equation of a straight line. Give an expression for the slope. Why does this plot always have a negative slope?
k = rate constant; A = frequency factor; Ea = activation energy (J/mol); R = gas constant (8.314 J/mol·K); T = temperature (K)
y= mx + b; ln k = (-Ea/R) * (1/T) + ln A
The plot always has a negative slope because increasing temperature lowers 1/T,
increasing k.
Show how the Arrhenius equation can be used to calculate the energy of activation of a reaction provided rate constants are known at two different temperatures.
Using the Arrhenius equation: ln(k2/k1) = (-Ea/R) * (1/T2 - 1/T1)
Rearranging for Ea: Ea = -R * (ln(k2/k1)) / (1/T2 - 1/T1)
This equation allows for the calculation of activation energy if rate constants are known at
two temperatures.
A study of the decomposition of ethylene oxide, C2H4O(g) → CH4(g) + CO(g)
yields a rate constant of 1.2x10-5s-1 at 650 K and a value of 2.19x10-11s-1 at 500 K. Calculate the energy of activation for this reaction.
238 kJ/mol
The rate of a particular reaction went from 3.00 M/s to 4.35 M/s when the temperature was raised from 18 ºC to 30 ºC. What is the activation energy of the reaction in kJ/mol?
22.7 kJ/mol
Describe what is meant by the molecularity of a reaction.
Molecularity refers to the number of reactant molecules involved in an elementary step.
Provide an example of a unimolecular and a bimolecular elementary reaction.
Unimolecular: N2O → N2 + O); Bimolecular: NO2 + CO → NO + CO2
temperature is a measure of
average kinetic energy of the molecules in a sample
What does an increase in temperature look like on a maxwell-boltzmann distributions curve?
the curve flattens and broadens
What experimental result provides information about the molecularity of a reaction mechanism ?
The rate law
Activation energy is always (positive/negative)
positive
Define reaction mechanism
A sequence of elementary steps describing the overall reaction