Hydrogen bonding
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
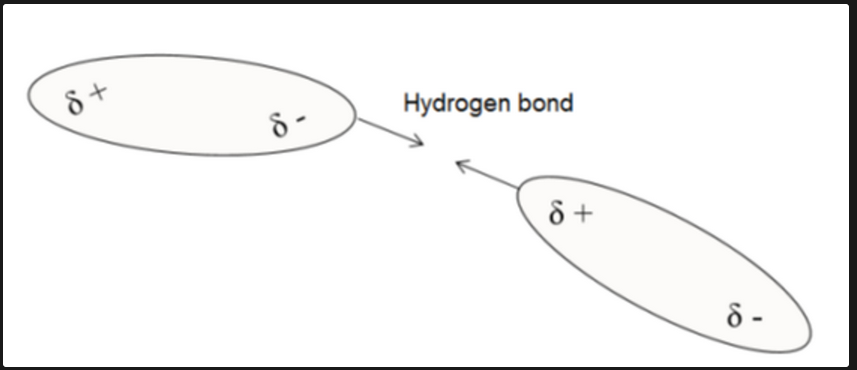
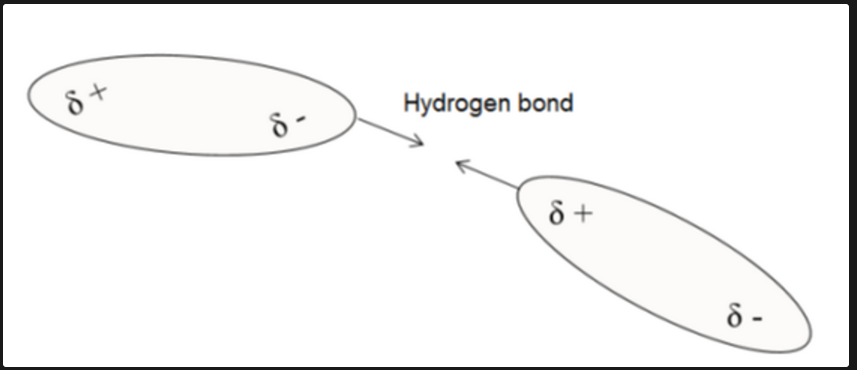
Hydrogen Bonding
A type of intermolecular force where polar molecules are attracted to each other, forming weak but important bonds.
Intermolecular Forces
Forces that occur between molecules, such as hydrogen bonds.
Covalent Bond
A strong bond formed by the sharing of electrons between atoms, different from hydrogen bonds.
Polar Molecules
Molecules that have a slight positive charge on one end and a slight negative charge on the other, allowing for hydrogen bonding.
Hydrogen Bond Representation
Represented with dashed lines (////) to signify attraction between molecules.
Requirements for Hydrogen Bonding
Must occur between two polar molecules containing O-H, F-H, or N-H bonds.
Role of Hydrogen Bonds in DNA
Hydrogen bonds hold the base pairs together, stabilizing the DNA structure.
Role of Hydrogen Bonds in Proteins
Help maintain the 3D functional shapes of proteins.
High Latent Heat
The energy required to change the state of a substance without increasing its temperature, such as melting ice or boiling water.
Latent Heat Energy in the Body
Evaporates water from sweat, helping to cool the body.
High Specific Heat
The amount of energy required to change the temperature of a substance, indicating how much energy is needed to raise the temperature.
High Surface Tension
Water's tendency to behave as if it has a skin on its surface, contracting to the smallest possible area.
Surfactant
A substance that reduces surface tension, necessary for smooth lung function and oxygen transfer into the bloodstream.
Importance of Surfactants
Essential for premature babies, as they are the last to develop, affecting their ability to breathe.
Water Cohesion
Water molecules' tendency to stick to themselves, contributing to high surface tension.