Ch. 27: Quantum Optics
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
true or false: nothing can go faster than the speed of light
true! c=3×10^8 m/s
classical physics: 3 major fields
Three major fields of physics in 1900 (Classical Physics=before 1900).
-Mechanics, Electromagnetism (including Optics), Thermodynamics
-The conventional view at the time was that light was completely described when it is treated as a wave. (*sometimes treat as particle)
*”quantam”=small, only discrete values (“quantized”)
classical physics: 5 outstanding issues
There were five famous outstanding issues that couldn’t be described by any of the above fields
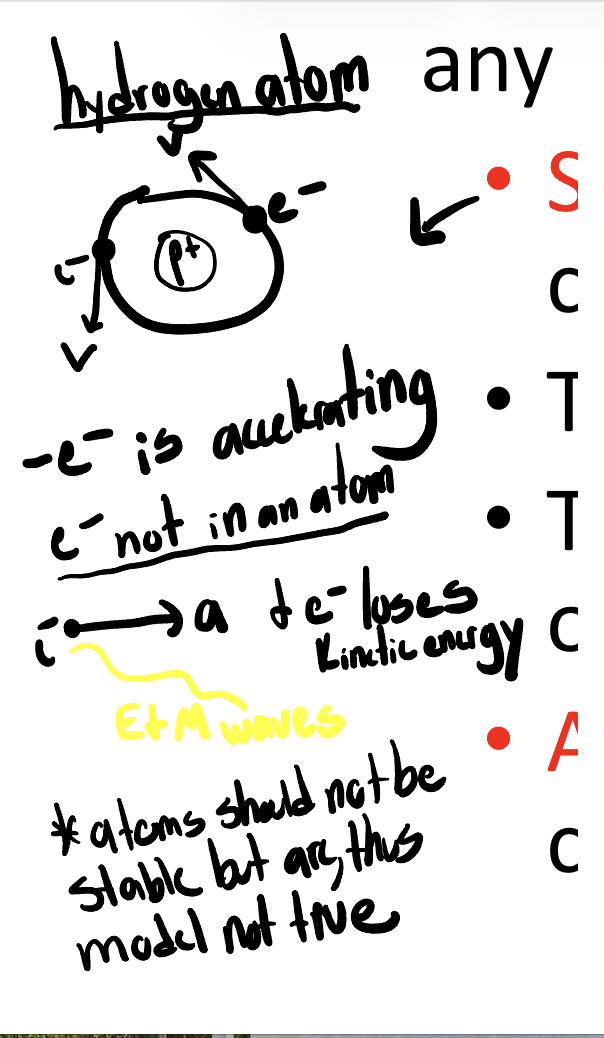
-Stability of atoms against the in-spiral of electrons predicted by classical Electromagnetism
-The nature of blackbody spectra (*all objects emit EM waves, depends on object temp)
-The photoelectric effect involving incident light knocking electrons out of a metal (*x-rays hit metal and knock out electron; basis of digital camera/absorb light)
-Atomic spectral lines indicated that excited atoms give off a discrete characteristic spectrum of light (*ex=red superimposed multiple other colors)
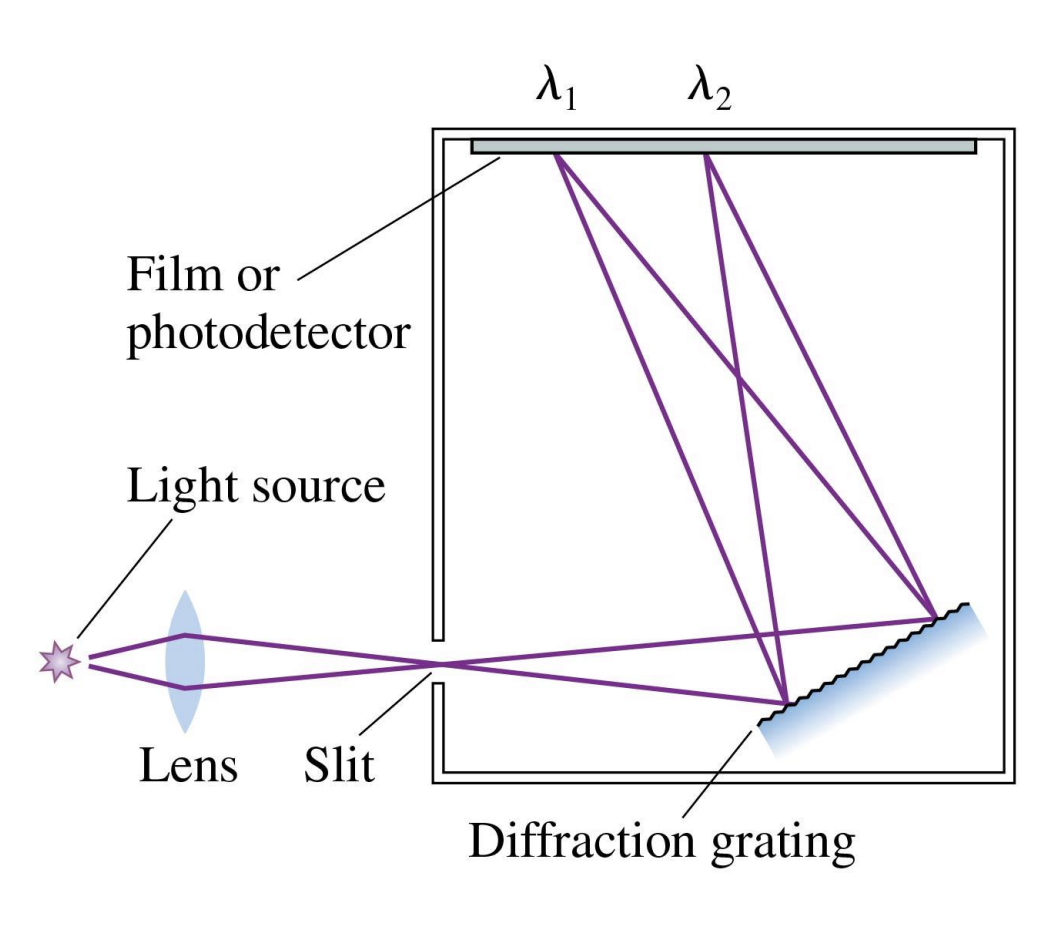
the spectrometer
-Making the diffraction grating slightly curved focuses the spectrum onto the photodetector
*spectra=range of wavelengths, fancy diffraction gradient
continuous spectra
-A hot, self-luminous object, such as the filament of an incandescent lightbulb, forms a rainbow-like continuous spectrum in which light is emitted at every possible wavelength
no gaps/no missing wavelengths (see all parts of spectrum)
-ex: lightbulbs, white lights
blackbody spectra
An object with surface area A and absolute temperature T radiates or absorbs heat energy at the rate: Prad=sigma*A*T^4 (*sigma=constant; A=SA of object; T=temperature in Kelvin!)
where σ = 5.67 × 10–8 W/(m^2 ⋅K^4) is the Stefan-Boltzmann constant
-A perfect absorber or emitter is called a blackbody, and the thermal radiation emitted by a blackbody is called blackbody radiation. Example: charcoal, the sun
-we will model objects as blackbodies
*all EM rays absorbed also get re-edmitted; anything w/ temp emits; ex=SA person, temp person in K=energy/sec

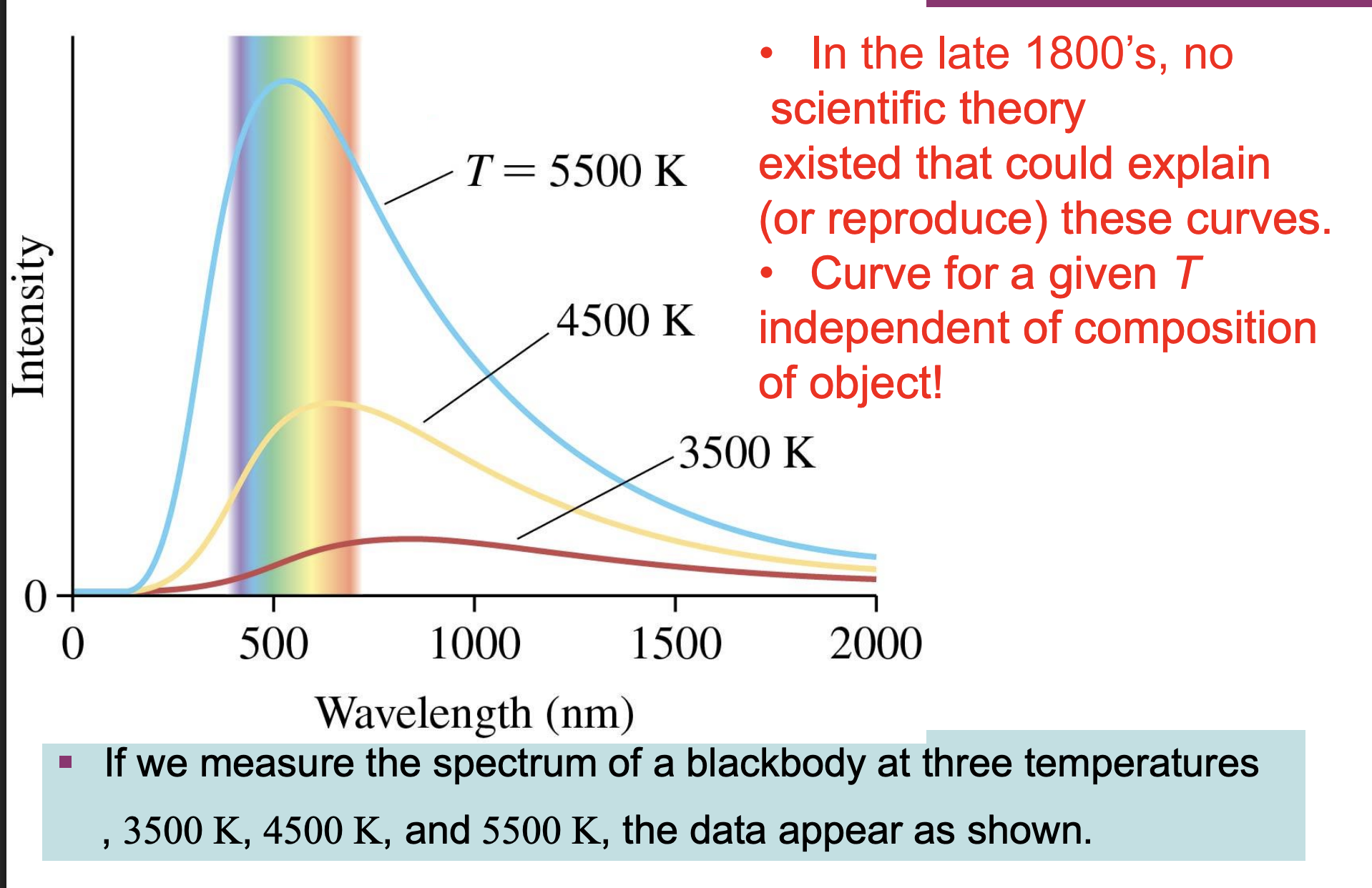
blackbody spectra graph
-In the late 1800’s, no scientific theory existed that could explain (or reproduce) these curves (*had no equation)
-Curve for a given T independent of composition of object! (*same temp has same curve though dif compositions)
*lower temp=lower peak
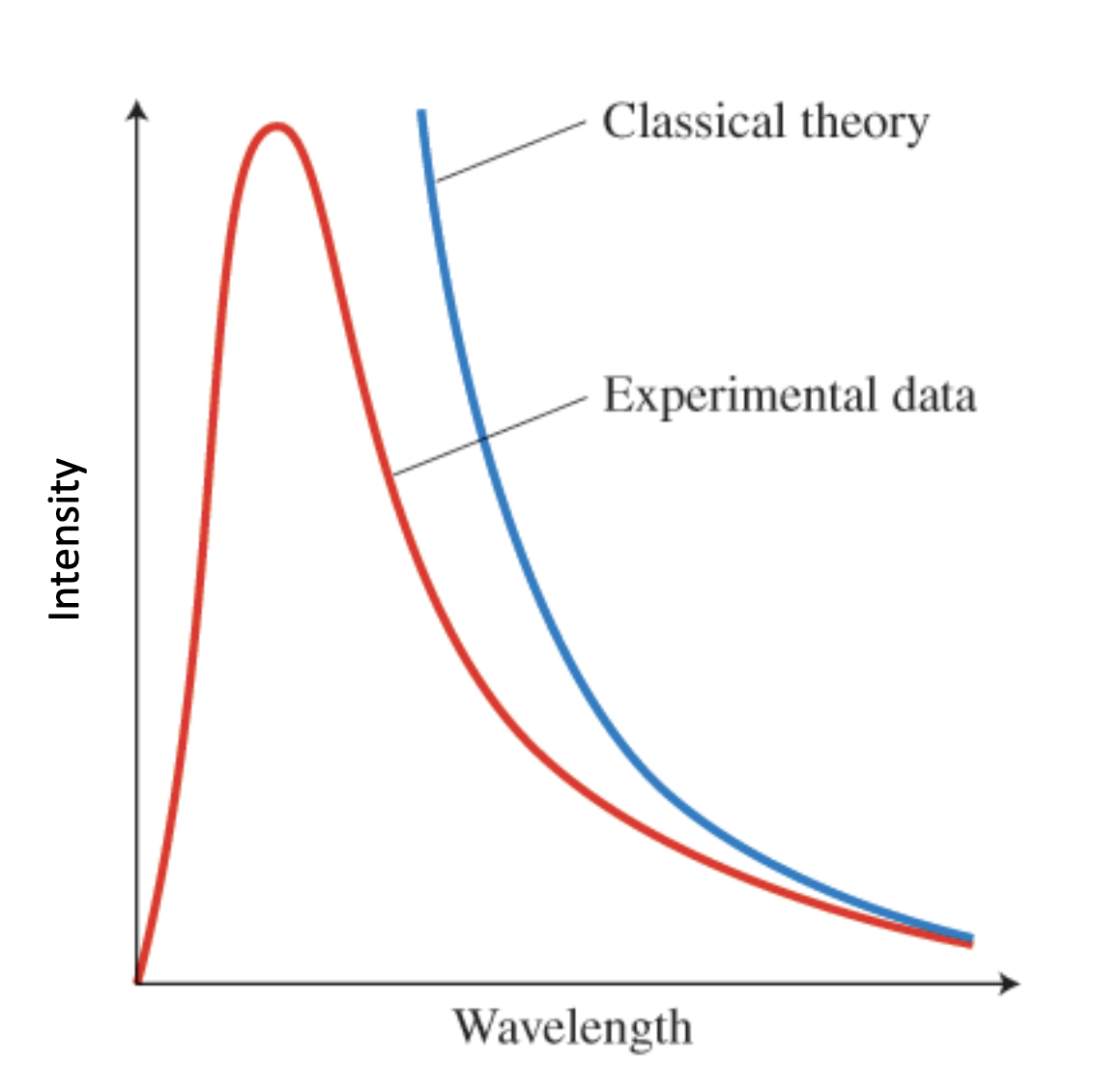
classical theory vs experimental data
-classical theory: lower wavelengths=higher EM waves (doesn’t make sense); how get to match experimental?
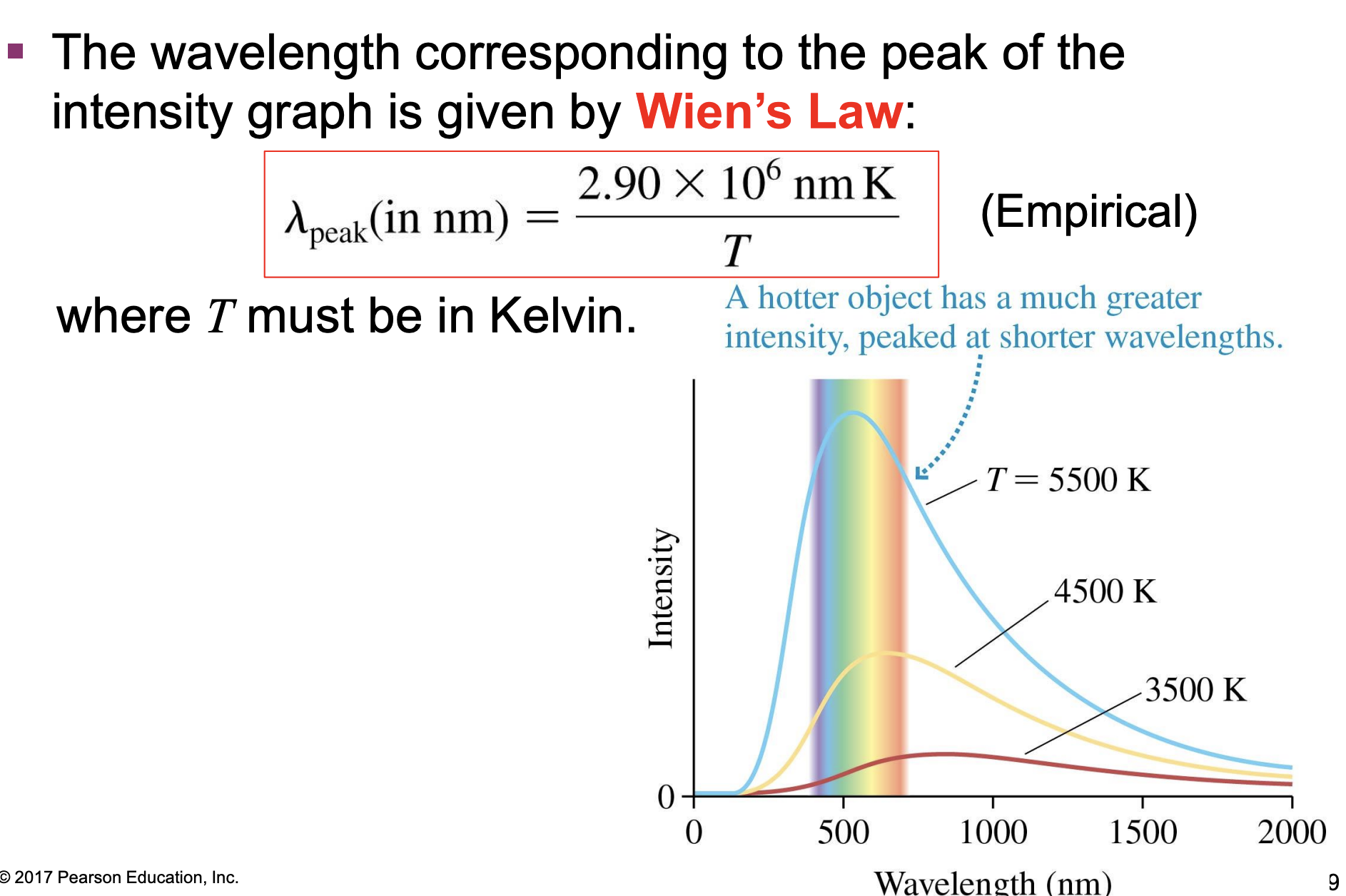
blackbody spectra: Wien’s Law
-The wavelength corresponding to the peak of the intensity graph is given by Wien’s Law: (picture); where T must be in Kelvin
-empirical=based on data
*body=mostly infared rays
why is the sun not green, though the wavelength that is most intensely emitted from the sun is green?
-green is just the peak wavelength, it edmits other colors too and the colors overlap so looks orange
*similar example: peak wavelength person=infrared, but we can see each other

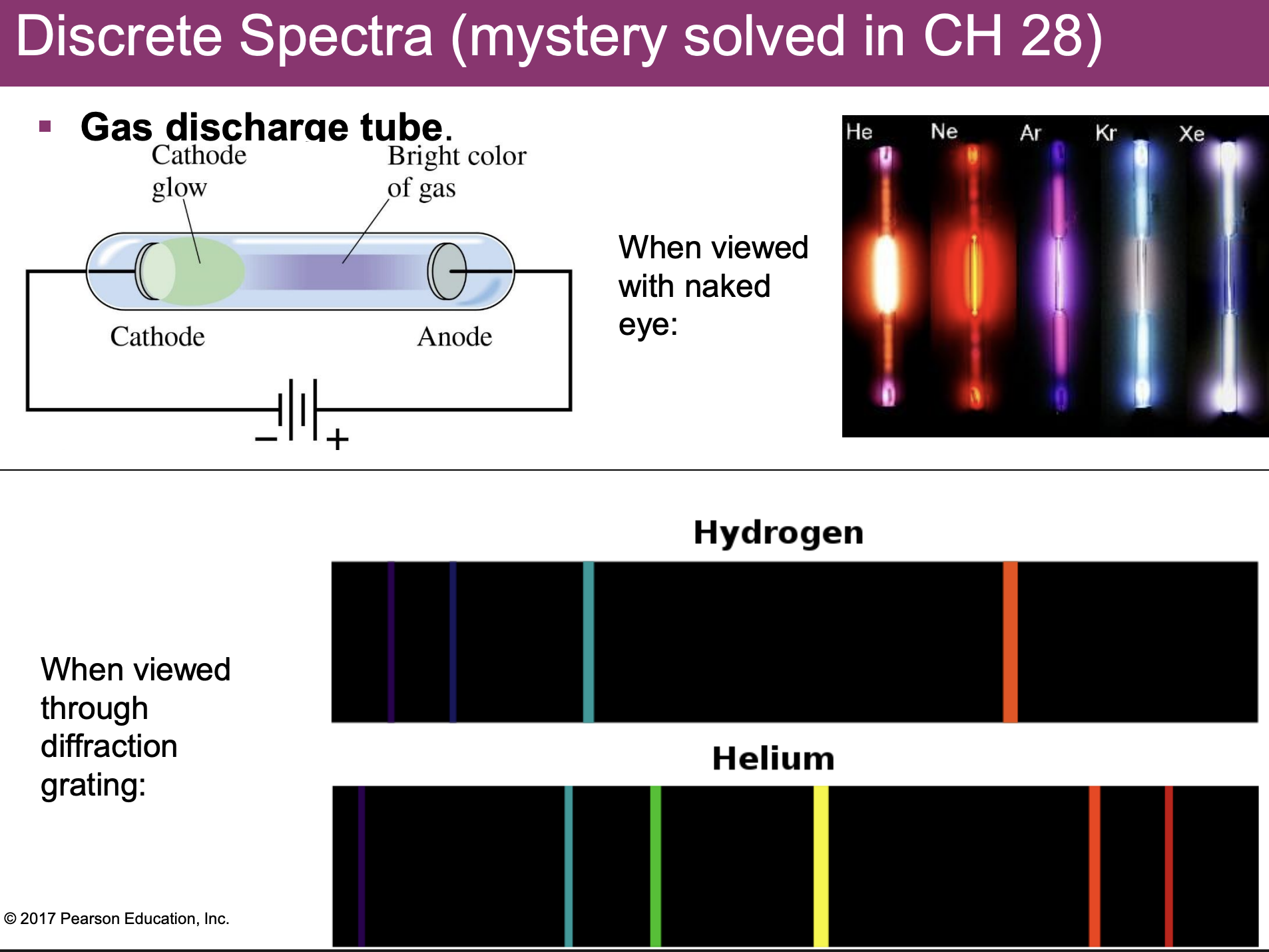
discrete spectra
-when view through diffraction grating: not a single color, but multiple colors that overlap; each has a specific wavelength
-every element makes its own signature spectrum, bc dif #’s of electrons
-Gas Discharge Tube: large potential difference so large electric current through gas, warms and glows
*top picture=what can wee w/ your eyes
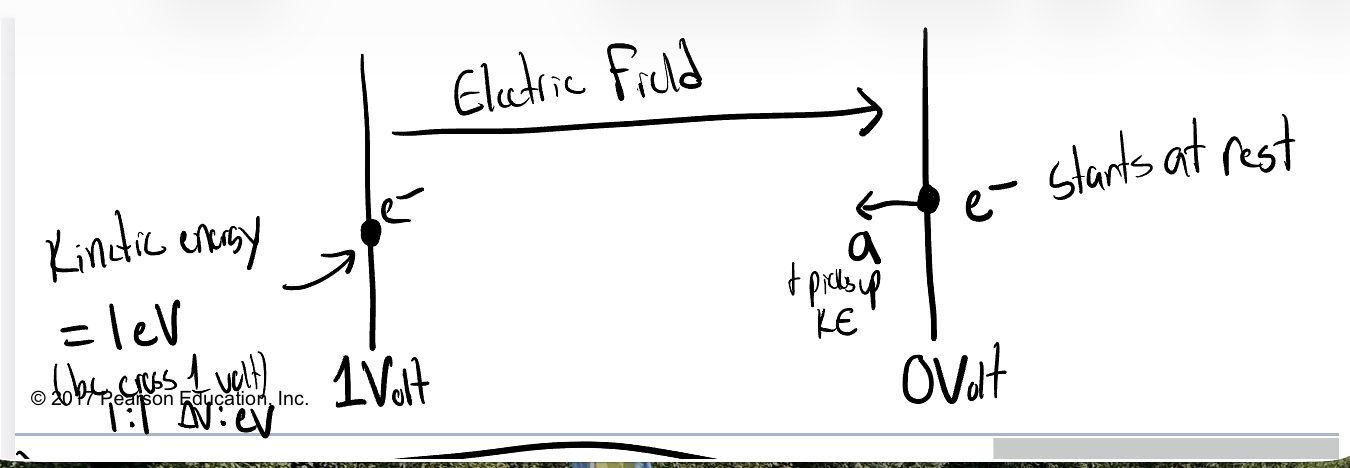
electron-volt
1eV = 1.6x10-19 J
-One electron-volt (eV) is the kinetic energy gained by an electron accelerated through a potential difference of 1 volt
*when deal w/ particles, bc convient; eV=unit; potential difference creates an electric field, and the electron that starts at rest accelerats and picks up kinetic energy; 1:1 change in voltgs:eV