CHEM FINAL
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
What is “Ground State”?
No energy has been absorbed
What is “Excited State”?
When energy has been absorbed.
What are valence electrons?
Electrons in the outermost principal energy level.
What are core electrons?
Electrons in the innermost principal energy level.
What are orbitals?
A region of space where there is greater than 90% probability of finding an electron.
What is an atomic radius?
The average distance from the nucleus to the outermost electron.
On the periodic table, as you go from left to right, atomic radius ______ in the rows and _____ in the columns.
Decreases and increases.
What is ionization energy?
The energy required to remove an electron from an isolated ground stable atom in the gas phase.
On the periodic table, as you go from left to right, ionization energy ______ in the rows and _____ in the columns.
Increases, decreases
What is the octet rule?
Atoms tend to react in such a way as to get eight electrons in their valence shell.
How many bonds does hydrogen form?
1 covalent bond
How many bonds do halogens form?
1 covalent bond.
How many bonds does oxygen like to form?
2 covalent bonds.
How many bonds does nitrogen form?
3 covalent bonds.
How many bonds does carbon like to form?
4 covalent bonds.
What can 2 bonds be?
Either 2 single or 1 double bond.
What can 3 bonds be?
Either 3 single, 1 triple, or 1 single+1 double
What can 4 bonds be?
Either 4 single, 2 double, 1 triple+1 single, 1 double+2 single
Carbon is the closest to absolute. True or False.
True.
What is electronegativity?
The tendency of an atom to pull the electrons in a covalent bond towards itself.
What is bond energy (bond strength)?
The energy required to break a bond. Or the energy required when a bond form.
The breaking of a bond _____ energy, but does not _____ it.
requires, release
What are bond lengths?
The average distance between nuclei of atoms forming a bond.
On the periodic table, as you go from left to right, electronegativity ______ in the rows and _____ in the columns.
Increases, decreases
Bond Energy increases how?
Single < Double < Triple
Bond Length increases how?
Triple < Double < Single
Formal Charge equation?
FC = (# of valence electrons on an atom) - (# of non-bonding electrons on the atom) - ½ (# bond electrons)
What is the Valence Shell Electron Pair Repulsion (VSEPR)?
The valence electron pairs surrounding an atom tend to repel each other.
What is valence bond theory?
A covalent bond is the result of the overlap of atomic orbitals.
There are two types of covalent bonds; they are?
Sigma bond
Pi bond
What are sigma bonds?
Direct overlap of orbitals between two atoms.
All single bonds are ____ bonds.
Sigma bonds.
What are pi bonds?
Side-ways overlap of p orbitals.
Double bonds consist of 1 ____ bond and 1 ____ bond?
sigma bond, pi bond.
Triple bonds consist of 1 _____ bond and 2 ____ bonds?
sigma bond, pi bond
What are hybrid orbitals?
Orbitals that are the result of combining two or more of the basic orbitals.
How many electrons can the s sublevel hold?
2
How many electrons can the p sublevel hold?
6
How many electrons can the d sublevel hold?
10
How many electrons can the f sublevel hold?
14

What kind of orbital is this?
S orbital

What kind of orbital is this?
P orbital

What kind of orbital is this?
D orbital

What kind of orbital is this?
F orbital
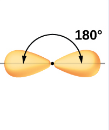
What kind of electron pair geometry is this?
Linear.
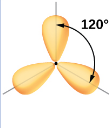
What kind of electron pair geometry is this?
Trigonal planar
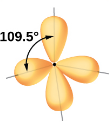
What kind of electron pair geometry is this?
Tetrahedral
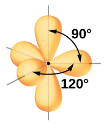
What kind of electron pair geometry is this?
Trigonal bipyramidal
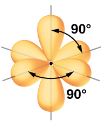
What kind of electron pair geometry is this?
Octahedral
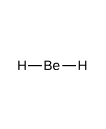
What kind of electron pair geometry is this?
Linear
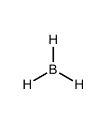
What kind of electron pair geometry is this?
Trigonal planar
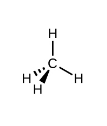
What kind of electron pair geometry is this?
Tetrahedral
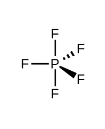
What kind of electron pair geometry is this?
Trigonal bipyramidal
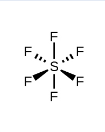
What kind of electron pair geometry is this?
Octahedral
In linear electron pair geometry, what are the number of regions and what is its angle?
Two regions of high electron density
180 degrees
In trigonal planar electron pair geometry, what are the number of regions and what is its angle?
Three regions of high electron density
120 degrees
In tetrahedral electron pair geometry, what are the number of regions and what is its angle?
Four regions of high electron density
109.5 degrees
In trigonal bipyramidal electron pair geometry, what are the number of regions and what is its angle?
Five regions of high electron density
Angles of 90 degrees or 120 degrees
In octahedral electron pair geometry, what are the number of regions and what is its angle?
Six regions of high electron density
90 degrees or 180 degrees.
How many valence electrons do non-metals want? What is the exception?
8 valence electrons, besides hydrogen which wants 2.
1 line between two atoms = ?
single bond, 2 electrons
2 lines between an atom = ?
double bond, 4 electrons
2 dots in a lewis diagram are called a ____ pair of electrons.
lone pair.
What are the four properties of a gas?
Pressure
Temperature
Volume
Amount (moles)
All gases behave the same. (True or False)
True.
Equation for pressure?
Pressure = Force / Area
What is a barometer?
The first device used to measure pressure.
1 atm = ?? mmHg = ?? torr
760 for both
Pressure is inversely proportion (if one goes up, the other goes down) to volume (true or false).
True
Temperature is inversely proportion (if one goes up, the other goes down) to volume (true or false).
True