chemcial stability 2
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
What is oxidation?
Addition of oxygen
Removal of hydrogen
Loss of electrons
What is reduction?
Removal of oxygen
Addition of hydrogen
Gain of electrons
Why is autooxidation important in pharmaceuticals?
Most important oxidation process in drugs
Molecular oxygen (O₂) usually the source of oxidation
What makes autooxidation reactions complex?
Involve multiple reactive oxygen species (ROS)
Reactions often multi-step and unpredictable
What ROS are produced in autooxidation?
Superoxide anion (O₂⁻·)
Hydrogen peroxide (H₂O₂)
Hydroxyl radical (HO·)
Singlet oxygen (¹O₂)
What is required for free radical-mediated autooxidation?
Involvement of light or redox-active metal ions
Common metals: Fe(II)/Fe(III), Cu(I)/Cu(II)
What are the 3 key requirements for ROS chemistry in vitro?
Transition metal ion
Good chelating ligand
Reducing agent
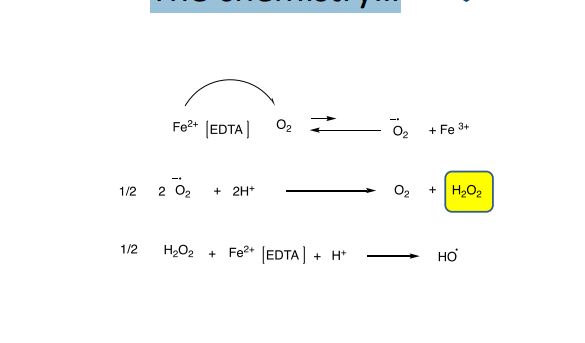
What is the Fenton reaction?
Reactants: H₂O₂ + Fe(II)
Generates hydroxyl radicals (HO·)
Purpose: produces reactive oxygen species (free radicals)
What is the Udenfriend reaction?
Reactants: Fe(II), ascorbic acid, EDTA, air
Complex multistep process → includes a Fenton-type step
Produces reactive oxygen species / free radicals
The chemistry…
How can agents like polyvinylpyrrolidone (povidone) and polyethylene glycol (PEG) degrade?
They can undergo autocatalytic degradation
This means they can reduce hydrogen peroxide by themselves
This process can cause their self-degradation without needing external catalysts or radicals
Udenfriend reaction – requirements & substitutes
Metal ion: Fe²⁺ → Cu⁺, Sn²⁺, Co²⁺, Ti²⁺
Chelator: EDTA → citrate, DTPA, pyrophosphate, drugs
Reducing agent: Ascorbic acid → phenols, bisulfite, catechols
Note: Any metal-chelating drug can react
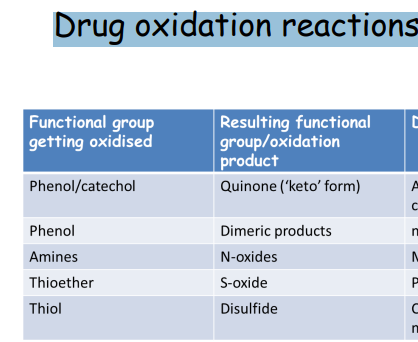
: Common drug oxidation reactions – functional groups and products
Phenol/catechol → Quinones (keto form)
Phenol → Dimeric products
Amines → N-oxides
Thioether → S-oxide
Thiol → Disulfide
Difference between homolytic and heterolytic bond cleavage?
Homolytic: bond breaks evenly → each atom gets 1 electron → forms radicals (unpaired electrons)
Heterolytic: bond breaks unevenly → one atom gets both electrons → forms ions (cation + anion)
What happens in carbon radical formation by homolysis?
Breaking C–Br bond evenly → carbon radical + Br· radical
Carbon radical has 7 valence electrons (one unpaired)
Carbon radical is planar, sp² hybridized
Compare to carbocation (6 valence electrons, positive charge)
Which radicals are more stable: methyl, primary, secondary, or tertiary?
Tertiary radicals are most stable
Stability order: tertiary > secondary > primary > methyl
Why tertiary is most stable:
More alkyl groups donate electrons (electron donation via hyperconjugation)
Better delocalization of the unpaired electron
Steric hindrance reduces unwanted reactions, stabilizing the radical
Which molecules are prone to C–H oxidation? Why?
Ethers, aliphatic amines, aldehydes
Why: These have C–H bonds next to heteroatoms or functional groups that weaken the bond, making hydrogen easier to remove during oxidation.
Which molecules are prone to O–H and N–H oxidation? Why?
Phenols (O–H), aromatic amines (N–H)
Why: The O–H and N–H bonds are reactive sites that can lose hydrogen easily, leading to oxidation products. Aromatic rings stabilize radicals formed after oxidation.
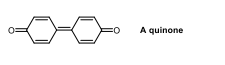
a quiunone sructure
Why are drugs containing phenolic groups often formulated at low pH?
At low pH, phenolic groups stay in their neutral form rather than forming the phenoxide ion.
The phenoxide ion has higher electron density, making it more reactive and more easily oxidised.
The phenoxide ion is more susceptible to oxidation, so keeping the pH low helps improve drug stability.
phenols are weak acids
Why are many basic drugs (e.g. aromatic amines) formulated as their salts?
Aromatic aminest.he nitrogen lone pair can partially delocalize into the ring, onates electron density into the ringmaking them basic.
When protonated (forming a salt), the lone pair is no longer available electron density on nitrogen drops → nucleophilicity decreases.
Salt formation also increases solubility in water, improving drug absorption and stability.
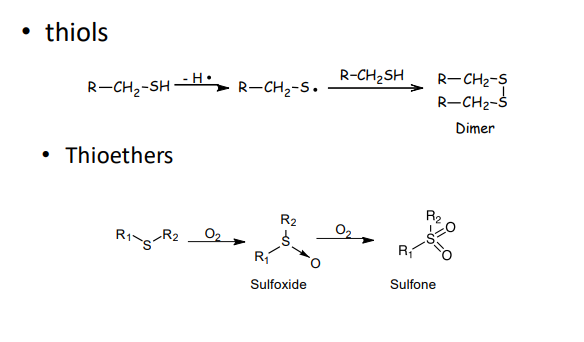
If we have oxidation of SH bonds what do we get?
thiols
thioesters
thiols=dimer
thioesters= sulfoxide + sullfone
oxidation happes in a chain reaction what are the three things?
initiation
propgation
termiantion
How can oxidation of drugs be minimised?4
all these prevent the iniitation
Protect from light
Limit peroxides (prevent initiation)
Use optimum pH:
Acidic drugs → degrade faster when ionised (high pH)
Basic drugs → more stable in acidic pH
Add chelating agents (e.g. EDTA, citric acid)
→ remove metal ions that promote oxidation
How do we stop propagation?
Exclude oxygen
Pack with nitrogen
Use tablet strips
Add antioxidants
Ascorbic acid → sacrificial
BHT → radical trap (stops chain reaction)
Two ways radicals are produced
Initiation (main way)
External factors create radicals
e.g. light, heat, metal ions
Starts the chain reaction
From peroxides (e.g. H₂O₂)
Peroxides can break down to form radicals (•OH)
Then enter radical chain reactions
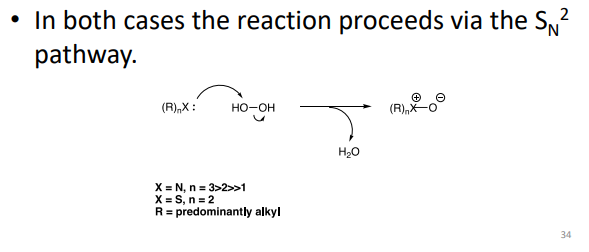
How do peroxides (e.g. H₂O₂) cause oxidation?
1. Radical pathway
H₂O₂ → forms •OH radicals
→ chain reaction (propagation)
2. Non-radical pathway
Directly oxidises amines & sulfides
→ SN2 mechanism (no radicals)
→ Peroxides can cause oxidation by both radical and non-radical mechanisms
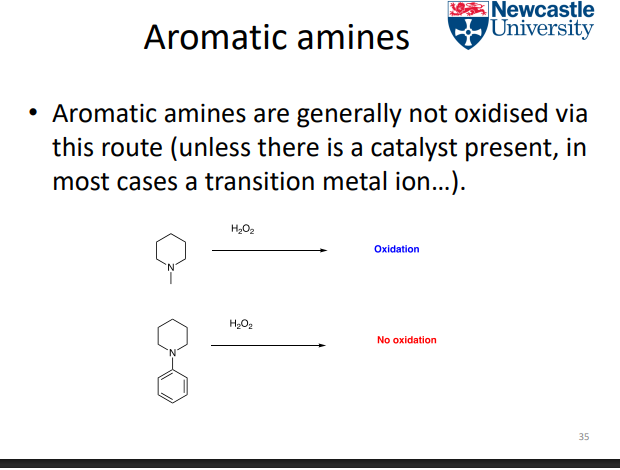
Why are aliphatic amines more easily oxidised than aromatic amines?
Aliphatic amines:
Lone pair localised on N
→ High electron density
→ More nucleophilic → more oxidation
Aromatic amines:
Lone pair delocalised into ring
→ Less available → less oxidation
How does pKa affect oxidation of amines?
Higher pKa = more basic
→ Lone pair more available
→ More susceptible to oxidation
How can oxidation of amines be prevented?
Convert to HCl salt (protonated form)
→ No lone pair on N
→ No nucleophilicity → no oxidation
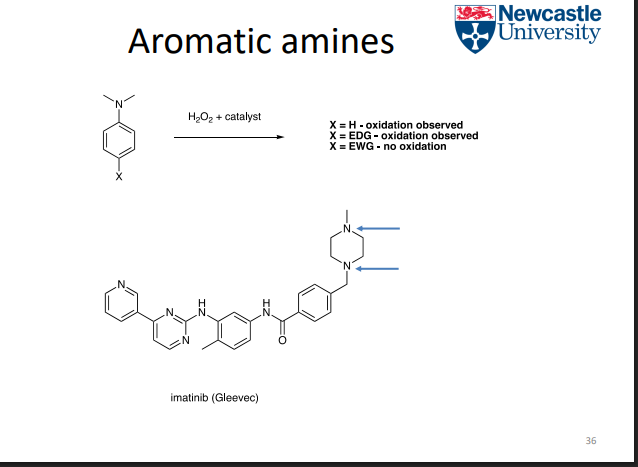
Aroamtic amines efefcts ofthe susbsutuient
Reactions of H₂O₂ with other functional groups
Carboxylic acids (RCOOH) → Carboxylic peracids (RCO₃H)
Bicarbonate (HCO₃⁻) → Peroxymonocarbonate (CO₄H)
Nitriles (R–C≡N) → Peroxycarbimidic acids
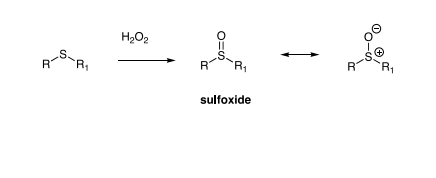
Oxidation of thioethers
Thioethers (R–S–R′) react with H₂O₂ or other peroxides.
Mechanism: Nucleophilic attack by sulfur on the peroxide.
Product: Sulfoxide (R–S(=O)–R′)