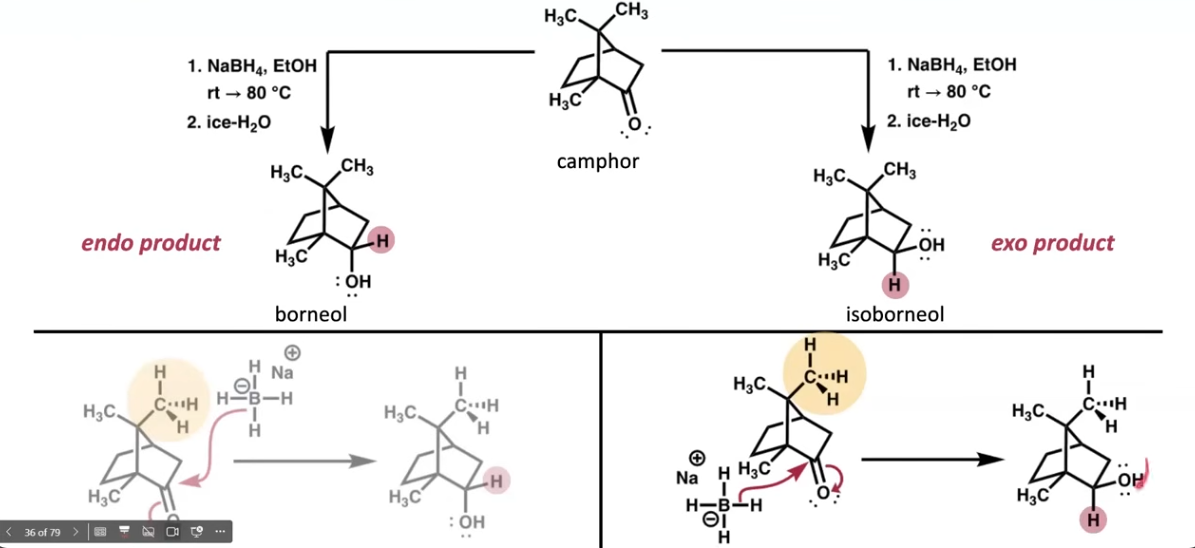
Borohydride Reduction of Camphor
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Reduction
Reduction is the gain of electron density on a carbon
Reduction is generally considered to be the addition of hydrogen to an organic molecule or by the breaking of a bond to an electronegative element (O, N, halogen)
What can often be used to perform a reduction?
Hydrogen gas, an appropriate catalyst, and perhaps some heat/pressure
Oxidation
Oxidation is the loss of electron density on a carbon
Removing a hydrogen
Ex: alcohol to ketone or aldehyde to carboxylic acid
Formation is C-O, C-N, or C-X
What metal hydride agents can be used to reduce carbonyl compounds?
Lithium aluminum hydride, LiAlH4 (LAH), and sodium borohydride, NaBH4
Lithium aluminum hydride, LiAlH4 (LAH)
Hydride (H-) is added to the carbonyl
Very powerful, indiscriminate reduction
Sodium borohydride, NaBH4
Hydride (H-) is added to the carbonyl
Mild, only reduced aldehydes and ketones
How is the carbonyl carbon reduced?
The carbonyl carbon is reduced when the hydride ion adds to the carbon which breaks the carbon–oxygen π straight pi-bond and forms a new carbon–hydrogen bond.
Can H- exist as an isolated charged particle in solution
No
Like a proton, a hydride ion, H–, does not exist as an isolated charged particle in solution
Why must scrupulously dried aprotic solvents be used with LAH?
LAH reacts violently with any proton source, such as water or alcohol, to liberate hydrogen gas. This reaction is frequently exothermic enough to ignite the escaping hydrogen. Therefore, only scrupulously dried aprotic solvents like diethyl ether or tetrahydrofuran, THF, are used with LAH.
Why must LAH reactions be quenched by the addition of an ester (ethyl acetate is commonly used) or a hindered alcohol?
You quench LAH with a mild substance first so it doesn’t react violently when you finally add water.
What solvent can sodium borohydride be used in? What conditions must be met in order for this to work?
Sodium borohydride is mild mannered enough that it may be used in protic solvents like ethanol and water. It does, however, react slowly with these solvents and must be used in excess.
Why are metal hydride reducing agents always used in excess?
Why we use excess metal hydride (like LAH or NaBH₄):
To make sure all the reactant reacts
You want all the carbonyl → alcohol.
Extra reagent helps guarantee nothing is left unreacted.
The reaction slows down as it goes
After each hydride transfer, the reagent gets bulkier (crowded).
This makes it harder for it to reach the next carbonyl.
Extra reagent = keeps things moving faster.
Some reagent is “wasted”
These reagents react with water in the air.
Even tiny amounts of moisture can destroy some of it.
Solvent can also consume it
For example, sodium borohydride slowly reacts with alcohol solvents.
So not all of it is available for your main reaction.
Purity isn’t perfect
The reagent you weigh out might already be partially used up.
What is an epimer?
Epimers are molecules that differ at only one chiral (stereocenter) while all the other stereocenters are the same.
How to calculate percent of borneol in product:
% borneol in product= (Integration of borneol)/(Integration of borneol + isoborneol)
Recrystallization
Recrystallization is the process of dissolving a solid material into a suitable solvent or mixture of solvents and allowing the material to crystallize (actually, recrystallize, since you normally begin with impure crystalline materials) from this solution
Purity of Recrystallization
Generally, molecules of a single (pure) compound tend to form the best crystal lattice, so a solid that crystallizes from the solution has fewer impurities.
This process can be repeated, with each recrystallization ideally yielding a more purified product.
Realize that if purification has occurred, most, if not all, of the impurities will be left in the solution, as will (regrettably) some of your desired product. Put simply, you should recover less material than you started with—otherwise no purification took place.
Important points to remember about your choice of a recrystallization solvent:
The solvent should not chemically react with the compound. (This should be obvious; remember, you want the purified compound back as itself when you’re done.)
The solvent should dissolve the compound while hot.
This same solvent should not dissolve the compound when cold.
The solvent should either dissolve the impurities at all temperatures or not dissolve the impurities at all. (Remember, you want to remove the impurities.)
Trick for a mixed-solvent system for recrystallization:
The key ingredient here is finding a pair of solvents—one which readily dissolves the compound and the other in which the compound is totally insoluble. Oh yes, these two solvents must be completely miscible—or totally soluble in each other—at all concentrations. Alcohol/water, alcohol/dichloromethane, and hexane/ethanol are all common mixed solvent systems.
Diastereotopic protons
Diastereotopic protons are two hydrogens on the same carbon that are not equivalent because the molecule has some asymmetry.
Purpose of isolation in this reaction:
Isolation is done to separate and collect pure borneol/isoborneol from all the leftover chemicals in the reaction mixture.
Purpose of Recrystallization in this reaction:
Recrystallization cleans a solid by letting only the pure compound crystallize while impurities are left behind in solution.
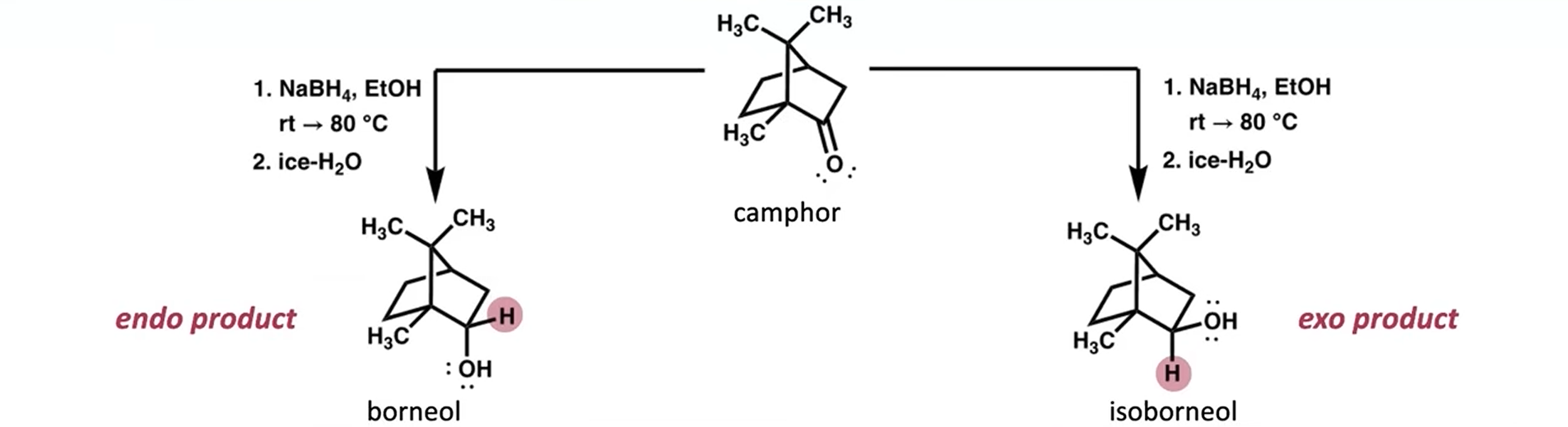
Draw the mechanism for both outcomes: